saturated vs unsaturated
Saturated hydrocarbon
Only single bonds
Maximum number of hydrogens attached
Unsaturated hydrocarbon
Has double or triple bonds
Fewer hydrogens attached
Why it matters (real life)
Saturated fats
Straight chains
Pack tightly
Sold (butter)
Unsaturated fats
Bent chains (kink from double bond)
Don’t pack tightly
Liquid (oil)
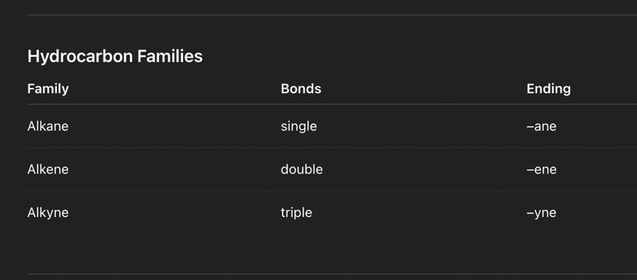
Alkenes
Contain at least one double bond
General formula:CnH2n
Number in a name = position of double bond
Example
Pent-2-ene — double bond starts on carbon 2
Alkynes
Certain triple bond
Ending: -yne
Isomers
Same molecular formula
Different arrangement of atoms
Different properties
example
pent-1-ene vs pent-2-ene
Key idea
Bond type determines physical properties
Structure affects behaviour of substances