Chp 5 Thermochemistry
5.1 Energy Basics
thermochemistry - the amount of heat absorbed or released during chemical and physical changes
Energy - the capacity to supply heat or do work
work - energy required to move something against a force
potential energy | energy an object has because of its relative position, composition, or condition |
kinetic energy | energy that an object possesses because of its motion |
kinetic energy →thermal energy - kinetic energy associated with the random motion of atoms and molecules
Heat (q) - transfer of thermal energy between two bodies at different temperatures
exothermic process | change that releases heat |
endothermic process | change that absorbs heat |
Ways of measuring energy?
calories (cal) - energy required to raise one gram of water by 1 degree C (1 kelvin)
Calorie - for food. capital C means kilocalorie
joule (J) - amount of energy used when a force of 1 newton moves an object 1 meter
1 J = 1 kg m2/s2
1 calorie =l 4.184 joules
heat capacity | 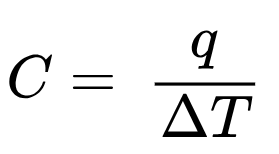 | body of matter is the quantity of heat (q) it absorbs or releases when it experiences a temperature change (ΔT) of 1 degree Celsius (or equivalently, 1 kelvin)
|
SPECIFIC heat capacity | 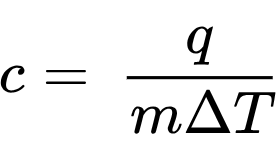 | quantity of heat required to raise the temperature of 1 gram of a substance by 1 degree Celsius (or 1 kelvin)
|
another kind of specific heat capacity
molar heat capacity - intensive property. heat capacity per mole of a particular substance and has units of J/mol °C
if the mass of a substance is known then:
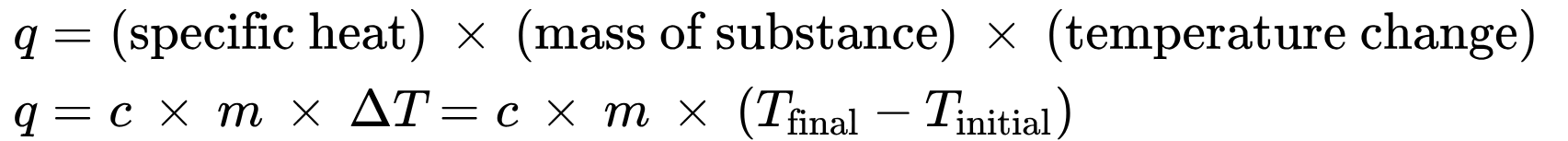
5.2 Calorimetry
calorimetry - measure the amount of heat involved in a chemical or physical process
system - substance(s) undergoing chem or physical change
surroundings - all other matter, including measurement tools, that provide or absorb heat from the system
ideally a calorimeter only measures the heat transfer between the substances & has no energy influence from the surroundings
aka the heat gained by substance M = the heat lost by substance W
so there is the same idea when
bomb calorimeter - used to measure energy produced by reactions w lots of heat & gaseous products. (e.g. combustion reactions)
nutritional calorie (Calorie) - 1 Calorie = 1000 calories, the amount of energy need to heat 1 kg of water by 1 °C
5.3 Enthalpy
internal energy (U) / (E) - the total of all possible kinds of energy present in a substance