Lecture 14: Equilibrium and Kc Concepts
Reversible Reactions
Definition: Reversible reactions can proceed in both forward and backward directions.
Example:
Hemoglobin (Hb) binds to oxygen (O2) reversibly, forming Hb.(O2)4.
Reaction:

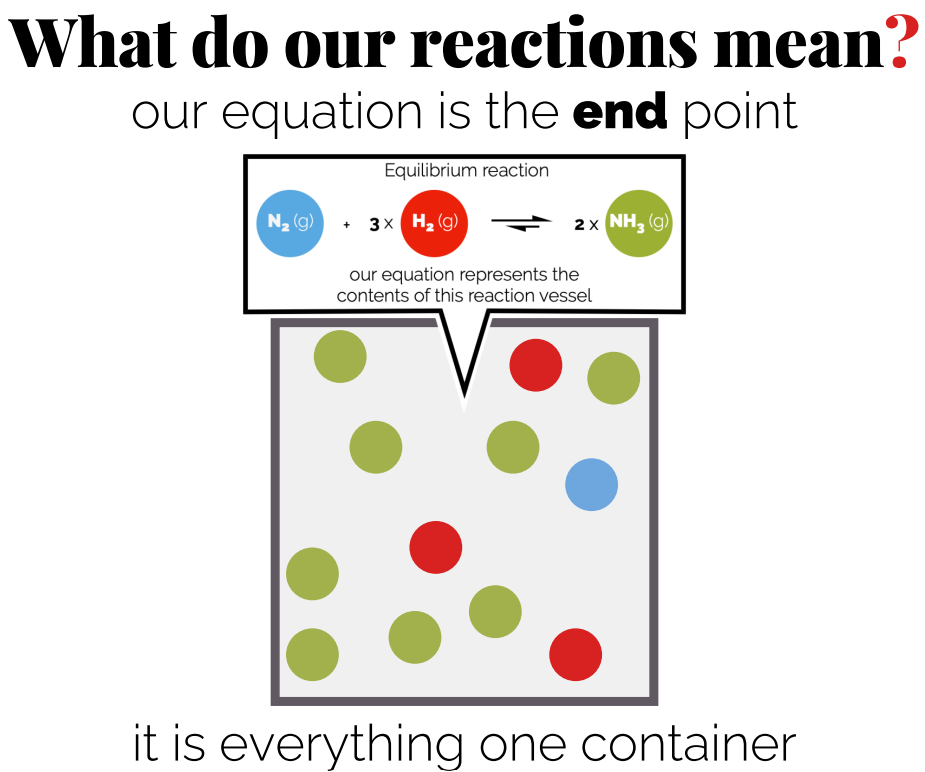
Chemical Equilibrium
Definition: Chemical equilibrium occurs when the rate of the forward reaction equals the rate of the reverse reaction.
You are getting to the point where the rate of reactions are the same , not the concentrations.
The concentrations of reactants and products do not need to be equal.

Equilibrium Constants (Kc)
General Formula:
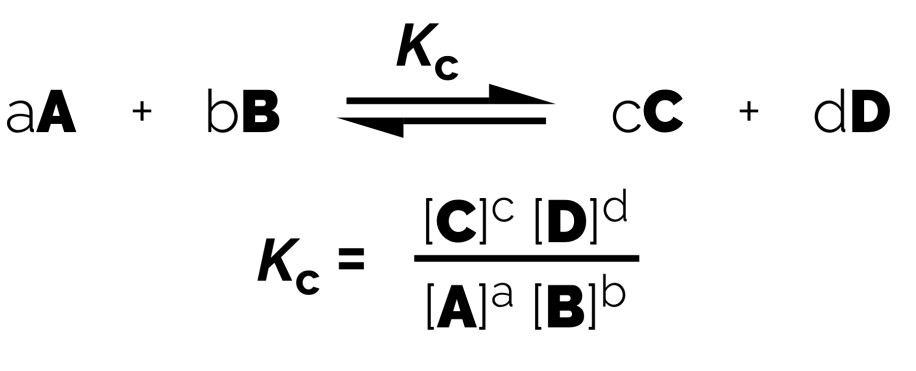
The lowercase letters is showing the number of moles: this becomes a power in the expression.
The uppercase letters represents are concentrations.
Products over reactants.
Always double check the equation is balanced before forming your expression.
Example
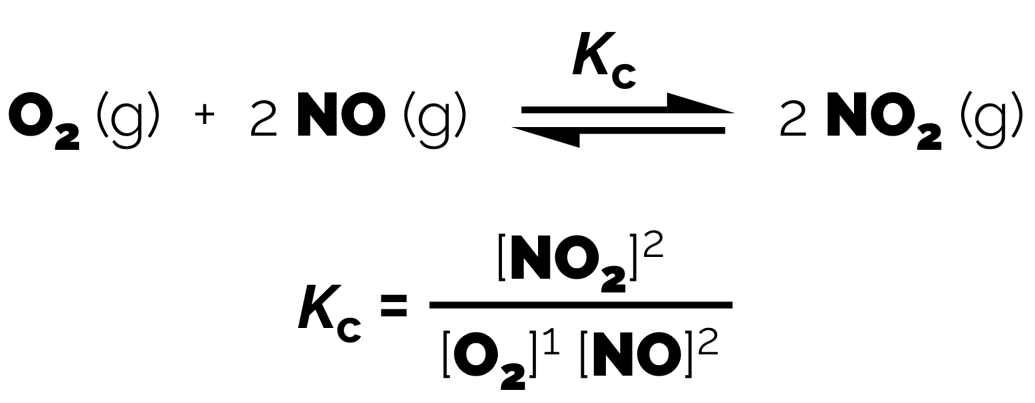
Key Characteristics:
Expresses the relationship between the amounts of products and reactants (Ratio of how many products versus how many reactants)
Kc has no units
Kc is constant at a given temperature
Pure solids and liquids (only don’t include aqueous solutions where it is not the bulk solvent) are not included in the Kc expression.
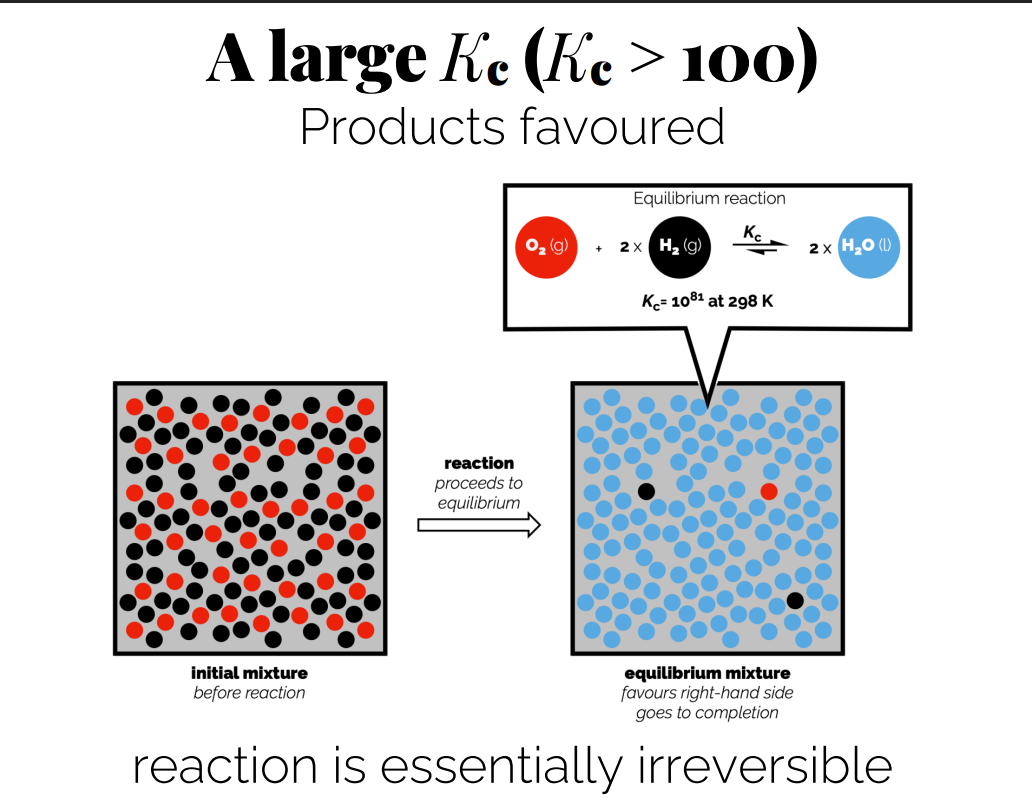
Interpretation of Kc Values
Large Kc (Kc > 100): Indicates that products are favored at equilibrium; the reaction proceeds towards completion.
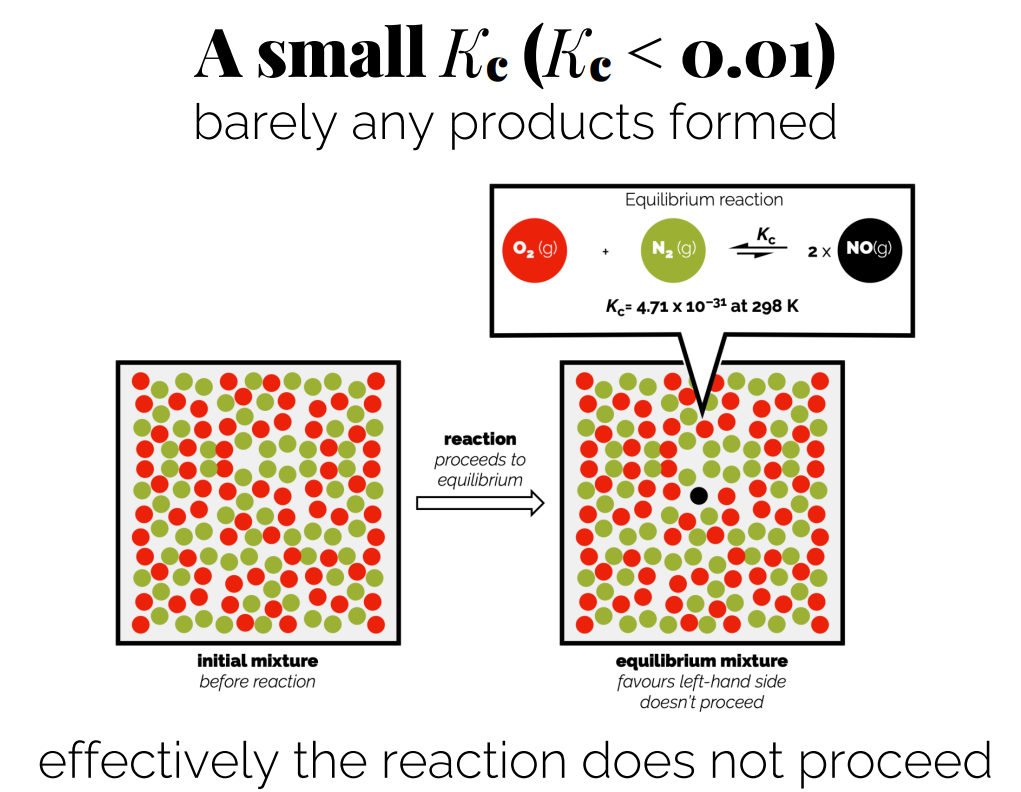
Small Kc (Kc < 0.01): Indicates that reactants are favored; little to no products are formed.
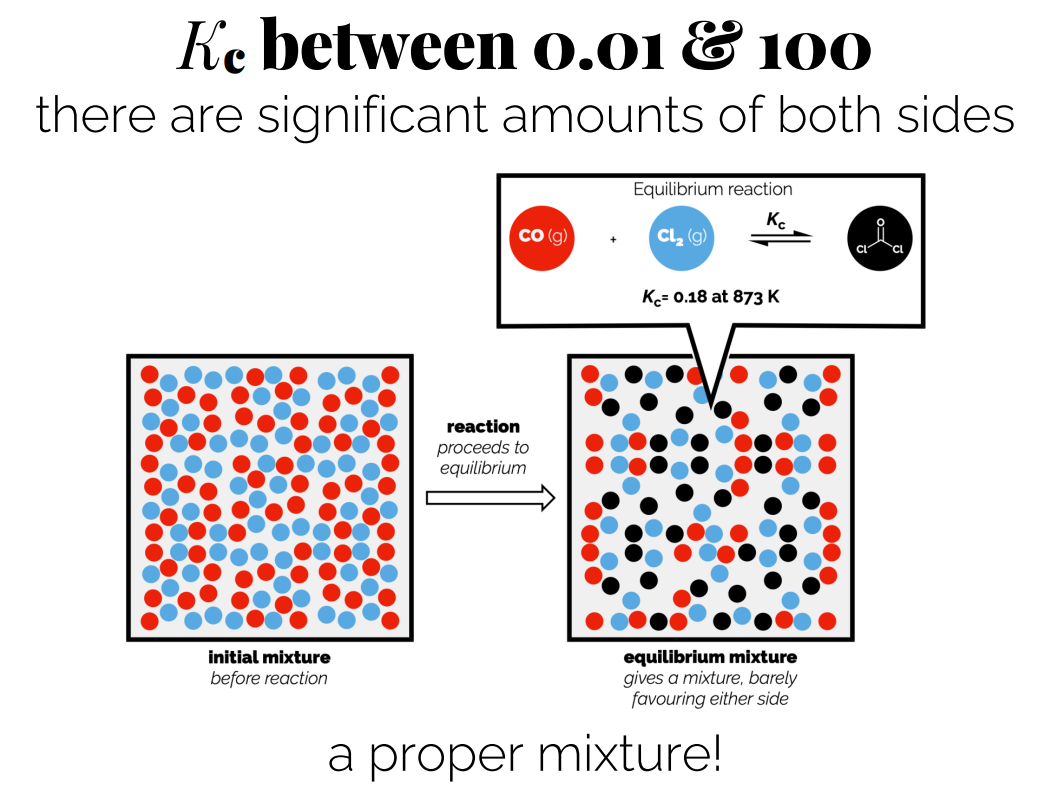
Kc between 0.01 and 100: Significant amounts of both products and reactants, a mixture.
(If Kc =1 then products=reactants).

Factors Affecting Kc
Kc is influenced by temperature changes; however, altering concentrations or pressure in a dynamic equilibrium does not change Kc.
Calculating Kc from Concentrations
Balance the equation
Form Kc expression
Calculate Kc by plugging in the concentrations numbers
Example Calculations:
