Osmosis in Plant Cells
In plant cells the water potential depends not only on the solute concentration in the cytoplasm and vacuole, but also on the pressure potential generated by the cell wall. As cells become turgid the cytoplasm pushes against the cell wall which resists further expansion, exerting a pressure on the cytoplasm and preventing further entry of water. The relationship between the solute, water and pressure potentials can be expressed by the equation:
Ψw = Ψs + Ψp
(pressure potential is a positive number, solute and water potentials are negative).
Plant cells become turgid if immersed into solutions with a higher water potential than the cell contents; water enters by osmosis and the cytoplasm and vacuole expand. When it is not physically possible for any more water to enter the cell, the water potential of the cell is 0. This means the solute potential and pressure potentials cancel each other out.
When plant cells are immersed in solutions that have a water potential that is lower than the cells, water leaves the cells by osmosis, from a higher water potential inside the cell to a lower water potential in the solution. The vacuole and cytoplasm shrink because of the loss of water and the cytoplasm pulls away from the cell wall. Down a microscope there is a visible gap between the cell membrane and cell wall.
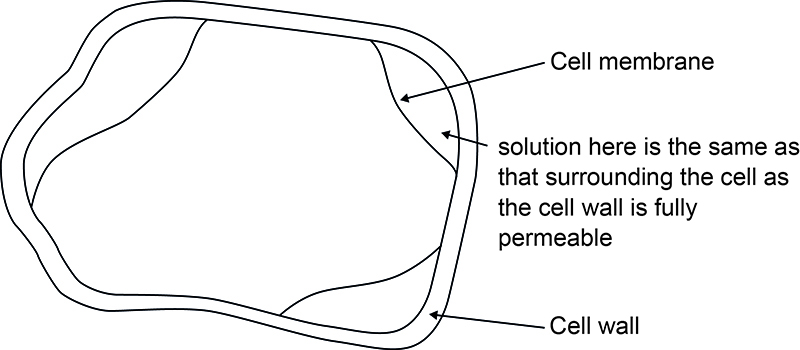
This phenomenon is known as plasmolysis and the cells are termed plasmolysed. As the cell wall is fully permeable, the solution diffuses through it and therefore the water potentials on both sides of the cell wall are the same. In a tissue, each cell has its own water potential. This means the cells plasmolyse at different sucrose concentrations. The point where 50% of the cells are plasmolysed is taken to be the point where the water potentials of solution and tissue are equal. This point is called incipient plasmolysis. When cells are plasmolysed the pressure potential is 0 kPa; the water potential and solute potential of the cells are equal.