chemical changes
Metal Oxides
metal + oxygen = metal oxide
oxidation because metals gain oxygen
reduction = loss of oxygen; oxidation = gain of oxygen
The Reactivity Series
when metals react, they form positive ions
the reactivity series arranges the metals in order of their tendency to form a positive ion (reactivity)
metals can be put in order from their reactivity with water and dilute acids
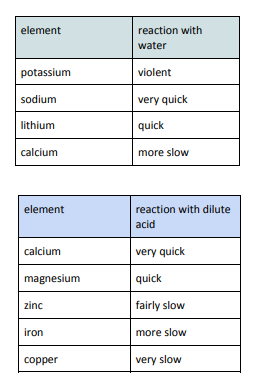
a more reactive metal can displace a less reactive metal from a compound
Oxidation & Reduction in terms of electrons
Oxidation is Loss of electrons; Reduction is Gain of electrons (OIL RIG)
sodium oxidised = Na → Na+ + e-
sodium+ reduced = Na+ + e- → Na
Identify elements which have been oxidised & reduced
eg. 2Na + 2HCl → 2NaCl + H2
write out ions
2Na + 2H+ + 2Cl- → 2Na+ + 2Cl- + H2
2Na → 2Na+ + 2e- so oxidised
2H+ + 2e- → H2 so reduced
2Cl- → 2Cl- so neither
Reactions of acids with metals
acid + metal → salt + hydrogen
redox reaction = one substance reduced & one oxidised
eg. 2HCl + 2Na → 2NaCl + H2
Neutralisation of acids
acids are neutralised by alkalis and bases
eg. metal hydroxides, metal carbonates & metal oxides
an alkali is a base dissolved in water
metal hydroxide + acid → salt + water
eg. 2NaOH + 2HCl → 2NaCl + 2H2O
metal carbonate + acid → salt + water + carbon dioxide
eg. CuCO3 + H2SO4 → CuSO4 + H2O + CO2
metal oxide + acid → salt + water
eg. Na2O + 2HCl → 2NaCl + H2O
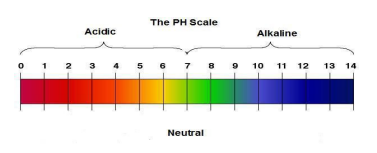
The pH scale & neutralisation
H+ + OH- → H2O
acids produce hydrogen ions in aqueous solution
alkalis produce hydroxide ions in aqueous solution
Strong & Weak acids
strong/weak is not the same as concentrated/dilute
strong acids completely ionise in aqueous solution
eg. hydrochloric, nitric and sulfuric acids
weak acids partially ionise in aqueous solution
eg. ethanoic, citric and carbonic acids
the stronger the acid, the lower the pH
as pH decreases by one unit, H+ concentration increases by a factor of 10
concentration refers to the amount of substance in a given volume whereas strength refers to the concentration of H+ in aqueous solution
Electrolysis
when an ionic substance is melted/dissolved, the ions are free to move within the liquid
electrolysis is passing a current through a molten solution so it can be broken down into its elements; the molten solution is called an electrolyte
positive ions move to negatively charged electrode - cathode (reduction: gain of electrons)
negative ions move to positively charged electrode - anode (oxidation: loss of electrons)
ions are discharged at each electrode
when a simple ionic compound is electrolysed in its molten state using inert electrodes, the metal is produced at the cathode and the non-metal is produced at the anode
Using Electrolysis to Extract Metals
metals that are more reactive than carbon are too reactive to be extracted by reduction with carbon so they’re extracted by electrolysis of molten compounds
metals that react with carbon can also be extracted by electrolysis
large amounts of energy are used in extraction for melting the compounds and produce the electrical current
aluminium is manufactured by the electrolysis of aluminium and cryolite using carbon as the anode
aluminium oxide is mixed with cryolite to lower the melting point as aluminium oxide is too expensive to melt
the positive electrodes need to be continually replaced because oxygen is formed which reacts with the carbon forming carbon dioxide and causing the cathode to wear
Electrolysis of Aqueous Solutions
ions discharged when an aqueous solution is electrolysed with inert electrodes depend on the relative reactivity of the elements involved
at the negative cathode, hydrogen is produced unless the metal is less reactive than hydrogen because more reactive ions want to stay within the solution
at the positive anode, if hydroxide and halide ions are present, one of the haline ions will be produced - if no halide is present oxygen is formed
this happens in aqueous solution as water molecules break down producing H+ ions and OH- which get discharged
Representation of Reactions as Half Equations
negative electrode (cathode)
X+ + e- → X
positive electrode (anode)
X- → X + e-