Enthalpy
Enthalpy change
When bonds break or form during a reaction, energy is often released or absorbed as heat, this heat change at a constant pressure is known as an enthalpy change
Exothermic vs Endothermic
Exothermic - release heat and have a negative enthalpy change (∆H), temperature of surroundings increases
Endothermic - absorbs heat from surroundings and have a positive enthalpy change, temperature of of surroundings decreases
Enthalpy Diagrams
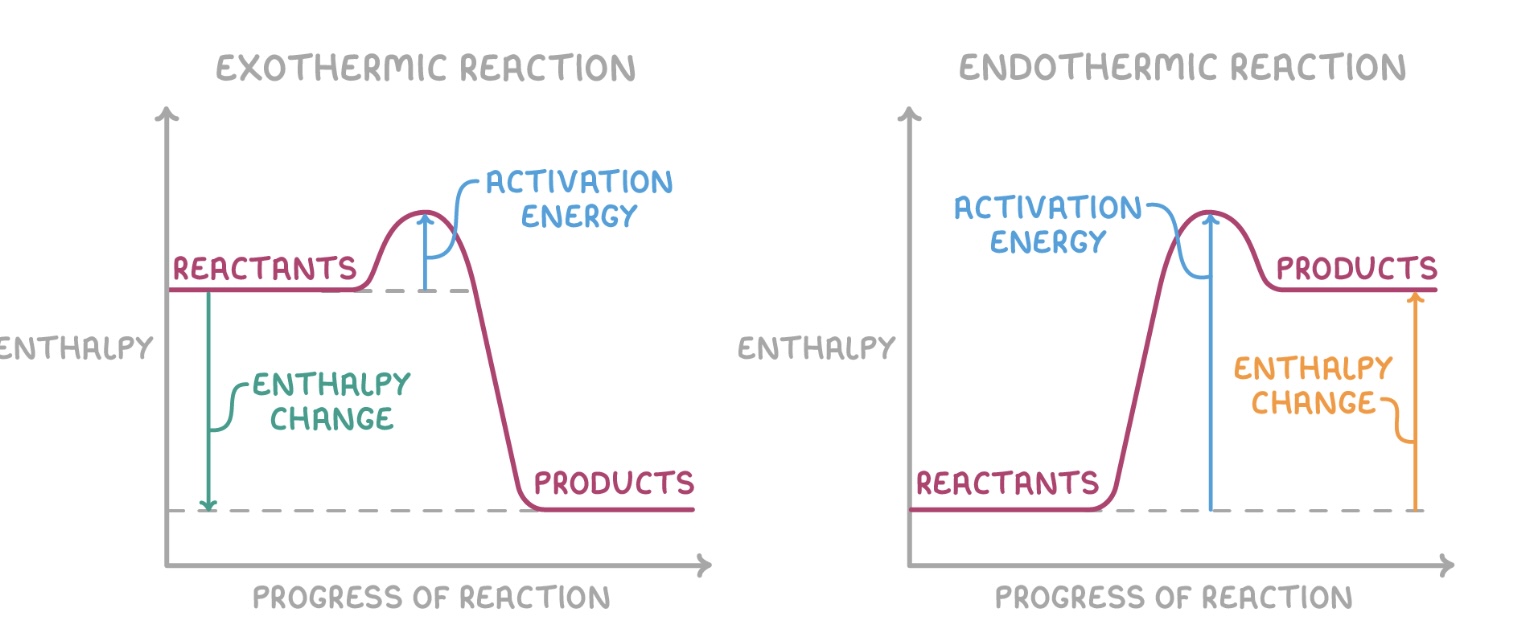
Standard conditions
temperature = 298K, Pressure = 101kPa
Types of standard enthalpy changes
Standard enthalpy of formation - the enthalpy change when 1 mole of a substance is formed from its constituent elements under standard conditions with all reactants and products in their standard states
Standard enthalpy of combustion - enthalpy change when 1 mole of a substance is completely burned in oxygen