alcohols and ethers
alcohols
All alcohols contain the hydroxy group (-OH) and have general form ROH
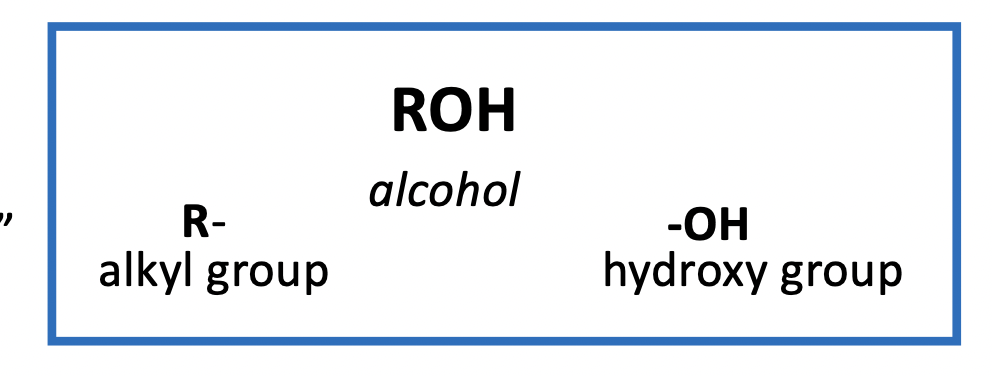
Nomenclature (naming):
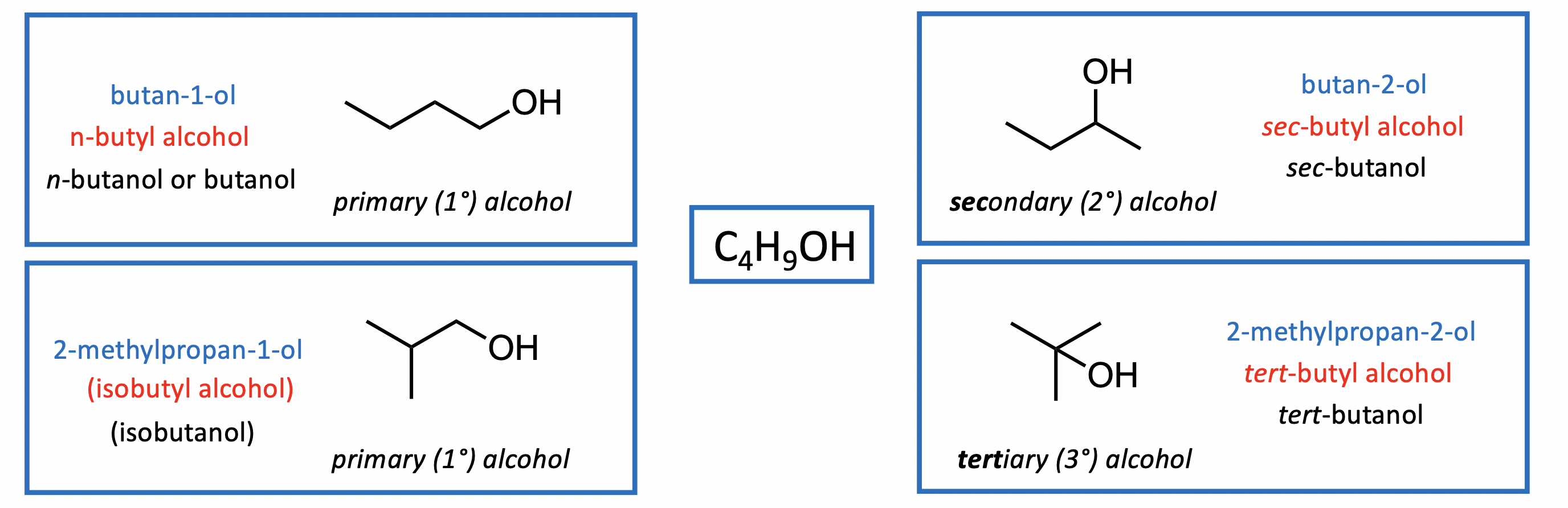
IUPAC system: remove the “e” from the alkane parent group and replace with “ol” A number before the ”-ol” indicates position of OH group on parent carbon chain
Common Names: Use “alkyl” group + “alcohol”
Mixture: common & IUPAC names are merged sometimes
reactions
Reactions of the O-H bond
deprotonation (step 1)
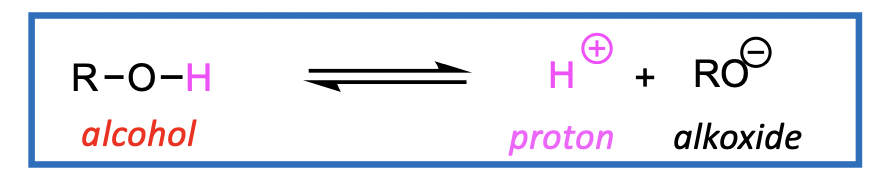
Can break the bond between O-H bond
H atom (1 è) H+ proton (0 è) H- hydride (2 è)
How easy is it to lose the OH proton (deprotonation)?
We can look to the acidity of alcohols to answer this.
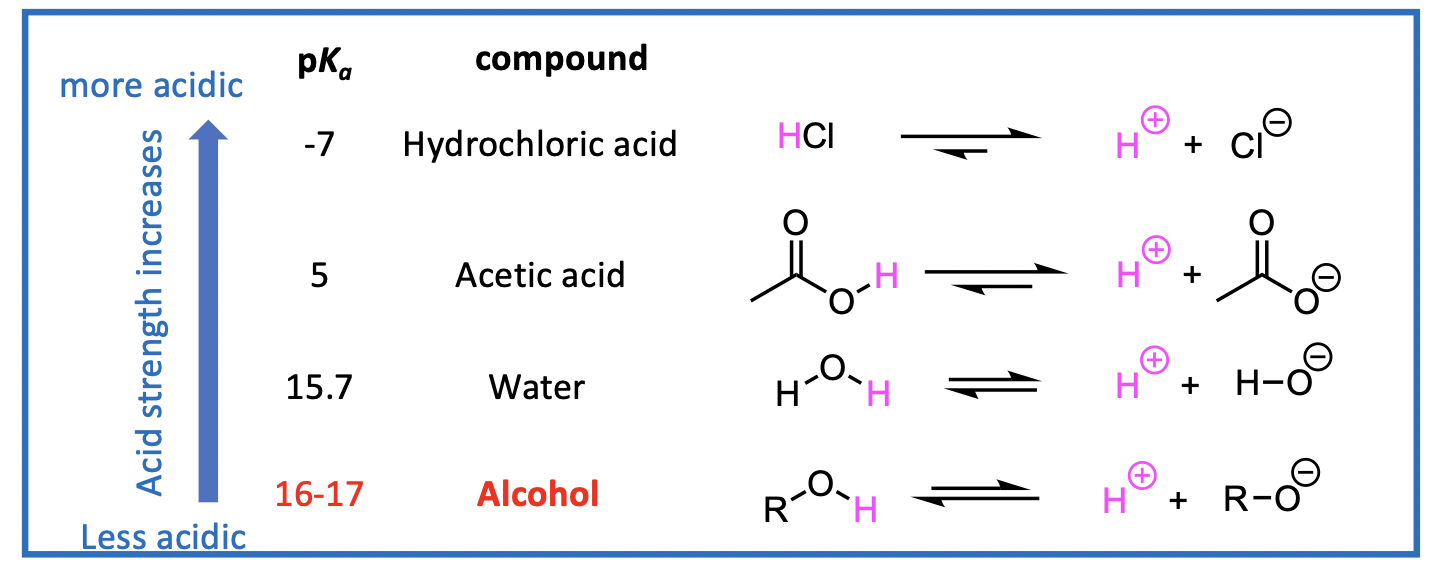
Lower pKa means more acidic proton, therefore equilibrium moves to the right.
This means an alcohol will not be deprotonated as easily as hydrochloric acid because the pKa of ROH is larger than pKa of HCl
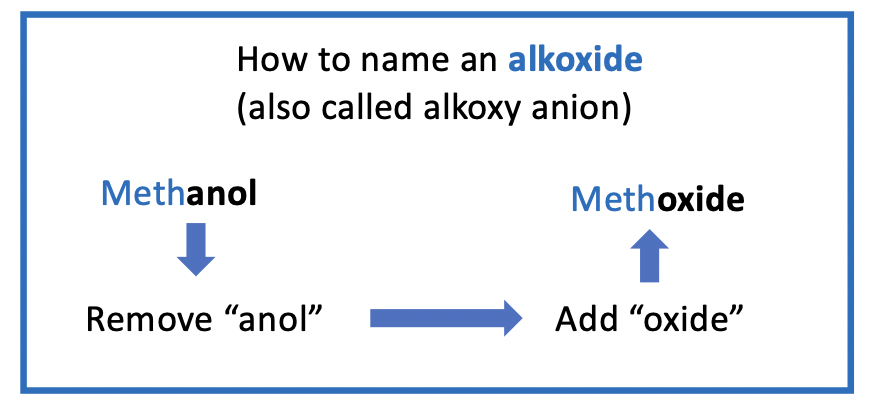
forming an alkoxide
an alkoxide is formed - an alkoxide is the conjugate base (CB) of an ROH
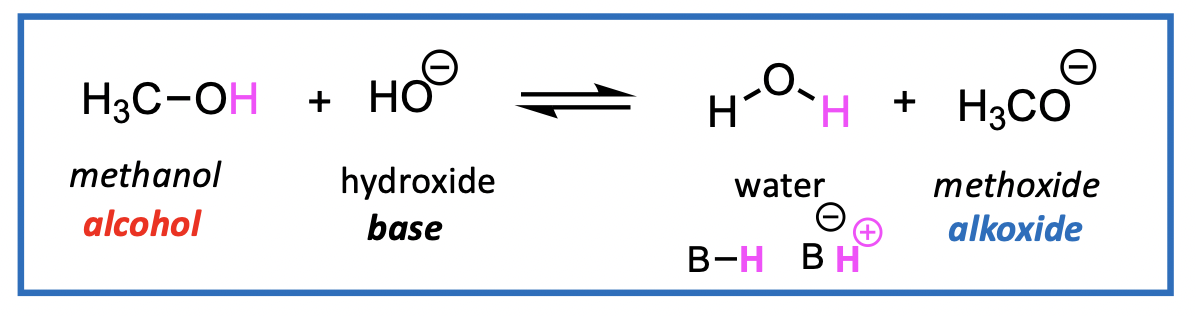
CH3OH and H2O have similar pKa, Therefore, get a mixture of HO- (hydroxy) and H3CO- (methoxy) anions
NOT a good method for making alkoxides
Creation of metal alkoxides using sodium or potassium metal
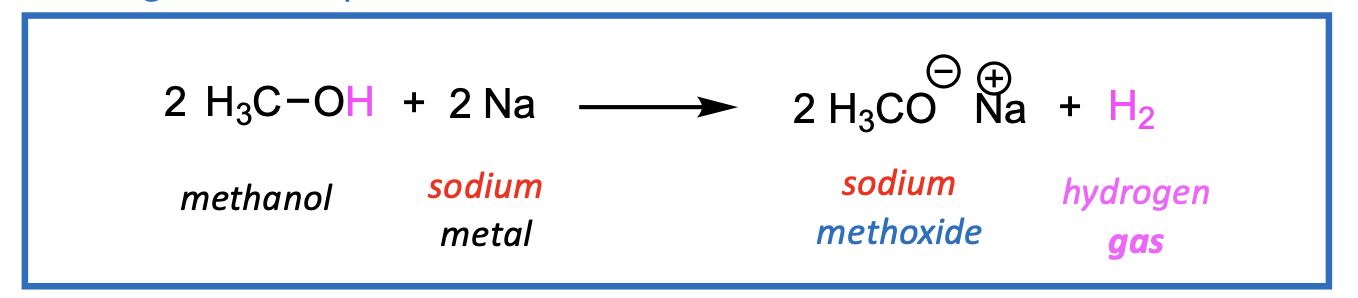
Creation of metal alkoxides using sodium or potassium metal
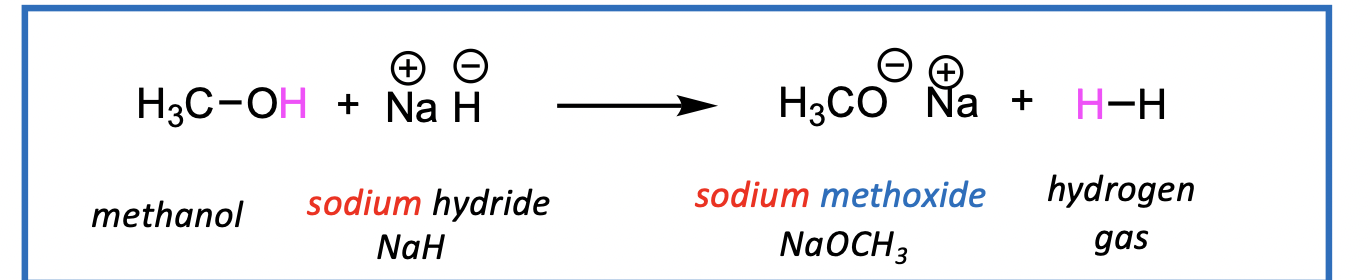
Reactions go to completion (no equilibrium) with H2 gas bubbling out of solution.
Also, metal alkoxides are useful as bases in elimination reaction
Reactions of the O lone pairs
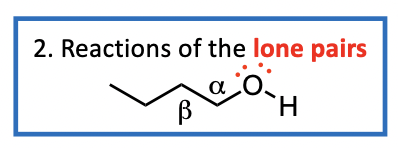
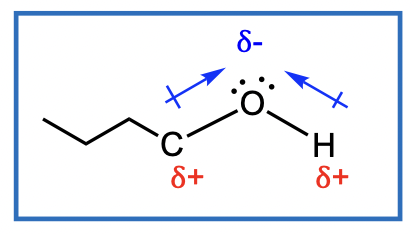
The oxygen of the alcohol has lone pairs of electrons
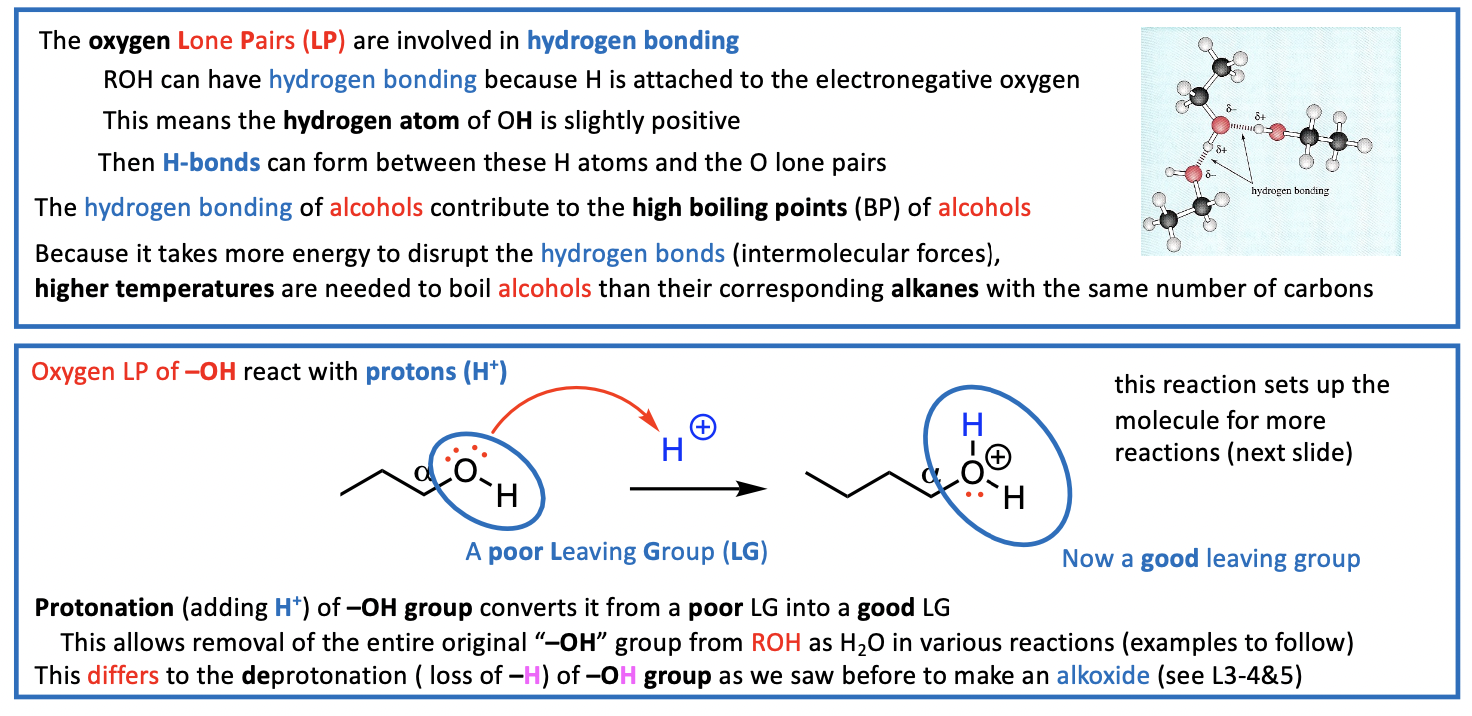
We now consider the reactions that can occur at the C-O bond of ROH Acid Conditions Needed
2 things can happen; Sn or E
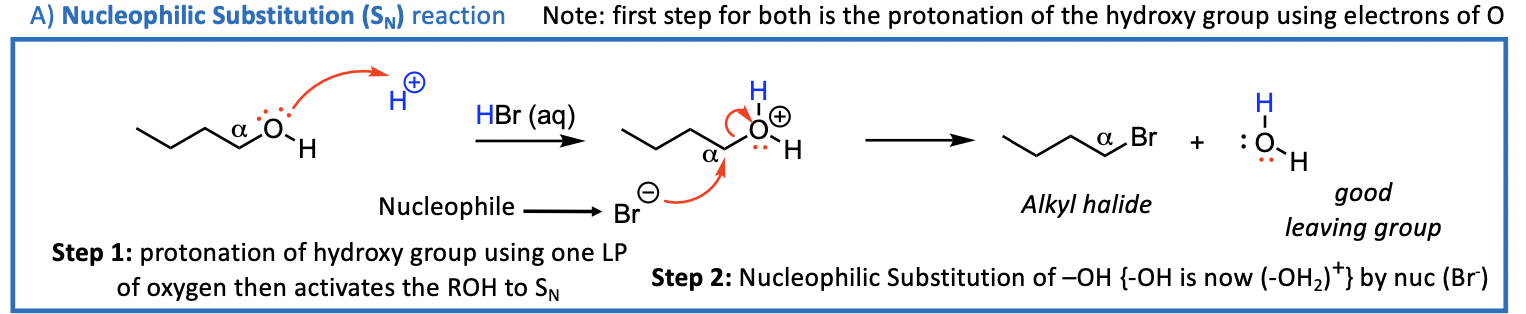
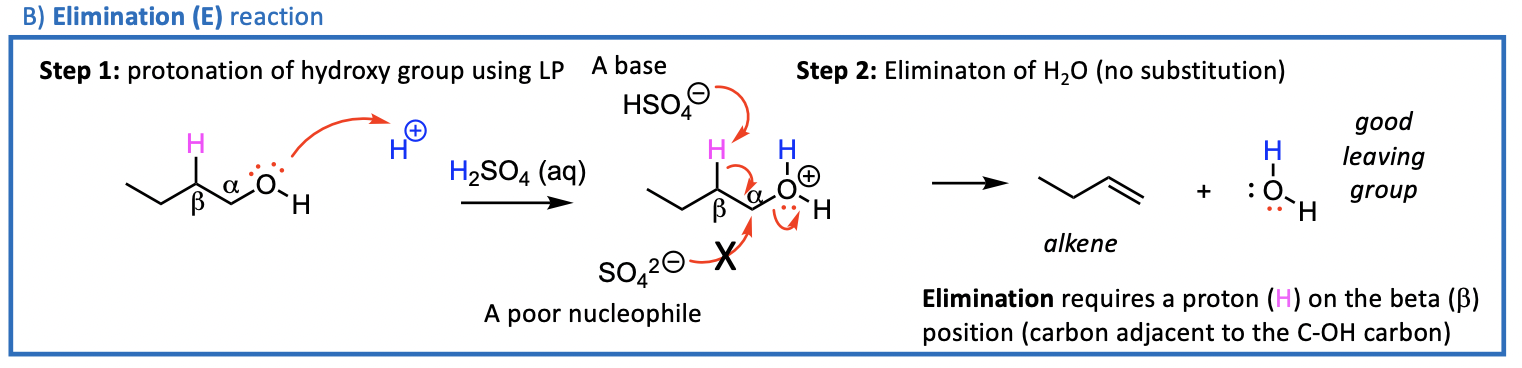
Reactions of the C-O bond
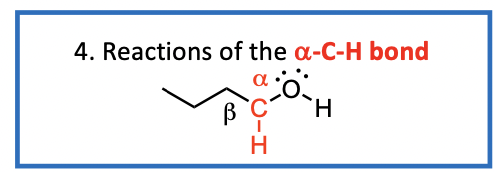
We now consider the reactions of the a-C-H bond of alcohols
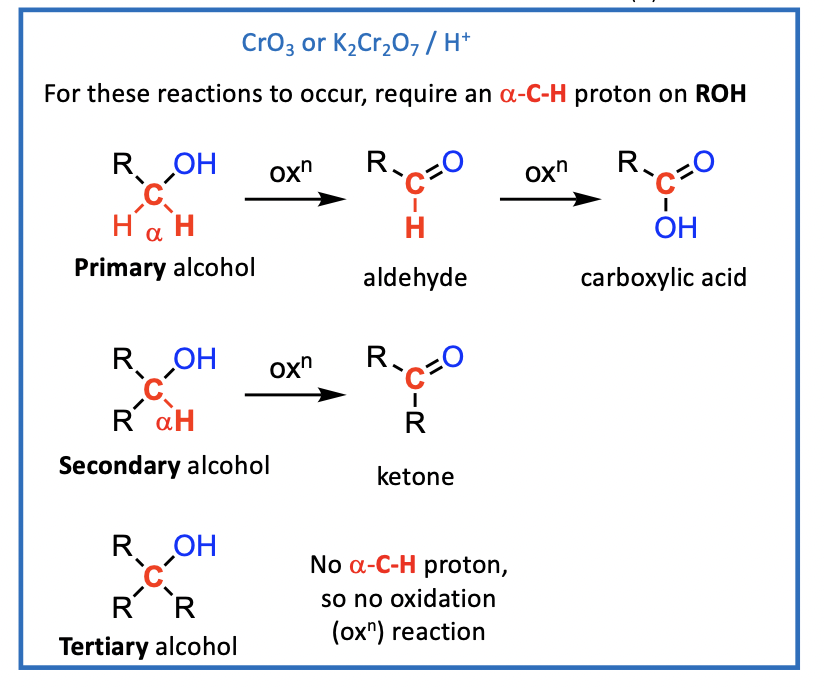
Reactions of the a-C-H bond
ethers
Ethers are compounds in which an oxygen atom is bonded to two alkyl (or aryl) groups The two R groups can be the same (symmetrical ethers) or different (asymmetrical ethers)
Nomenclature (naming):
IUPAC system: ethers are named as substituted alkanes (“alkoxy”+”alkane”)(RO-R)
A number before the ”alkoxy” indicates position of OR group on parent carbon chain
Common Names: write “alkyl” groups in alphabetical order then add “ether” (R-O-R)Additional : historical names also apply
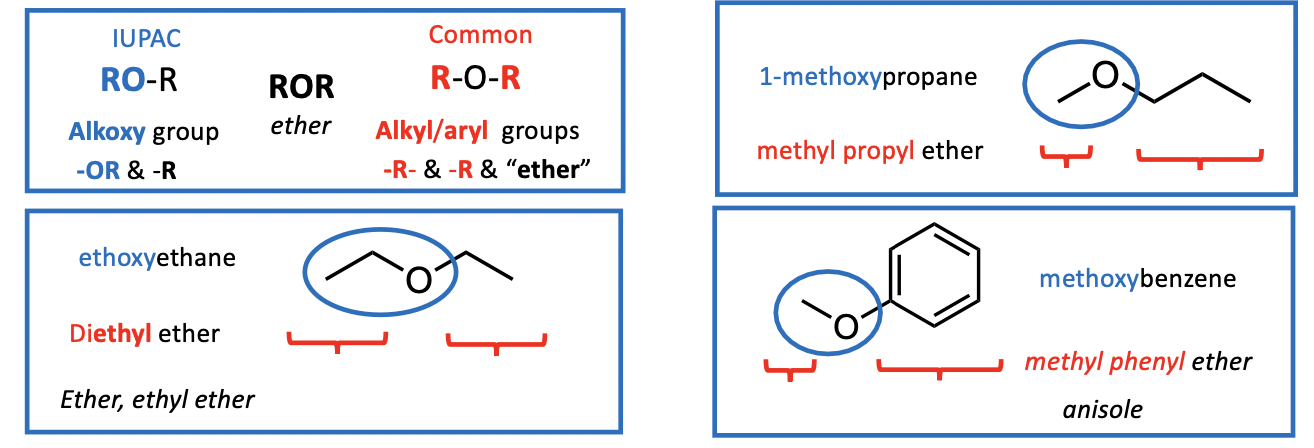
properties
Ethers are relatively inert compounds
Ethers are often used as solvents for organic reactions
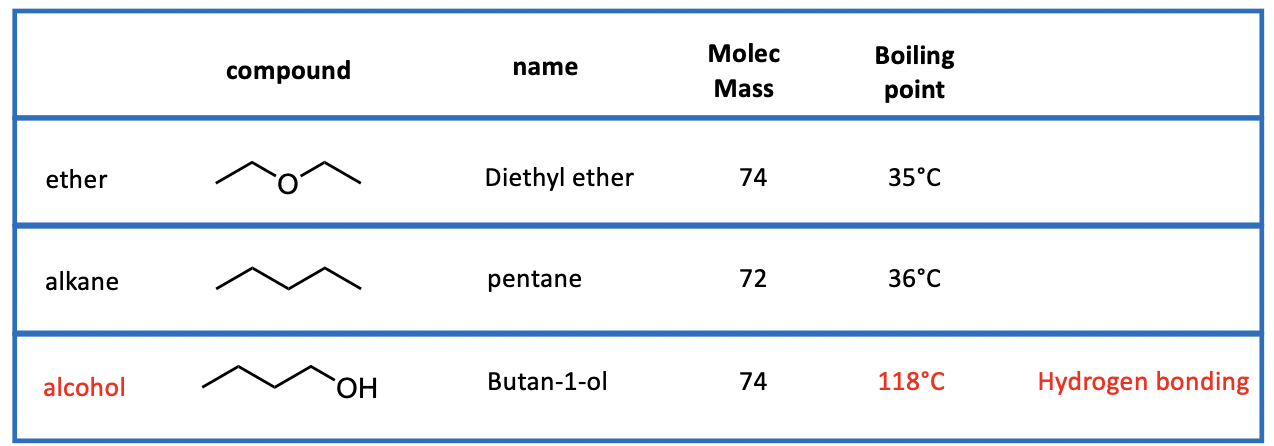
Positive aspects of Ethers
Good solvents
Low boiling points (35 deg)
Not reactive
Does not mix with water (immiscible)
Excellent for extraction of organic material from water
Negative aspects of Ethers
Highly flammable
Forms explosive peroxides (RO-OR)