Grade 10 Review
Polyatomics
phosphate | PO43- |
|---|---|
carbonate | CO32- |
sulfate | SO42- |
bicarbonate | HCO3- |
chlorate | ClO3- |
nitrate | NO3- |
ammonium | NH4+ |
bromate | BrO3- |
iodate | IO3- |
cyanide | CN- |
hydroxide | OH- |
acetate | C2H3O2- (CH3COO-) |
hydrodium | H3O+ |
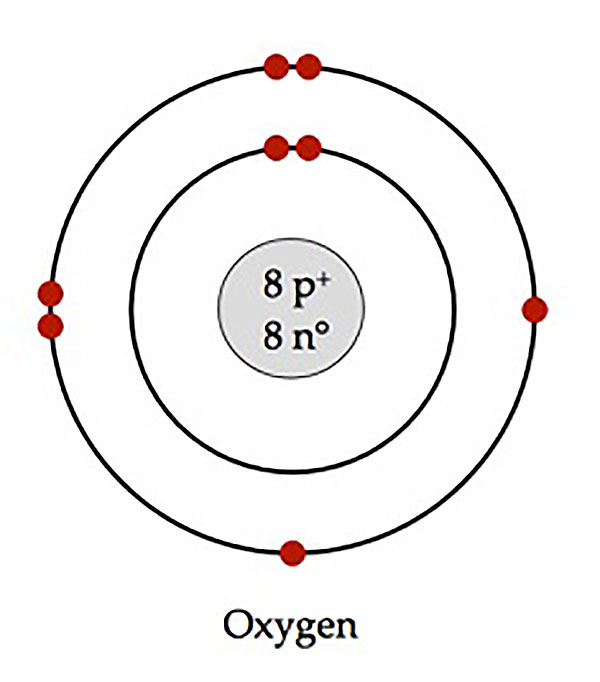
Bohr -Rutherford Diagrams
shows number and location of subatomic particles
Lewis Dot Diagrams
shows valence electrons on valence shells

Ions
ion = a charged atom that gains or loses electrons to get a full outer shell
all noble gases are stable
electron configuration
Metals
loses electrons, become cations (positive)
imbalance of protons and electrons
easier to lose one than gain seven
charge = group number
Non-Metals
gain electrons, become anions (negative)
imbalance of protons and electrons
charge = group number - 8