Water
The chemical reactions of all living things take place in an aqueous (water based) environment. Thus, water is one of the most important compounds found in living things.
A. Water: the single most abundant compound in most living things
B. Chemical formula: H20 (2 part hydrogen, 1 part oxygen)
C. Bohr model for water (covalent bonds)
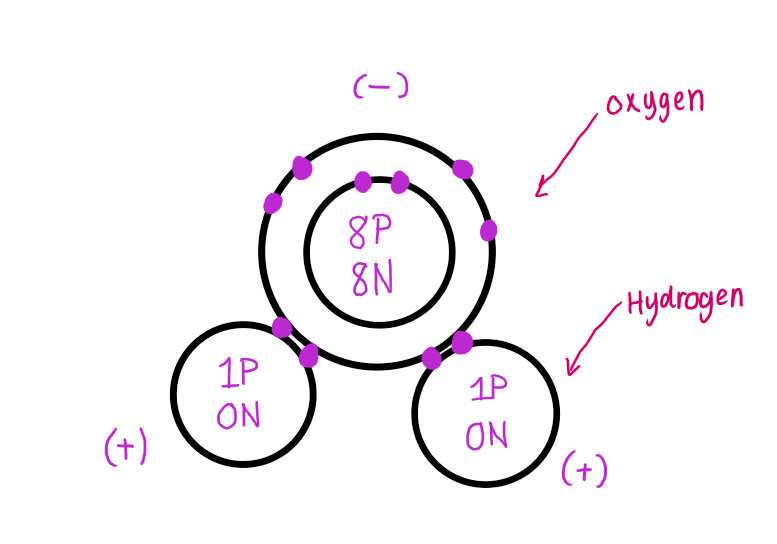
D. Water is a polar covalent compound
Polar: the electrons are unevenly shared between the hydrogen and oxygen atoms.
Oxygen pulls on the e- greater than the H so at any moment, the shared e- are more likely closer to the oxygen.
What effect does “polar” have
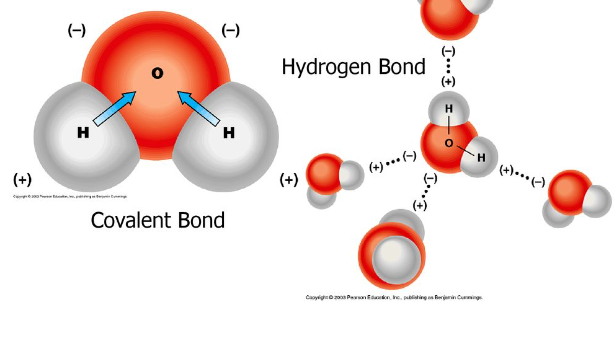
water has a partial positive pole and partial negative pole
like a magnet
E. Hydrogen bonding
the partial positive and partial negative charges allow H20 molecules to attract to each other or stick together through hydrogen bonds
Hydrogen bonds are weak bonds that can be broken
H-bonds are responsible for waters specials properties
F. Properties of water
Strong cohesion and adhesion forces
Cohesion = attraction between molecules of the same substance
Ex. cohesion leads to water having a high surface tension due to the hydrogen bonding of surface molecules to each other
Adhesion = attraction between molecules of different substances
Capillary action = the movement of water within spaces of a porous material due to the forces of adhesion, cohesion, and surface tensio
Surface tensions = The slightly elastic quality along the surface of water allows certain insects to walk on water.
Waters high specific heat capacity
Water can absorb heat and large amounts of thermal energy before its own
Water acts like a heat buffer for the globe
Heat capacity: it is caused by hydrogen bonding among water molecules, when heat is absorbed, hydrogen bonds are broken, and water molecules can move freely
Ice
ice is less dense than liquid water
Ability to form solutions
water is very effective in dissolving many other substances
Water is the universal solvent
Solution: liquid consisting of uniform mixture of two or more substances (Kool-Aid)
Solvent: Liquid (dissolving agent) (ex. water)
Solute: substance dissolved (ex. Kool-Aid powder/sugar)
G. Remember the rule to make a solution
Since water is polar, it will dissolve ions and other polar substances
Hydrophilic = water lover
Water will NOT dissolve non-polar substances