Mineralogy Midterm!!!
Mineral: a naturally occurring, crystalline solid, with a definite chemical composition. >6000 minerals to date
**Glass is not a mineral because it lacks long-range atomic order (non-crystalline)
Biomineral: minerals that constitute an integral part of biologic structures. (ex. calcite and aragonite CaCO3, tooth enamel made by hydroxylapatite!)
Mineraloid: minerals that lack long-range crystalline structure, including amorphous solids and glasses.
Crystal: mineral bounded by regular crystal faces produced as the crystal grew.
Mineral Formulas: Cations first and in order of decreasing coordination number!!!
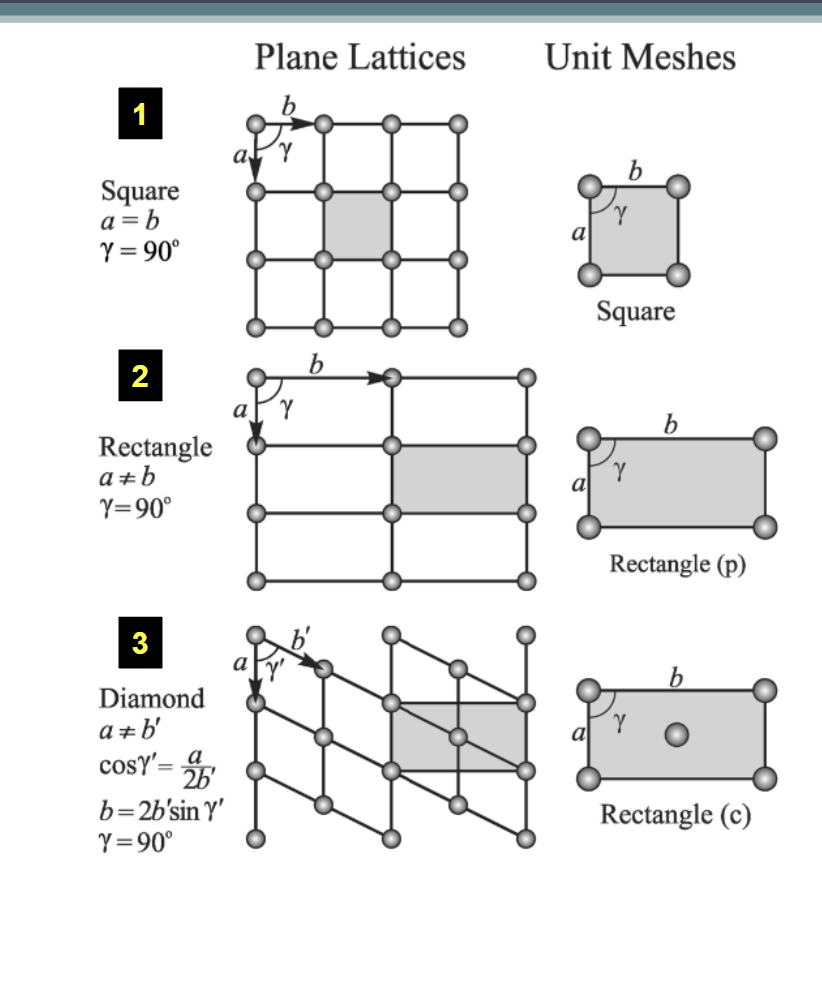
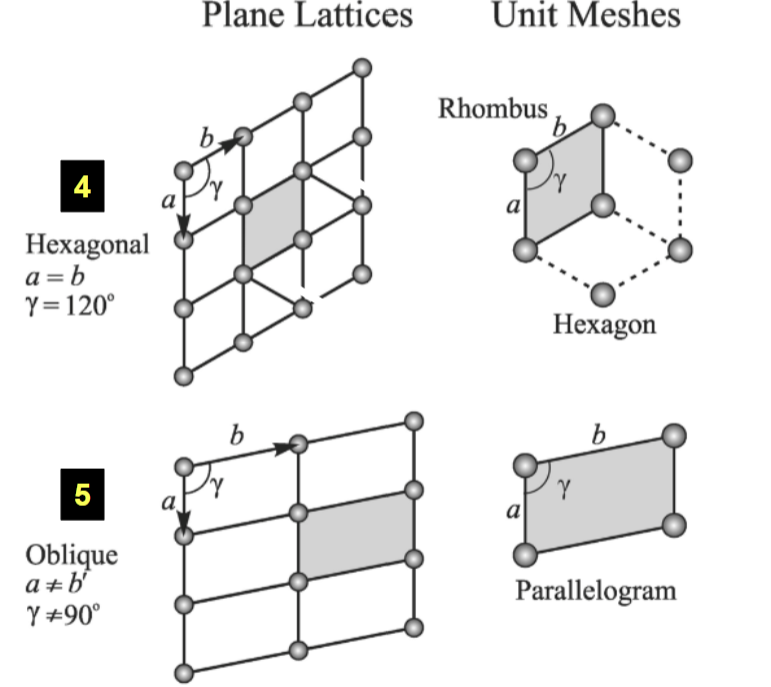
Unit Meshes:
Crystal Systems
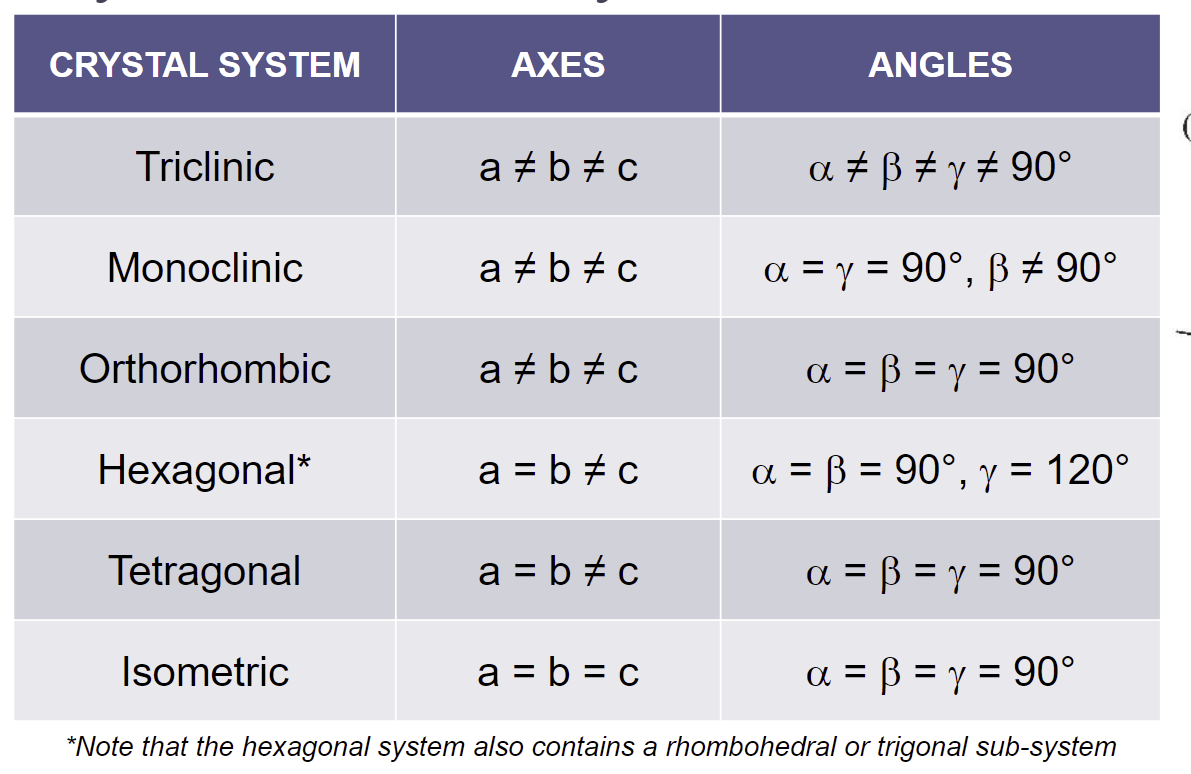
CHEAT SHEET
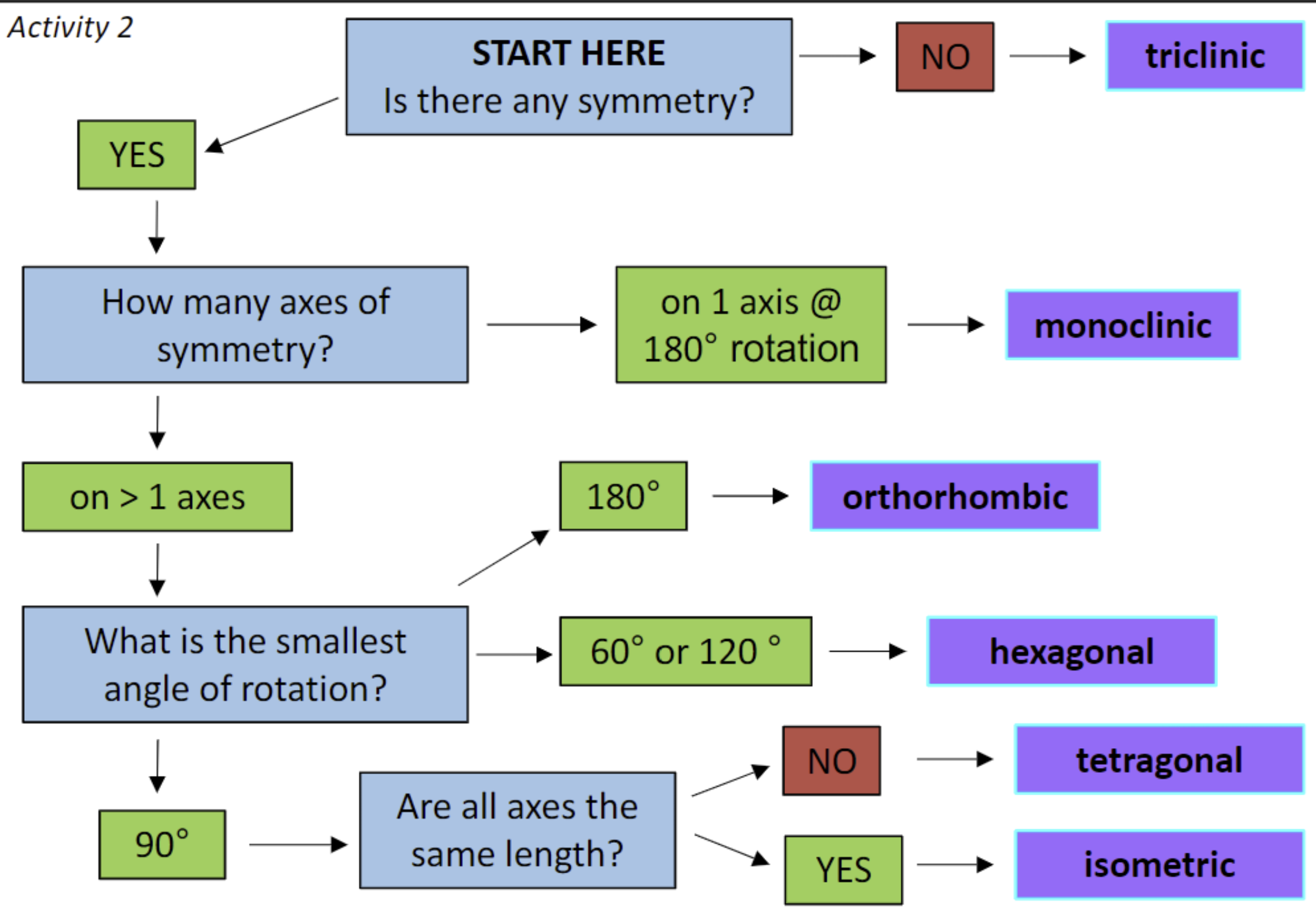
Triclinic —> no symmetry!! (ex. K-feldspar)
Monoclinic —> 1 axis of symmetry, all axes are different (ex. orthoclase, muscovite)
Orthorhombic —> all axes different, but all angles are equal
Tetragonal —> one axes is different length, but all angles are equal
Hexagonal (Trigonal, Rhombohedral) —> self explanatory
Isometric (cubic) —> self explanatory
Crystal Chemistry
Ionic Bond: Electrostatic attraction between positive and negative ions
Covalent: Sharing of electrons when atomic orbitals overlap
Metallic Bonding: lattice of cations in a sea of delocalized electrons
OXIDATION IS LOSS. REDUCTION IS GAIN. (OF ELECTRONS)
PROTONS (ATOMIC NUMBER Z) DEFINES ELEMENT!!!
Crystal Structure + Pauling’s Rules
Coordination Principle: coordination number (number of nearest cation neighbors is determined by radius ratio (radius of cation over radius of anion)
Electrostatic Valency Principle: strength of bond = cation charge / coordination number (ei. the larger the charge and the closer the bond, the stronger)
Sharing of Polyhedral Elements 1: corners < edges < faces shared destabilize the structure the most
Sharing of Polyhedral Elements 2: if there are differently charged cations, the highly charge cations minimize the number of anions to keep a max distance apart
Principle of Parsimony: crystal constituents are simple.
Isostructural: two or more minerals whose atoms are arranged in same type of crystal structure (ex. NaCl and PbS have same symmetry, cleavage, and crystal habit (isometric))
Polymorphism: the ability of a chemical compound to crystallize into more than one structure (ex. SiO2 => quartz, tridymite, coesite)
high pressure => high density structures, low pressure => low density structure
Reconstructive: REQUIRES ENERGY TO BREAK BONDS then rearranges atoms/ions into new structure
Displacive: NO BREAKING BONDS just bending of crystal structure (ex. alpha quartz becomes beta quartz at high temperatures)
Order-Disorder: RAPID COOLING —> cation distribution disorder, SLOW COOLING —> order (ex. K-feldspar sanidine is rapid cooling and disordered, K-feldspar microcline is slow cooling and ordered (Si and Al tend to be the same sites))
rapid cooling —> volcanic igneous, slow cooling —> plutonic igneous
Mineral Growth
NO QUARTZ IN MAFIC ROCKS (basalt), QUARTZ AND OTHER SILICA POLYMORPHS IN FELSIC VOLCANIC ROCKS (rhyolite)
Simple Substitution: cations with same charge and size
Coupled Substitution: basically simple substitutions
Omission: ions with different charge leave sites vacant (ex. swapping 3 Fe2+ for 2 Fe3+)
Interstitial: ions with different charge, extra ions placed in normally vacant sites (ex. swapping Si4+ with Al3+ and K+)
Homogeneous Nucleation: atoms/ions find each other spontaneously and randomly form the nucleus of a crystal
Heterogeneous Nucleation (dominant mechanism): new minerals nucleate by taking advantage of an existing crystal structure
Contact Twinning: twinning from one point
Penetrative Twinning: crystals directly overlap and are intergrowth (related by rotation) (ex. staurolite, pyrite)
X-Ray Crystallography
Powder X-ray Diffraction: finely ground powder
X-rays generated when stream of high energy electrons strike material, diffraction occurs when electrons hit atoms of object
d = distance between atoms in a crystal structure
Diffraction intensity differs among minerals because it depends on number and kinds of atoms present (the heavier the element, the more electrons, thus the more diffraction)
Physical Properties
Zircon: ZrSiO4, tetragonal, prismatic crystals
Transition elements are mainly responsible for colour in minerals
Colours:
Idiochromatic: “self-coloured” due to compositon
Allochromatic: coloured due to impurities
Pseudoachromatic: “false coloured” due to light diffraction tricks
Fluorescence: when a higher energy electron drops into a lower energy level, releasing energy in the form of EMF causing photoluminescence
Magnetism: due to unpaired electrons in orbitals
Curie Temperature: temp at which magnetic material lose magnetism when heated
For magnetite: 585 C
Magnetite acquires magnetization parallel to earth’s magnetic field
Cleavage:
Perfect: breaks easily
Good: breaks easily but not on even/continuous surfaces
Distinct/Indistinct/Poor: decreasing quality of break surfaces
Types:
Cubic (galena)
Octahedral (fluorite)
Rhombohedral (calcite)
Prismatic (amphiboles)
Basal (micas)
Mineral Classification
Mineral Class: mostly based on anions or anionic properties
(Silicate) Mineral Subclass: bonding of tetrahedra is used to group structurally similar minerals (ex. tectosilicates) —> depends on how many O2- are shared between tetrahedra
Mineral Group: two or minerals with similar structure and chemically similar elements (feldspars within tectosilicates)