Acids and Bases
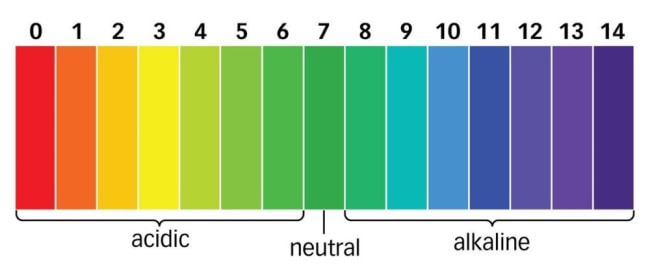
pH scale:
Ranges from 0 to 14, with 7 being neutral.
Values below 7 indicate acidic solutions, while values above 7 indicate basic solutions.
The further the pH value is from 7, the stronger the acid or base, respectively, with 0 being the strongest acid and 14 being the strongest base. This can be used to indicate the strength of an alkali or acid but only if the acids are at the same concentration.
pH range:
0 - 3, strong acid - Hydrochloric acid
4 - 6, weak acid - Ethanoic acid
7, neutral - Water
8 - 10, weak alkali - Ammonia solution
11 - 14, strong alkali - Sodium hydroxide
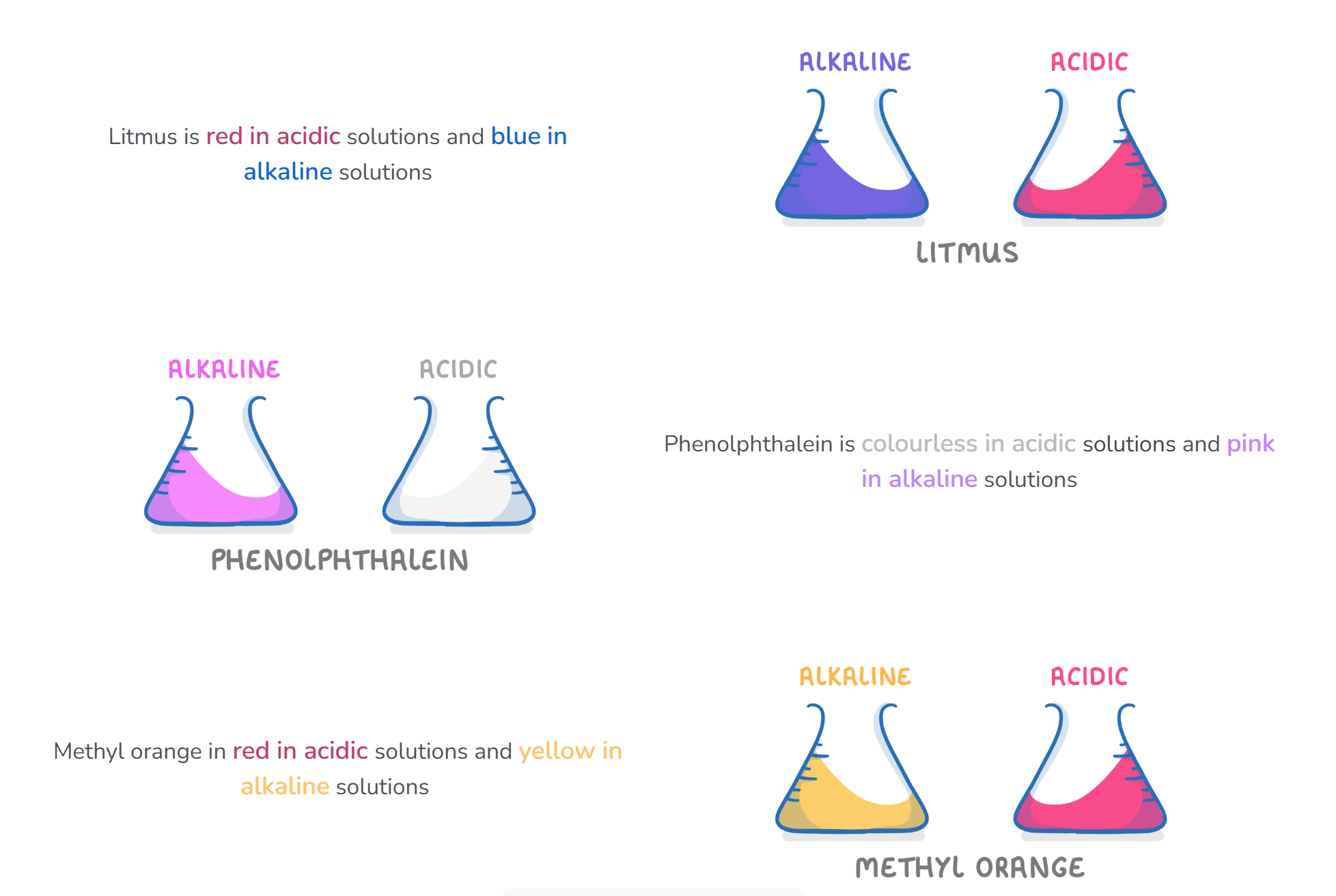
Different indicators have different colours depending on whether the solution is acidic, alkaline or neutral but they do not show the pH
INDICATOR | ACID | NEUTRAL | ALKALI |
litmus solution | red | purple | blue |
phenolphthalein | colourless | colourless | pink |
methyl orange | red | yellow | yellow |
red litmus | stays red | stays red | turns blue |
blue litmus | turns red | stays blue | stays blue |
METAL HYRDOXIDE + ACID ——— SALT + WATER
NaOH + HCl ——— NaCl + H2O
METAL OXIDE + ACID ——— SALT + WATER
MgO + 2HCl ——— MgCl + H2O
METAL CARBONATE ——— SALT + WATER + CARBON DIOXIDE
AMMONIA + ACID ——— AMMONIUM SALT

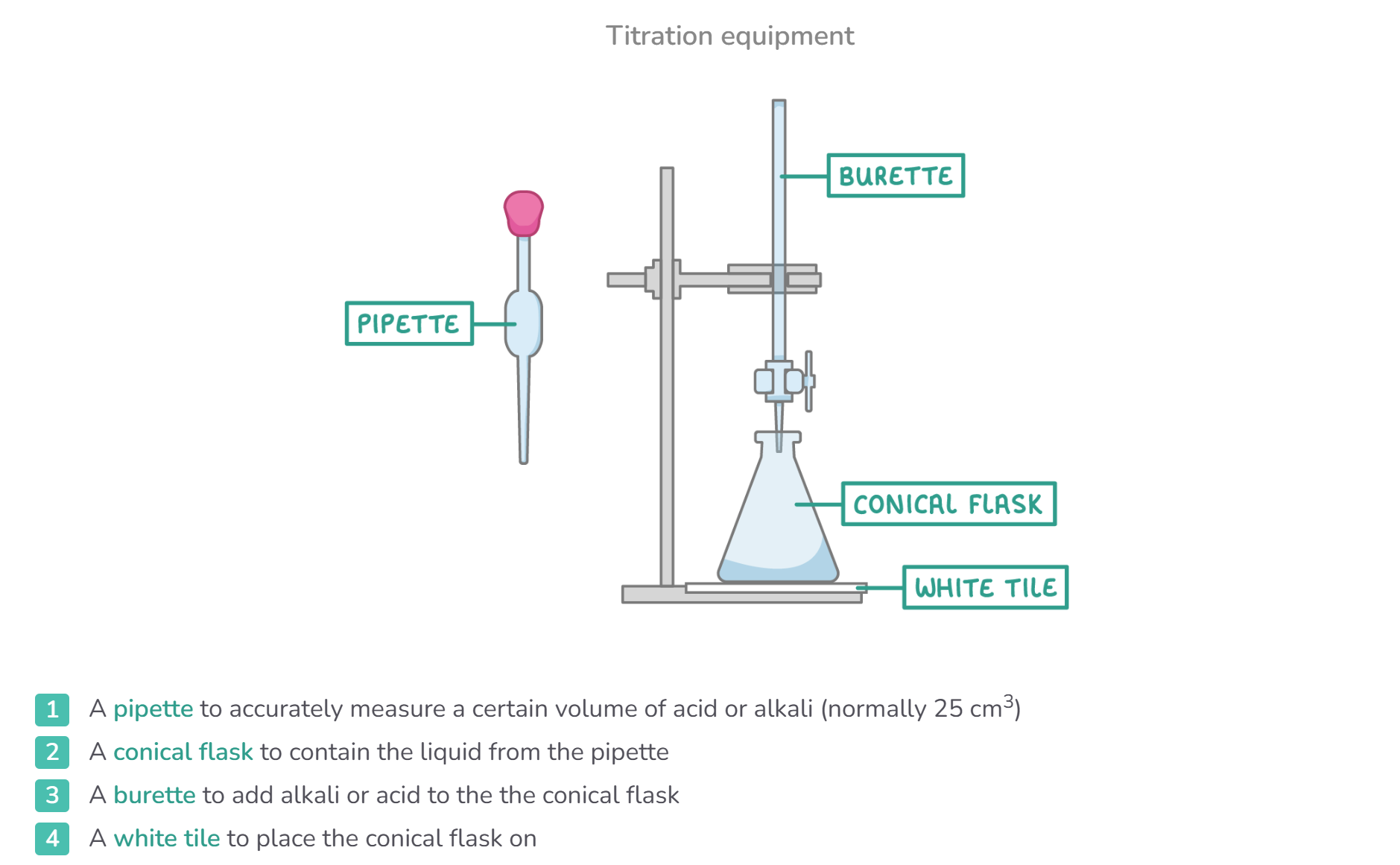
TITRATION