Collision Theory
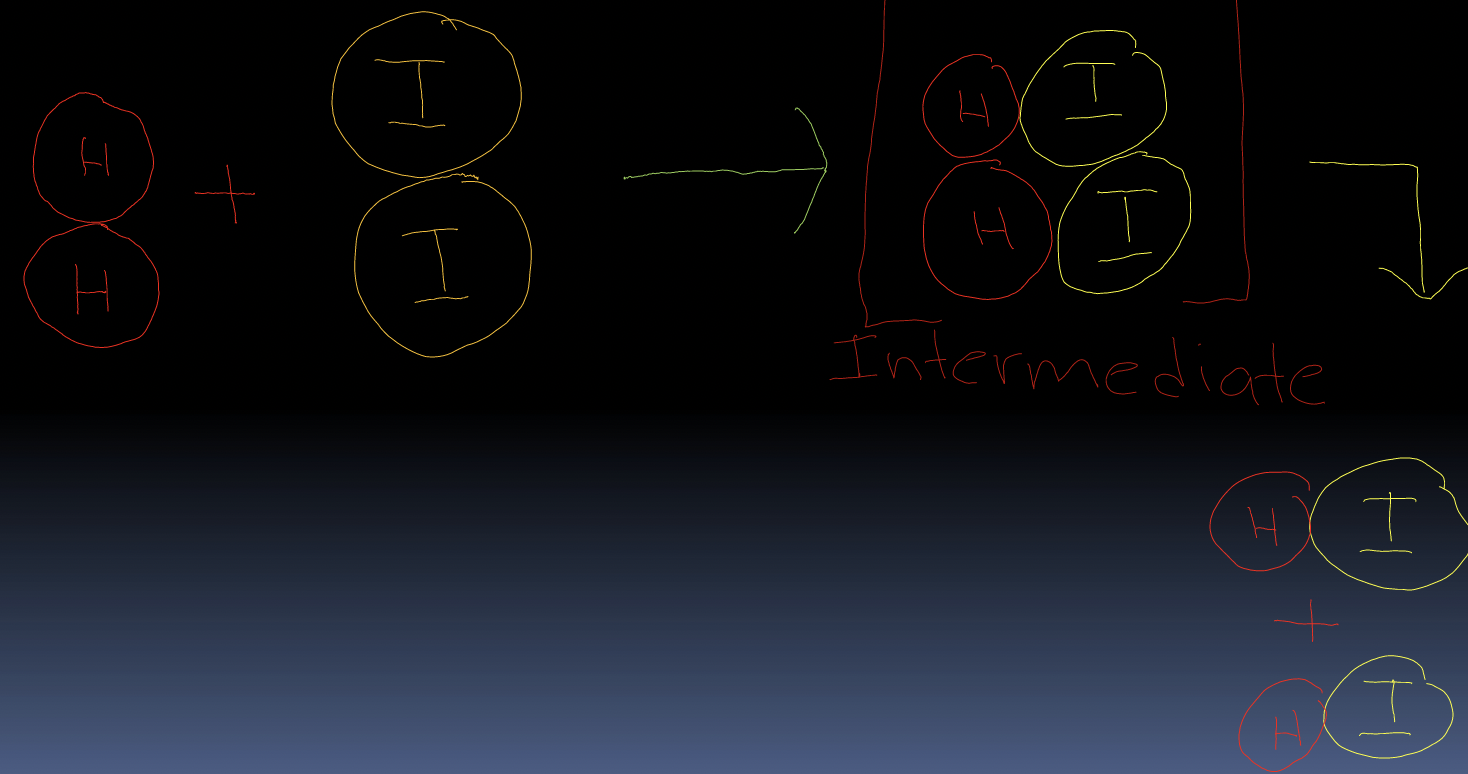
- Molecules act as balls that bounce off each other and transfer energy during collisions
For molecules to react there must be a collision
\
Concentration
If [ ] (concentration) increases more collisions are possible and reaction rate increases.
Temperature
If the temperature increases, then the kinetic speed of the molecule increases so the molecules start moving faster which makes them collide even more which increases the reaction rate.
\
Enthalpy and Chemical Reactions
Potential Energy
Stored energy within a bond. Position in space
Kinetic Energy
Energy in particles that are moving. Due to movement of the entire system or molecules.
Bond Energy
Energy required to break a bond between 2 atoms
Enthalpy = H
H = total kinetic and potential energy combined in a system at constant pressure.
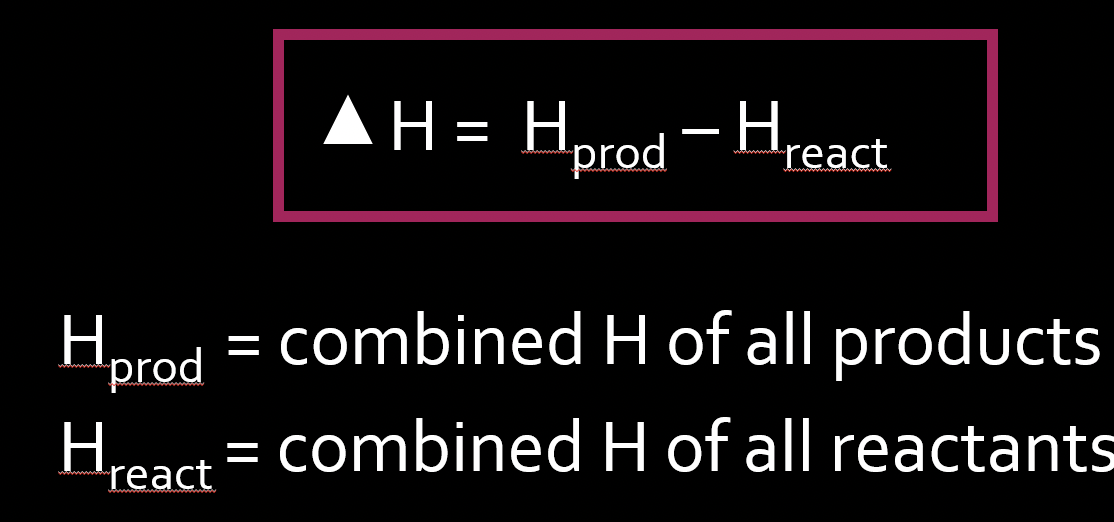 prod = final : react = initial
prod = final : react = initial
Check powerpoint for more
\
\
\
\