Pure Chem-Atoms+Elements+Compounds+Isotopes+Ions+Ar
Elements
-An element is a pure substance made up of only one type of atom. (e.g. oxygen, carbon)
-Represented by chemical symbols like “O” for Oxygen or “H” for Hydrogen
-About 118 known elements
-Cannot be broken down into simpler substances
Compound
-Two or more different elements chemically bonded in a fixed ratio. (e.g. H2O, CO2)
-Represented by chemical formulas such as H2O for water or CO2 for Carbon Dioxide
-Cannot be separated physically
-Chemical reactions are needed to break compounds into their individual elements
Mixtures
-Two or more elements or compounds physically mixed, not chemically bonded; can be separated by physical methods (e.g. filtration, distillation)
-Components in a mixture retain their individual properties
-Can be seperated by physical means
Key differences:
• Elements: one type of atom, cannot be broken down by chemical means.
• Compounds: can only be broken down into elements by chemical reactions.
• Mixtures: properties of parts stay the same; can be separated physically.
Atoms v.s. Molecules
Atom - smallest indivisible unit of an element
Molecule - group of atoms bonded together (can be made of the same or different elements)
Molecules v.s. Compounds
A compound is a specific type of molecule. Not all molecules are compounds.
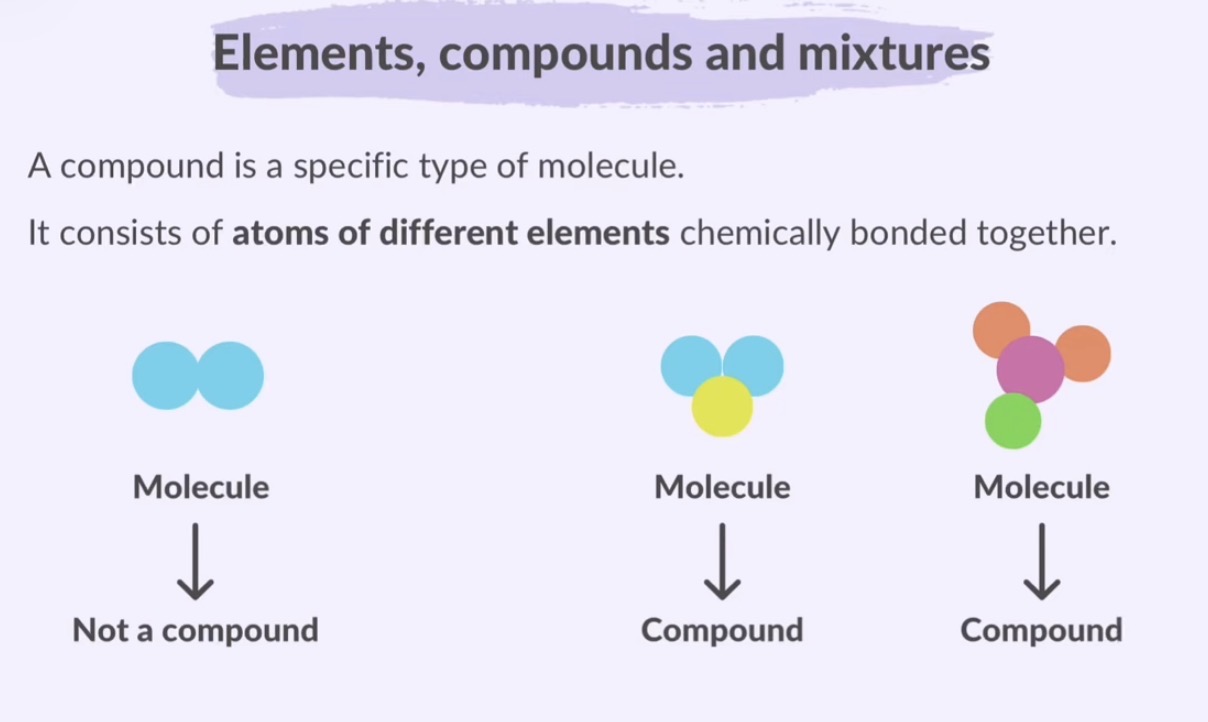

Substances
Substances made of 1 type of atoms are called elements.
→ Atoms
→ Molecules
Substances made of more than one type of atoms are called compounds.
Mixtures are made of two or more elements or compounds not chemically bonded together.
Atomic Structure and the Periodic Table
Atoms:
smallest unit of matter that makes up everything around us
basic building blocks of all substances
come in different types known as elements
Consist of subatomic particles known as:
Protons
Neutrons
Electrons
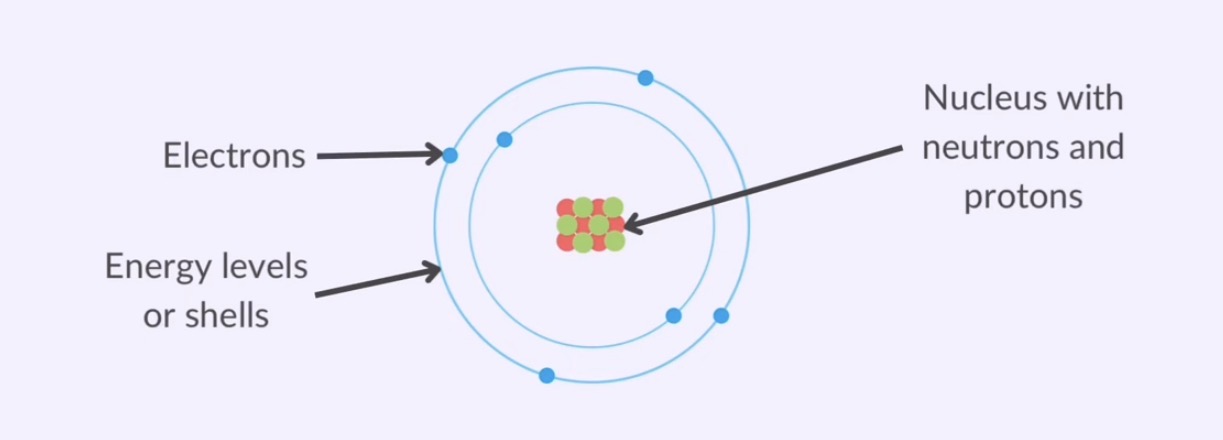
Nucleus
the nucleus is the central core of an atom
contains two types of subatomic particles
Protons-positively charged particles
Neutrons-electrically neutral particles
Electrons are negatively charged particles that orbit the nucleus in shells
much lighter in mass
Electron shells
electrons are arranged in energy levels or electron shells around the nucleus
Atomic Structure:
center called the nucleus with neutrons and protons
electrons arranged in energy levels or shells
electrons orbit the nucleus
Proton Number
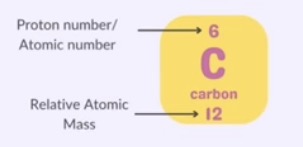
the proton number or atomic number is the number of protons in the nucleus of an atom
the number of protons are always equal to the number of electrons in a neutral atom
the atomic number also helps us identify where the element is located on the periodic table
Each type of atom is characterised by its unique number of protons.
Smaller of the two numbers, whereas on place.
Mass Number
the mass number or nucleon number is the total number of protons and neutrons in the nucleus of an atom
it represents the total mass of the atom
relative atomic mass is not the same thing as mass number
Relative atomic mass v.s. mass number
Relative atomic mass is the average mass of all the isotopes of an element.
Mass number is the total number of protons and neutrons in an element.
Calculating relative atomic mass
Relative Atomic Mass (Ar) is the average mass of all the isotopes of an element compared to 1/12th of the mass of a carbon-12 atom.
The fixed mass of a carbon-12 atom is like a standard reference point for comparing the masses of all other atoms.
Figuring out Ar
We figure out Ar by looking at the relative masses of each isotope and how common each isotope is (it’s abundance).
Increase the number of terms added based on the number of isotopes


Relative atomic mass is rounded to the whole number except for chlorine.
*For most cases, you can use relative atomic mass as a substitute for mass number, except for Chlorine.
Calculating Neutrons
Mass number/Nucleon number - Proton number/Atomic number = Neutron number
Electron Configuration
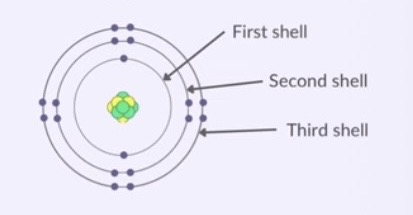
way of describing how electrons are arranged in an atom
electrons are arranged in shells around the nucleus
each shell can hold a certain amount of electrons
1st shell: 2 electrons
2nd shell: 8 electrons
3rd shell: 8 electrons
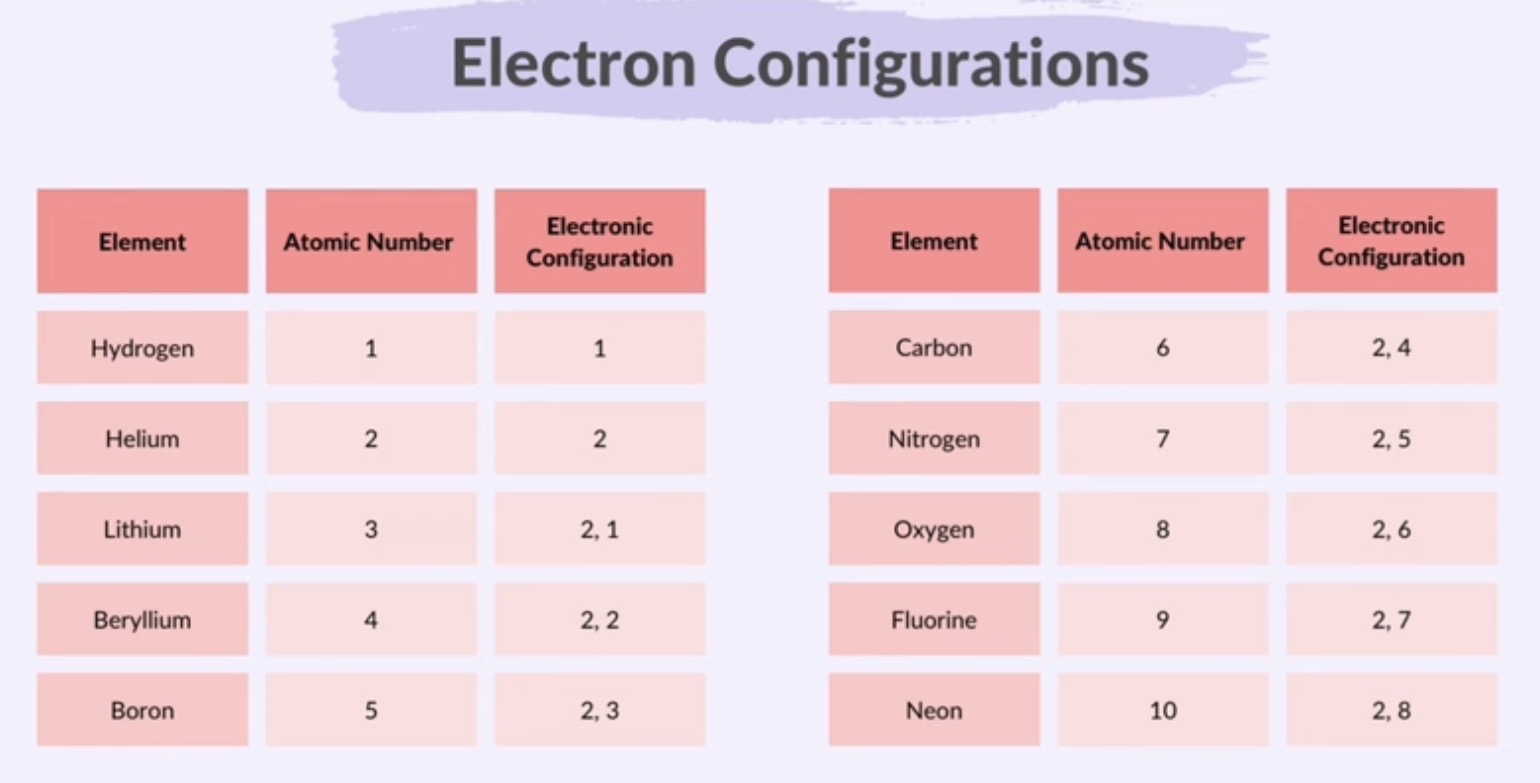
How to write the electron configuration of the element:
Atomic number of the element
Arrange the electrons taken into account with maximum electron number in each shell
Shown using shell diagrams or notation
Ions
Ions: atoms with net electric charge due to the gain or loss of electrons
Electron Configuration of Ions
Atoms become ions because they want a stable, full, outer shell of electrons.
This can be achieved in two ways:
Taking in electrons
Giving away
For example: Sodium (NA)
Sodium has an electron configuration of 2,8,1.
It has 1 electron in its outer shell.
To achieve full outer shell, they can:
Lose 1 electron so they have a full outer shell of 8 electrons
Gain 7 electrons so they can have another full outer shell of 8
Simpler option: 1 - Losing 1 electron
Sodium will give away its extra electron to achieve a full outer shell.
Sodium ion electron configuration: 2,8
Periodic Table
Columns: numbered using roman numerals from 1-8 (groups)
Rows : numbers using digits from 1-7 (periods)
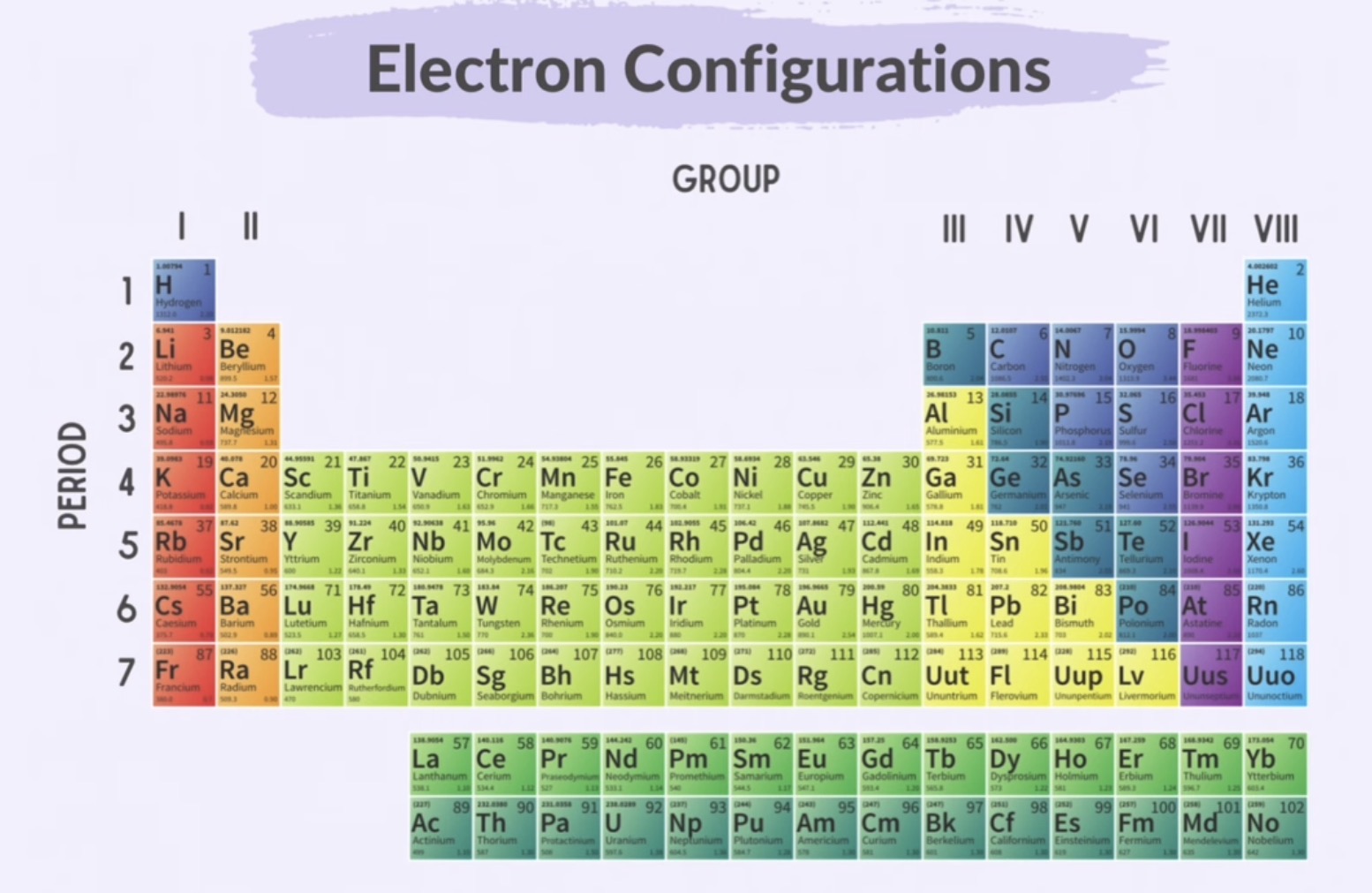
Group 8 - Noble Gases
They have full outer shells
Highly stable/inert
Do not react with other elements because they are satisfied with their electron configuration
The number of outer shell electrons is equal to the group number in Groups I to VIII.
The number of occupied electron shells is equal to the period number of 1-7.
Isotopes
Isotopes: different atoms of the same element that have the same number of protons but different numbers of neutrons.
Same atomic number but different mass number.
Same chemical properties because they have the same number of electrons.
Same electronic configuration.
Example:
Carbon - 12 and Carbon - 14
