EARTH SCI
All planets in the solar system share the same origin but only Earth has life
Certain factors have influenced the formation and continuation of life on Earth
Life would not exist without liquid water
Water is an important component of many biological processes
WATER ON EARTH IS THOUGHT TO HAVE ORIGINATED FROM 2 SOURCES:
Volcanism: in the form of steam
Icy Meteors: meteors would’ve partially melted even before impact
Many planets in our solar system also have water; this water is frozen in ice caps such as on Mars
Water may also be found dispersed through a planet’s thick atmosphere in the form of water vapor such as on Venus
Earth is the exception to the previous situations as we are located in the so-called habitable zone where water should exist
Heat is a necessary force in driving the different systems that support life on Earth
Earth receives heat from 2 main sources:
EXTERNAL HEAT (OUTSIDE EARTH)
Originates mainly from the sun
Heat enters Earth via radiation; this heat is affected by the greenhouse effect
INTERNAL HEAT (WITHIN EARTH)
Comes from radioactive decay of materials in the core and mantle (ex. uranium)
Heat is released and felt through tectonic activities like volcanism and plate movement
It is a layer of gas surrounding the planet
Gravity keeps this layer of gas around the planet as it would normally keep rising and eventually diffuse into outer space
The atmosphere contains vital gases for the respiration of living organisms
Atmosphere also serves to regulate the temperature of a planet and protect living organisms from excessive radiation
The atmosphere has several distinct layers
Each layers in the atmosphere have features that distinguish them from the others
Amount of matter on our planet is fixed
Resources that we obtain from our planet are fixed and do not regenerate
The waste we produce cannot be disposed
Law of Conservation of Mass
Matter can neither be created nor destroyed in a chemical reaction
Amount of matter cannot change
Mixture of gases that surround a planet
Responsible for meteorological phenomena
Earth’s atmosphere contains roughly;
78% nitrogen
21% oxygen
0.9% argon
0.1% trace gases
Solid parts of the earth
Composed of minerals called rocks, loose particles of rocks called regolith
Includes geologic landforms
Totality of Earth’s water
Includes the permanently frozen parts that make up the cryosphere
Considered separate from the water in the atmosphere but are ultimately connected
All forms of life and organic matter that haven’t decomposed yet
Formed by interactions between the Earth’s other subsystems
4 subsystems are linked closely via biogeochemical cycles
The cycles serve as pathways where chemicals move through the biotic and abiotic components of the Earth
Any significant activity in one ecosystem can greatly affect the other subsystems in various ways; effects are distributed via the biogeochemical cycles
Water cycle is an example of a biogeochemical cycle that is easily affected by a change in any of the subsystems; due to the ability of water to dissolve a wide range of substances
An example of a disruption would be when pyroclastic eruptions occur (Mt. Pinatubo and Taal)
Lahar: large amounts of ash released by volcanoes mixed with rainwater creating a dangerous mixture
The carbon cycle is not immediately affected by changes in the subsystems but its effects are greater in the long run; this is mainly manifested and felt as global warming via the greenhouse effect.
Greenhouse effect: atmospheric phenomenon wherein the atmosphere traps some of the heat it receives instead of releasing it all back into space (Greenhouse gases like CO2 and CH4 amplify this effect)
Naturally occurring, inorganic solids with definite chemical compositions and ordered internal structures
Each mineral is unique but displays general characteristics
NATURALLY OCCURRING
Minerals are formed naturally and exist without human intervention
Iron ore; mineral
Steel; man-made alloy
INORGANIC
Minerals formed only through inorganic processes
Materials made from or by organic materials and processes are not minerals
Diamond; mineral
Coal; NOT MINERAL
SOLIDS
Inorganic liquids and gases are not considered minerals
Ex. natural ice
DEFINITE CHEMICAL COMPOSITION
Minerals have exact chemical formulas containing elements and compounds in specific ratios (recipe)
Some minerals exhibit atomic substitution as an exception to the rule
ORDERED INTERNAL STRUCTURE
Atoms in minerals have an organized, repetitive, geometric pattern called a crystal structure
Halite & Sea salt = NaCl
Volcanic glass, although formed naturally, is not a mineral due to being amorphous
Ex. obsidian
Materials that fulfill all the requirements of being a mineral except having an organized internal structure
AMBER
Fossilized tree resin
Formed millions of years ago from tree sap
May contain trapped organisms inside
OPAL
Hydrated amorphous form of silica
Although similar to a mineral, its variable molecular structure disqualifies it from that category
PEARL
Hard, glistening objects formed by shelled mollusks
Mainly composed of CaCO3
May also be created artificially by implanting stimulus in shellfish
Classified according to the elements that compose them
Elements are abundant in the Earth’s crust
Minerals can be classified as:
Silicates
Oxides
Sulfides
Sulfates
Halides
Carbonates
Native metals
SILICATES
Composed primarily of silicon-oxygen tetrahedrons
Ex. olivine & quartz
SiO2 or SiO4
SULFATES
Consist of metal cations bonded to Sulfate anionic group
Usually precipitates out of water
Ex. include gypsum & copper sulfate
SO4–2
HALIDES
Consist of halogen ions bonded with other elements
Ex. rock salt & fluorite
Halogen ion
CARBONATES
Possess a carbonic ion
Ex. calcite & dolomite
Carbonic ion CO32-
NATIVE METALS
Single or pure metals (elemental)
Ex. copper & gold
Pure metals
CRYSTAL STRUCTURE OF MINERAL
Minerals with similar compositions tend to have similar crystal structures, thus belonging to the same crystal system.
Crystal structure of minerals depend on their composition:
Isometric, Tetragonal, Orthorhombic, Monoclinic, Triclinic, Hexagonal
Crystal form and habit
Cleavage and fracture
Luster
Color and streak
Hardness and density
CRYSTAL FORM AND HABIT
Since minerals have definite compositions, they form a definite crystal structure which crystallizes into a specific crystal form
Outward appearance of the mineral’s crystal form is its habit
Crystal habits can be described as:
Granular (dioptase)
Dendritic (manganese, ice)
Geode (amethyst, quartz)
Fibrous/Acicular (natrolite, asbestos)
Reniform (hematite)
Lamellar (gypsum)
Prismatic (quartz)
CLEAVAGE AND FRACTURE
How a mineral would break depending on its molecular bonding and structure
Cleavage: tendency of a mineral to break along planes weakness
Minerals with excellent cleavage tend to break into smooth, flat, and parallel surfaces
Minerals with good cleavage will result in small, smooth, step-like flat surfaces
Minerals with poor cleavage have cleavage surfaces that are difficult to identify
Minerals with no cleavage will fracture either in an irregular manner or as conchoidal fractures (smooth curved surfaces)
LUSTER
Pertains to the appearance of light as it is reflected off the surface of a mineral
Dull luster
Greasy luster
Metallic luster
Silky luster
COLOR AND STREAK
Color is not a reliable property in minerals as impurities can alter their original colors
Streak is the color of a mineral in its powdered form; obtained using a streak plate
Ex. quartz; normally colorless but with the addition of impurities colors can be formed
HARDNESS
Resistance of a mineral to scratching
Can be measured by scratching it with another mineral or reference material with a known hardness
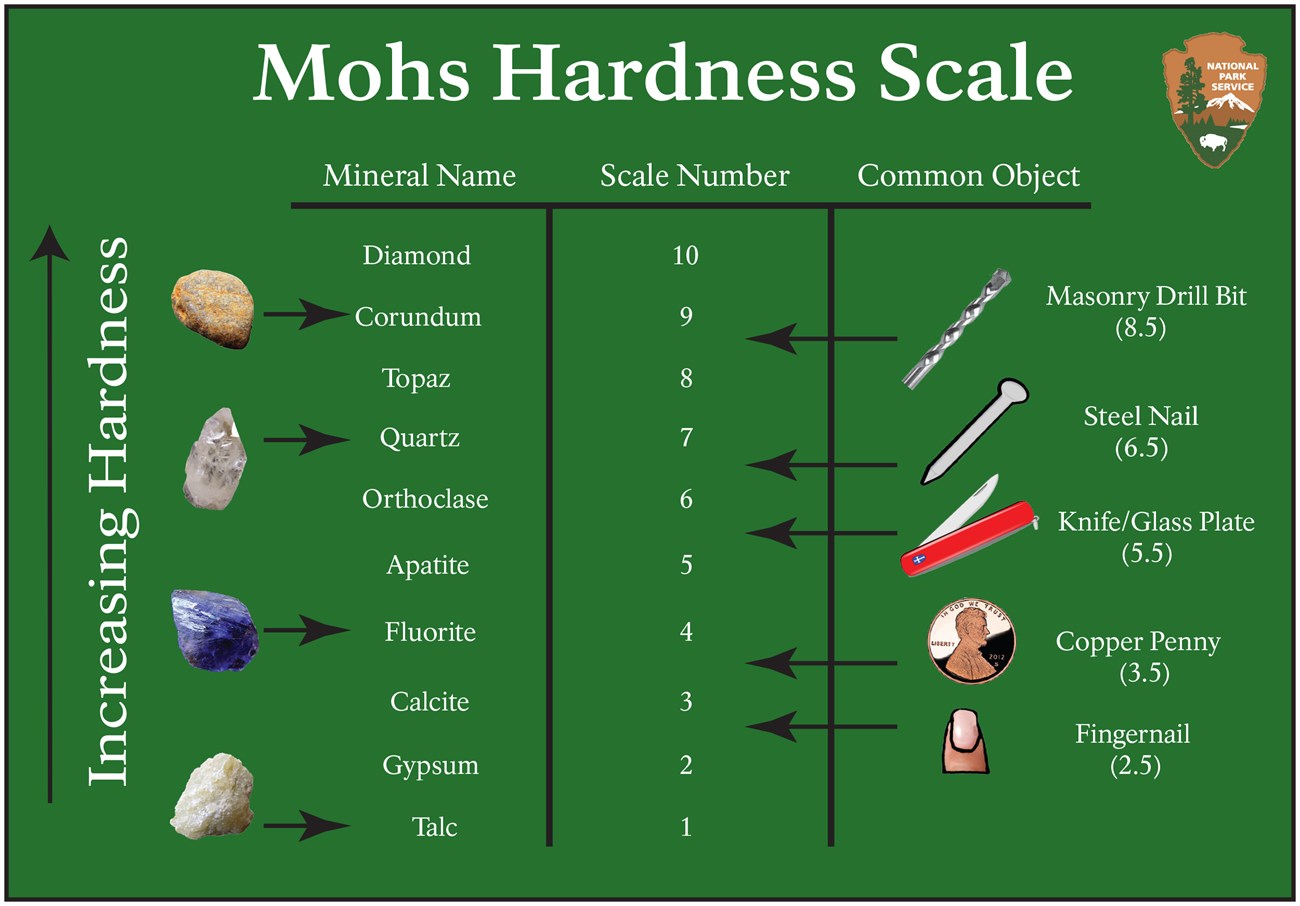
DENSITY
Specific gravity is a measure of the density of a mineral
Specific gravity value is expressed upon how much greater the weight of the mineral is to an equal amount of water
Water has a specific gravity of 1
OTHER PROPERTIES OF MINERALS
Magnetism
Taste
Effervescence (reaction to acids)
Feel
98% of the Earth’s crust is composed of 8 elements
Most of the common rock-forming minerals are silicate minerals composed of silicon and oxygen
Naturally occurring coherent aggregates of minerals or solid materials such as natural glass and organic matter
Found in the lithosphere
Lithosphere: “lithos” - stone
Lithosphere: rigid, rocky, outermost part of Earth
Composed of the crust and upper mantle
Rock cycle that describes all the processes by which rocks are formed, modified, transported, decomposed, melted, and reformed
Processes occur on the Earth’s surface and underneath
A dynamic cycle of processes and products
Formed by cooling and solidifying magma or lava
“Igneus” - fiery/fire
Form at higher temperatures than other rocks
Ex. obsidian, pumice, basalt, granite
Below the surface
From slowly cooling magma (intrusive or plutonic) ex. Granite
On the surface
From rapidly cooling lava (extrusive or volcanic) ex. Basalt
On the surface
From consolidation or particles erupted by explosive volcanic activity (pyroclastic igneous rocks) ex. Breccia
Crystalline igneous rocks
ex. granite
Products of the lithification or particles produced by the weathering of other pre-existing rocks
Parent material can come from igneous, sedimentary, and metamorphic rocks
“sedientarius” - sitting
Composed of clasts that vary depending on their sizes
Ex. sandstone, halite, conglomerate, coquina
CLASTIC
From the cementation of sediments that have been deposited, buried, and compacted over a long period
Ex. sandstone, conglomerate
CHEMICAL SEDIMENTARY ROCKS (CRYSTALLINE)
From the precipitation of minerals from ions in solutions
Ex. halite, gypsum
BIOCLASTS
From the compaction or cementation of animal or plant remains
Ex. coal, coquina
Formed from pre-existing parent rocks are altered by heat, pressure, and the chemical activity of fluids
“Metamorphism” - change in form
Metamorphism occurs underneath the Earth’s surface, although not as deep as igneous rocks
Ex. marble, quartzite, slate, gneiss
FOLIATED
Dominant altering factor is pressure, producing multiple bands of rocks that are aligned to the axis of pressure
Ex. gneiss
NON-FOLIATED (CRYSTALLINE)
Dominant altering factor is heat
Usually undergoes changes in texture due to the recrystallization of minerals
Ex. quartzite
ORES
Naturally occurring materials that can be profitably mined
May be mineral or rock and metallic or nonmetallic
Deposit is considered a potential ore body if its localized abundance is greater than normal
ROCK OR MINERAL IS CONSIDERED AN ORE BASED ON THE FF:
Overall chemical composition
Percentage of extractable resources concerning its total volume
Market value of the resources
Ore bodies are unevenly distributed throughout the Earth’s crust
A country will never be self-sufficient in terms of resources due to the unequal distribution of resources
This is exacerbated by the fact that all resources are finite
Potential ore bodies are located by recognizing the geologic processes that are responsible for the creation of a specific material
Mining: set of processes in which useful resources are withdrawn from a stock of nonrenewable resource
Mining is an important part of society because some vital resources cannot be grown by man
PROSPECTING
Assess the area
DRILLING
Testing the area
MODELING
Designing a structure conducive to the extracting of ore
IMPACT ASSESSMENT
If the mine will severely impact the environment
Harm should be reversible and not permanent
CONSTRUCTION
Mines should be constructed properly to avoid collapsing
EXTRACTION
Extracting of ores and minerals
MILLING AND REFINING
Breaks what is mined down into resources
DECOMMISSIONING
Proper closure of mine opened and return to original state as much as possible
Philippines is located within a very active and dynamic tectonic setting
The country has a unique setting in mineral and ore development and extraction
Philippines is the 5th most mineralized country in the world
Erosion
Sinkholes
Water pollution
Acid rock drainage
Heavy metal poisoning
Deforestation