3.4: Microbiology
Bacteria overview
Millions of bacteria live in small amounts of soil and water.
Our gut microbiome contains more bacteria than cells (10*13 - 10*14)
500-1,000 different species.
Bacteria are extremely small, ranging from:
Archaea, which can be 0.4um in diameter, making them the smallest known organism which can reproduce independently.
This is only limited by the size of molecules necessary for life.
The largest is a sulphur bacterium, which is 750um in diameter and visible to the human eye.
Most of the cell is a vacuole.
Classifications
Shape
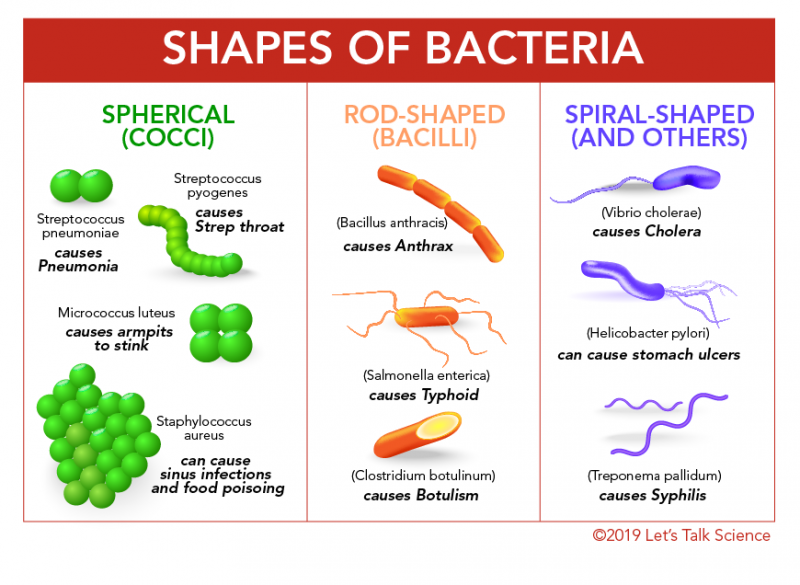
There are three different shapes:
Coccus (spherical), such as Staphylococcus.
Bacillus (rod-shaped), such as E.coli.
Spiral (corkscrew-shaped), such as Spirillum.
This can be further classified by their groupings:
Single, such as Helicobacter.
Pairs, such as Diplococcus pneumoniae.
Chains, such as Streptococcus.
Clusters, such as Staphylococcus.
Gram stain test
This test allows scientists to distinguish between the composition of bacterial cell walls:
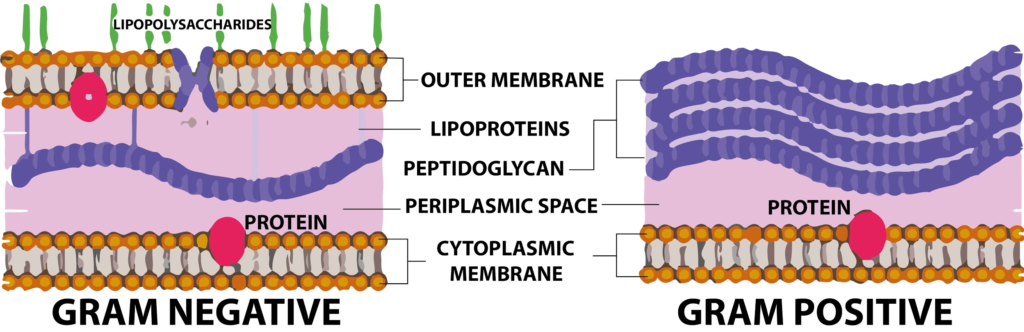
Positive bacteria remains purple throughout, due to its extra lipopolysaccharide layer.
Negative bacteria loses its purple and becomes pink, due to only having a thick peptidoglycan (murein) cell wall.
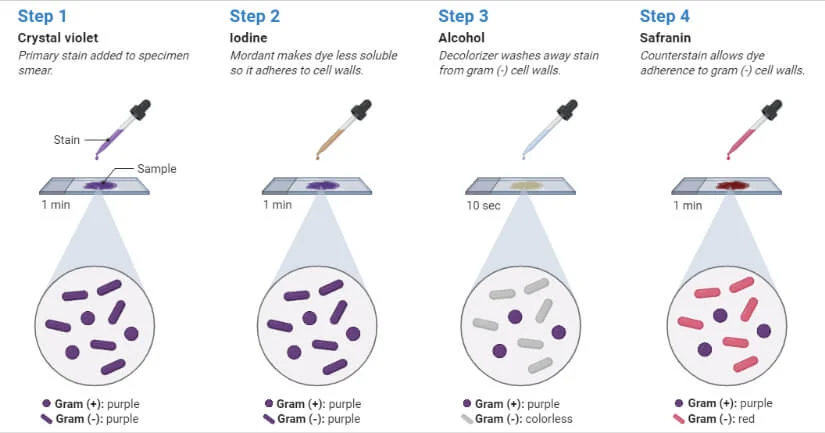
This test happens in 5 steps:
Bacteria are inoculated, spread on a glass slide and heated. It is currently transparent.
Crystal violet (CV) is added to stain the bacteria. This binds to the peptidoglycan, staining all bacteria purple.
Lugol’s iodine is added to bind the CV to the peptidoglycan more strongly. This is known as a mordant (fixes a dye to a material). The bacteria remain purple.
Acetone-alcohol is added as a decolouriser, which removes unbound CV. Due to the lipopolysaccharide molecules, the purple dye washes off gram negative bacteria. Gram positive bacteria remains purple, and gram negative becomes transparent.
Safranin is added as a counterstain, in order to make the gram negative bacteria visible. The gram negative bacteria become red, and gram positive remain purple.
Gram stain positive
This bacteria remains purple, and includes Staphylococcus and Streptococcus.
The lack of a lipopolysaccharide layer in the cell walls allows the stain to bind effectively.
They are also more susceptible to antibiotics (such as penicillin) and the enzyme lysozyme than gram negative bacteria.
Penicillin works by preventing bonds between peptidoglycan from forming. This prevents new cell wall creation during mitosis, making the cell wall weak and allowing the cell to burst via osmosis.
Lysozyme is an antibacterial enzyme, in human tears and saliva, which works by hydrolysing bonds holding peptidoglycan molecules together.
Gram stain negative
This bacteria loses the purple and becomes red, and includes Salmonella species and E.coli.
This has a more chemically complex cell wall, as the outer cell membrane is supplemented with large lipopolysaccharide molecules.
These protect the cell from dyes, makes them penicillin resistant and unaffected by lysozymes.
To control them requires antibiotics which interfere with the cell’s ability to make proteins.
These do not harm the eukaryotic (human) cells as they have different protein making cellular machinery.
Culturing bacteria
Microorganisms undergo asexual reproduction and reproduce quickly in suitable environments.
In optimum conditions they can divide every 20 minutes.
This optimum can vary across bacteria.
Necessary conditions
The following conditions are required:
Nutrients - Supplied using a nutrient media, either a liquid medium known as a nutrient broth or a medium solidified using agar. Mediums provide water and:
A carbon and energy source, usually glucose.
Nitrogen for amino acid synthesis, in organic and inorganic forms, such as nitrate ions.
Growth factors:
Vitamins, such as biotin.
Mineral salts, such as Na+ (sodium), Mg*2+ (magnesium), Cl- (chlorine), SO4*2- (sulphur), PO4*3- (phosphate).
Temperature - Enzymes regulate bacterial metabolism.
A range of 25-40 degrees is suitable for most bacteria.
Mammalian pathogens have an optimum of 37 degrees - human body temperature.
pH:
Most bacteria favour slightly alkaline conditions (7.4).
Fungi have an optimum of neutral to slightly acidic.
Oxygen need - There are 3 categories of oxygen need:
Obligate aerobes - Oxygen is required for metabolism, such as Mycobacterium tuberculosis.
Facultative aerobes - Microorganisms that do better with oxygen, but can survive in its absence, such as E.coli.
Obligate anaerobes - Microorganisms that respire without oxygen and cannot grow in its presence, such as Clostridium bacteria.
This produces toxins or poisons in a wound, destroying body tissue in a condition known as moist gangrene.
Aseptic technique
This is also known as sterile technique, where all bacteria and apparatus are kept clear of microorganisms to prevent contamination of the bacteria culture but also the environment.
To prevent culture and apparatus contamination:
All apparatus and media is sterilised before use to prevent initial contamination.
Culture vessels are flamed before opening and closing.
Sterile loops are used to prevent subsequent contamination.
To prevent environmental contamination:
Work surface is sterilised before and after use using a disinfectant.
During inoculation (introduction of bacteria to encourage it to grow):
The culture bottle is held in one hand, and the cap is held in the other hand. It should not be placed on the work surface.
The mouth of the bottle should be flamed for 2-3 seconds.
Bunsen burners should be continuously heated as this causes bacteria to rise due to a convection current - therefore away from the apparatus.
The inoculation loop should be heated until red hot and then cooled.
The petri dish lid should be lifted just enough for the loop to enter.
There are two techniques for this, spreading (a pipette drop and then spread) and streaking (done using an inoculation loop. Three streaks are made, then a slight turn and three more. This is done five times, making a shape that resembles a hexagon.).
The lid should be secured with tap, but not all the way as this would close off oxygen, encouraging the growth of anaerobic and likely pathogenic microorganisms.
Incubate at 25 degrees, avoiding 37 degrees as this would encourage pathogenic microorganisms to grow.
Petri dishes should not be opened after incubation (when bacterial colonies grow).
In labs, autoclaves are used.
These are sealed containers where glass and metal equipment is heated to 121 degrees in steam under pressure for 15 minutes after the optimum pressure is reached.
Disposable materials after use, such as petri dishes, are sealed inside autoclavable plastic bags, autoclaved, then disposed of.
Measurements
Estimating the growth of a bacterial population in a suspension (a fluid in which particles are dispersed but not totally dissolved) is important.
It is done by environmental health officers taking food samples.
Water authorities check water supplies daily.
Food manufacturers must check the food they sell.
Food and drugs are produced using bacteria grown in industrial fermenters, and bacterial count is important.
There are two methods:
Directly - Counting individual cells.
Viable counts only count living cells.
Total counts include living and dead cells.
Indirectly - Measuring the turbidity (cloudiness) of the culture.
Directly
It is not possible to count all bacteria, so only a small sample is counted.
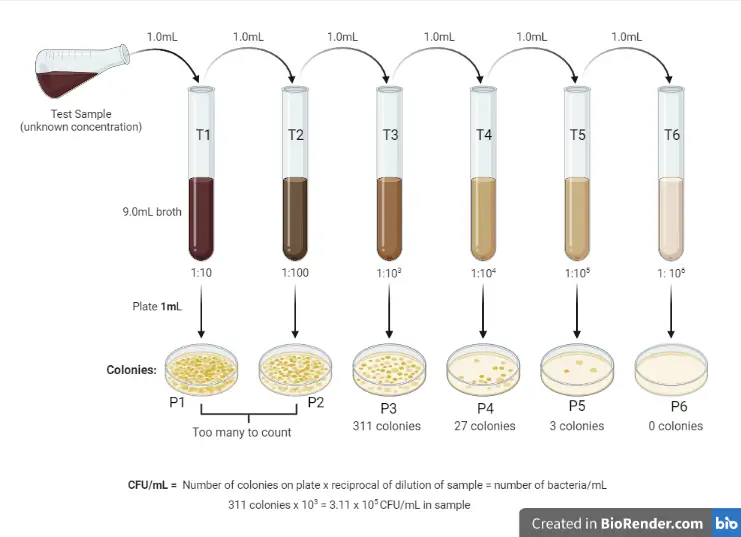
Dilutions are also necessary in order to count all the bacteria due to high population density.
As this only counts bacterial colonies, only live cells are counted.
Serial dilution is used, where a substance is diluted multiple times, usually of 1cm (moves up in 10-1) or 0.1cm (moves up in 10-2) suspension to 9cm or 9.9cm dilution.
1cm of each diluted sample is spread over a sterile agar plate and incubated over 2 days at 25 degrees.
If a sample isn’t diluted enough, colonies merge (clumping) together and make counting inaccurate.
Only a dish containing 20-100 viable colonies that are distinct and separate are chosen.
This sample is then multiplied by the dilution factor.
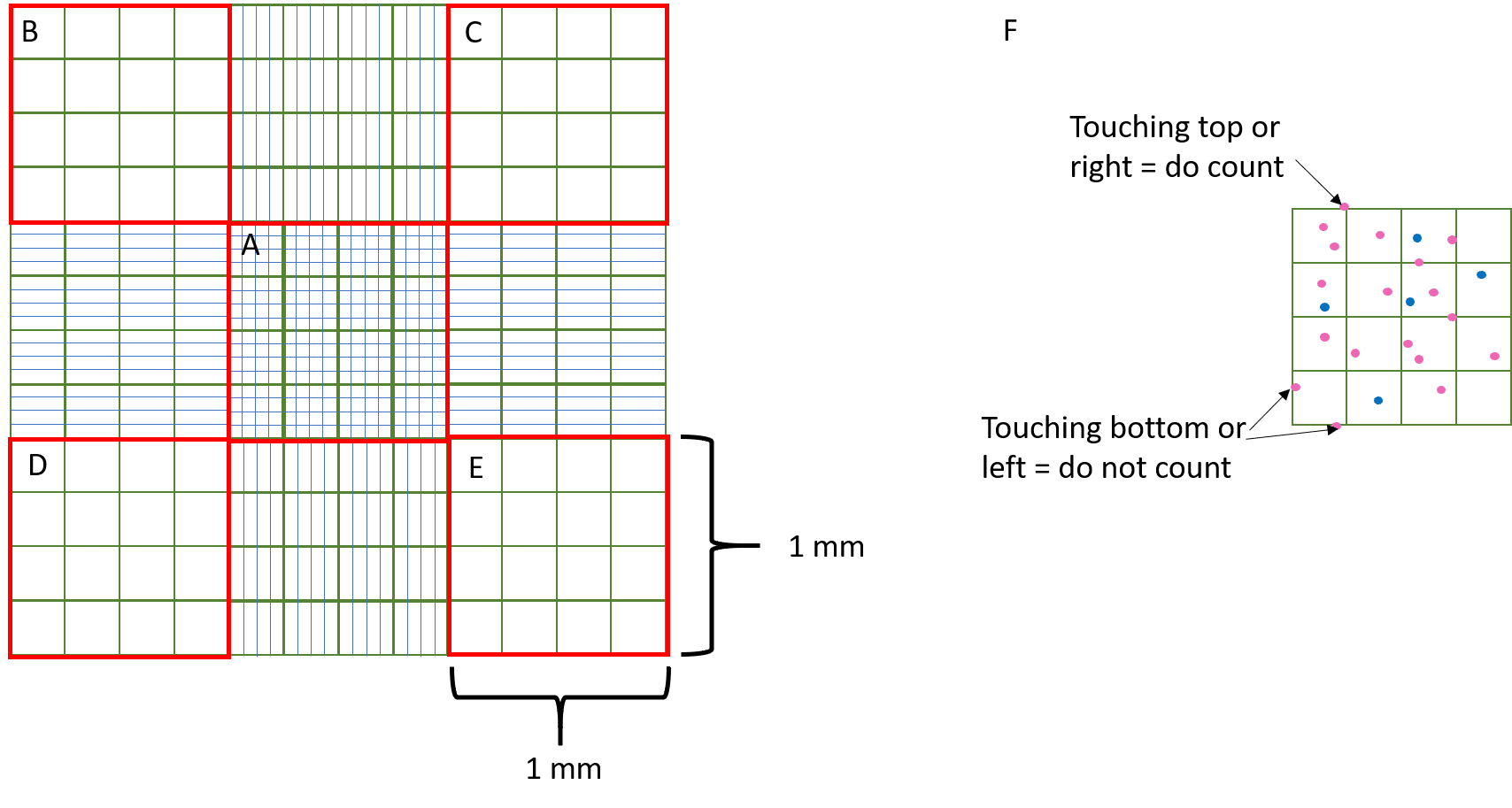
A haemocytometer can also be used, a more accurate method.
This is a specialised microscope, with a raised platform in the centre with perpendicular lines, allowing for individual counting.
However, this counts both living and dead cells.
Indirectly
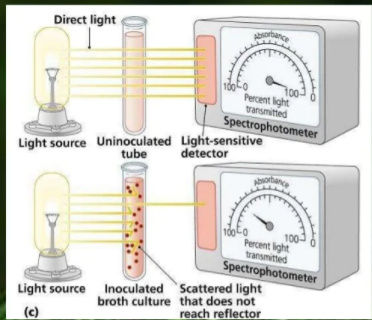
Turbidimetry is used, where a colorimeter is used to measure the turbidity of the culture, which would increase alongside the cell concentration.
This would measure the population by finding the exact absorbance of a suspension and comparing it to a graph of light suspension alongside bacterial cell count.
This provides a total cell count, as colorimeters do not distinguish between living and dead cells.
This is used in the field, such as in river water samples.