1 | EMS
bohr model and the quantum mechanical model both explain how electrons exist in atoms and how those electrons affect the atons and how thosew electrons affect the chemical and physical properties of elements
Niels Bohr and Erwin Schrödinger and Albert Einstein all played a role in the development of quantumn mechanics but were also confused buy their own theory of wave duality for the electron
Light is a form of electromagnetic radiation
Equation: 3.0 x 10^8 (m/s) or (186,000 mi/s)
Light has properties of both WAVES and PARTICLES
A wave is a disturbance that can travel through a medium from one location to another
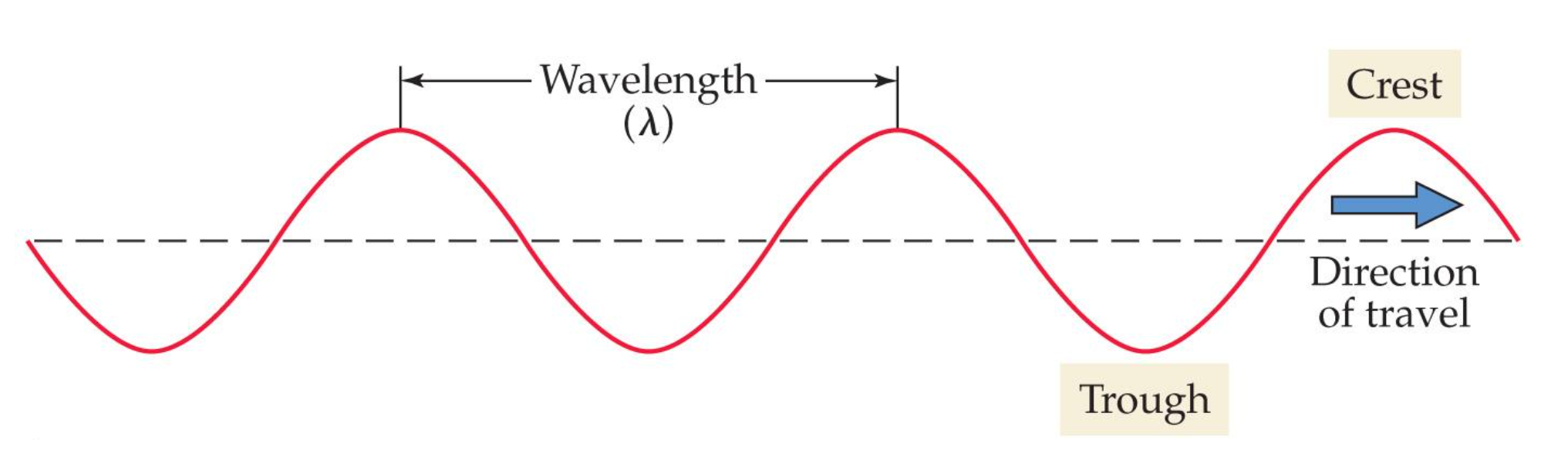
properties of waves include:
wavelength |
represented by lambda → λ
describes the distance between adjacent wave crests
color is also dependent on wavelength with red light having the longest wavelength of 750 nm and violet light havent the shortest wavelength of 400 nm
(1 nm = 1 x 10^-9 m)
\
\
\
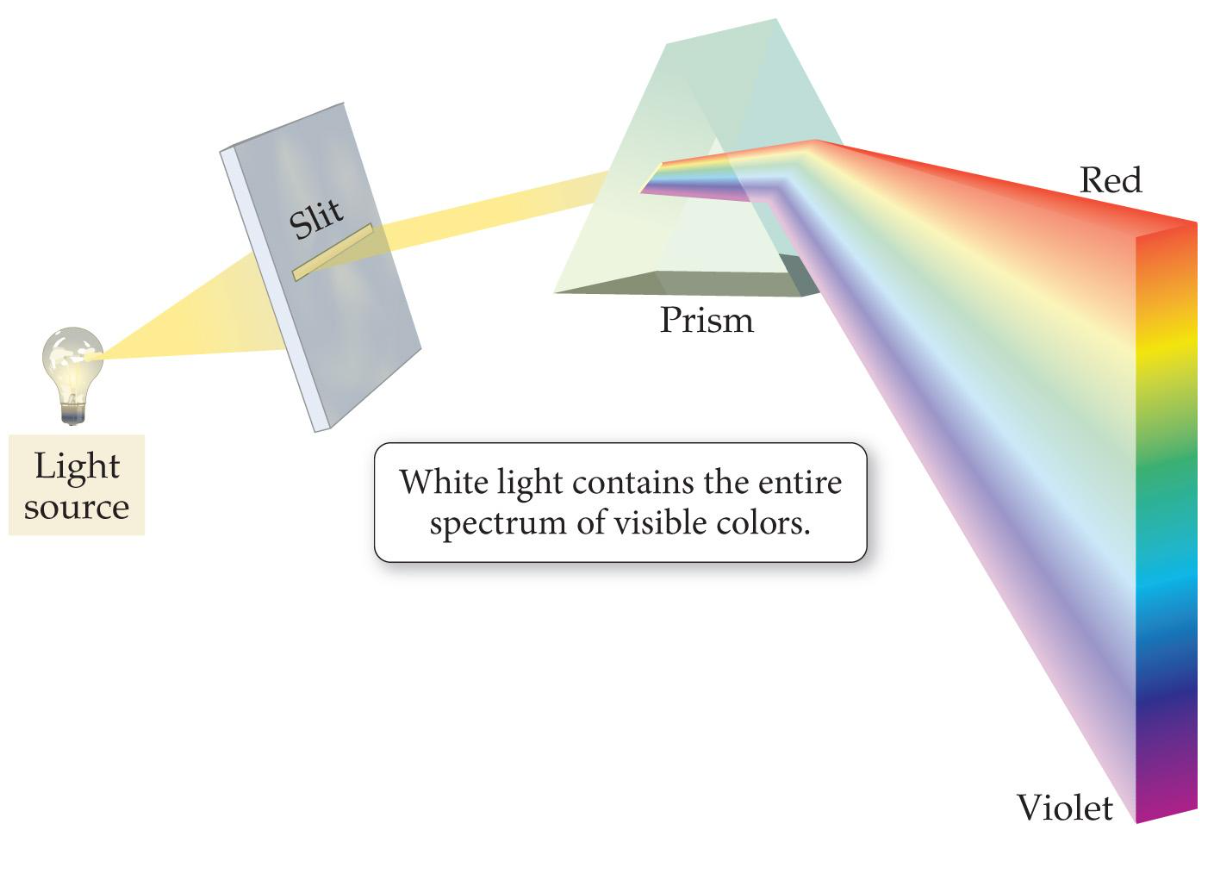
\ \ color |
light is seperated by color when it is passed through a prism
A red shirt may appear red because it reflects red light while absorbing the other colors and our eyes can only see reflected light
\
\
\ \ frequency |
represented by → v
the number of cycles or crestss that pass through a stationary point in one second
\ the shorter the wavelength the higher the frequency
particles of light |
Light can also be seen in a stream of particles
a particle of light is a photon
The amount of everygy carried in a photon depends on the wavelength of the light - the shorter the wavelength the greater the energy
light waves carry more if their crests are closer together (higher frequency shorter wavelength)
violet light carries more energy per photon than red light because biolet light has a shorter wavelength
\ the electromagnetic spectrum |
the electromagnetic spectrum shows the waves with long and low frequency wavelengths on the right and short wavelength and high frequencies on the right and visible light is on the small sliver in the middle
\
\
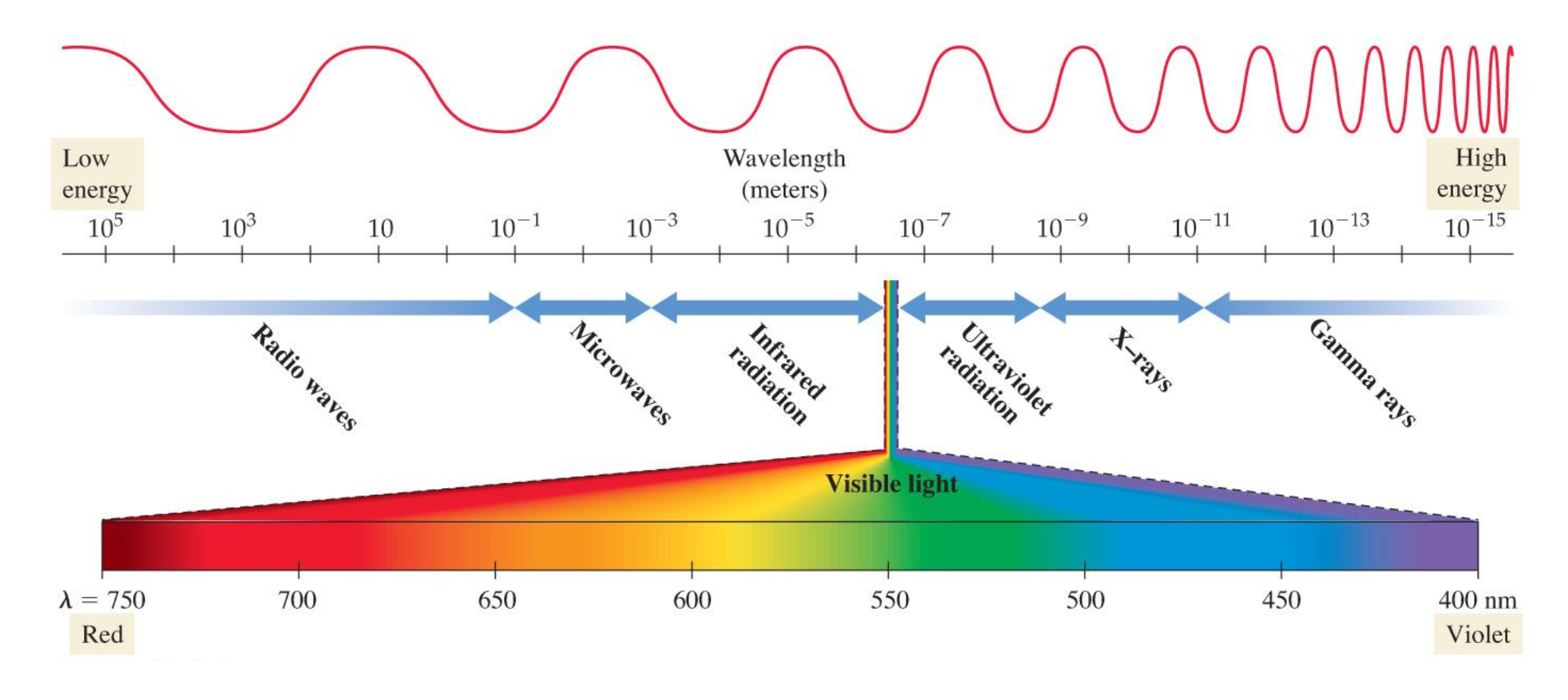
\ \ the shortest wavelength and the most energetic photons are those of gamma rays which are produced by the sum, stars, and unstable atomic nuclei on earth. Excessive human exposure is dangerous and can hurt biological molecules
x-rays are longer wavelengths but less energy than gamma rays. these are found from meducal use. Like gamma ray photons, these can carry enough energy to damage biological molecules (x-rays and gamma rays are sometimes called ionizing radiation because the high energy in their photons can ionize atoms and molecules which are usually used to destroy cancer cells)
UV or ultraviolet light is a component of sunlight that causes sunburn or suntan. Even though they are nto as energetic as gamma-ray or x-ray photons, UV photons carry enough energy to damage biological molecules
visible light ranges from violet to red light and photons of visible light do cause molecules in our eyes to rearrange which sends a signal to our brains that result in vision
infared light is the heat you feel when you place your hand near a hot object. All warm objects (including human bodies) emit infared light. Even though we can see infared light, sensors can detect it and are often used in night vision to see in the dark
microwaves are used for radar and in microwave ovens. This light has longer wavelengths and lower evergy per photon than visible or infared light
the longest wavelengths are radio waves which are used to transmit signals used by am and fm radio and other forms of communication