Pressure in Gases (collapsing can practical)
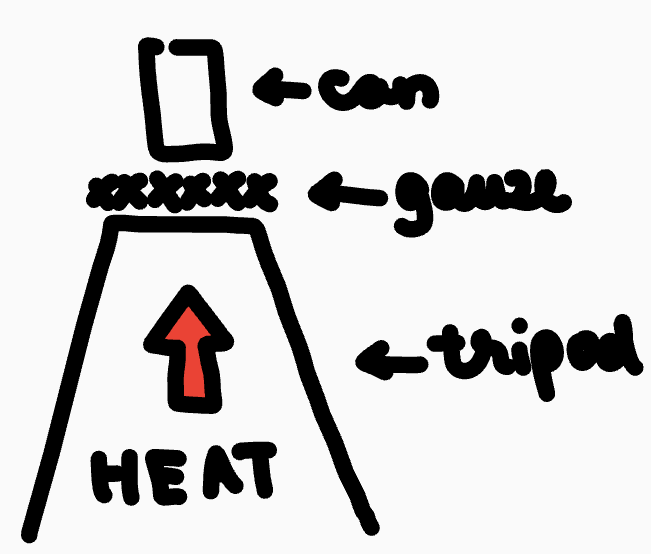
Before you start heating
Pressure is equal inside and outside the can
Water inside starts to heat up
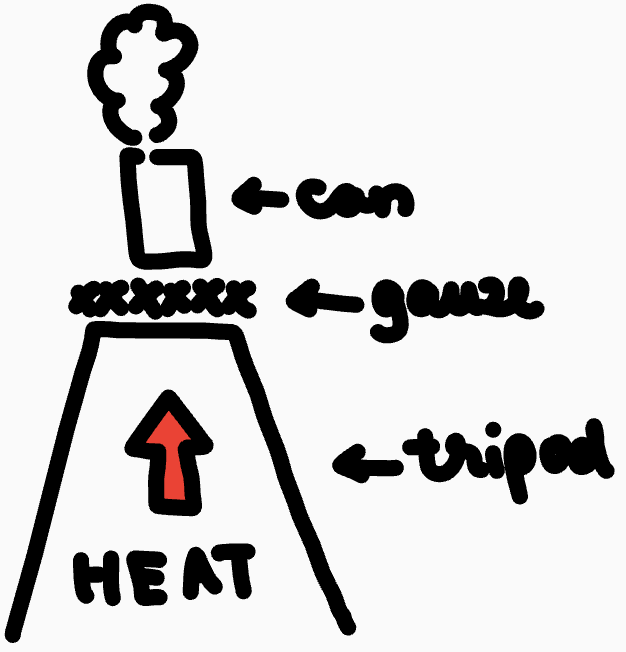
Steam comes out
Water becomes steam and starts to evaporate
Pressure stays the same as the steam is able to leave the can
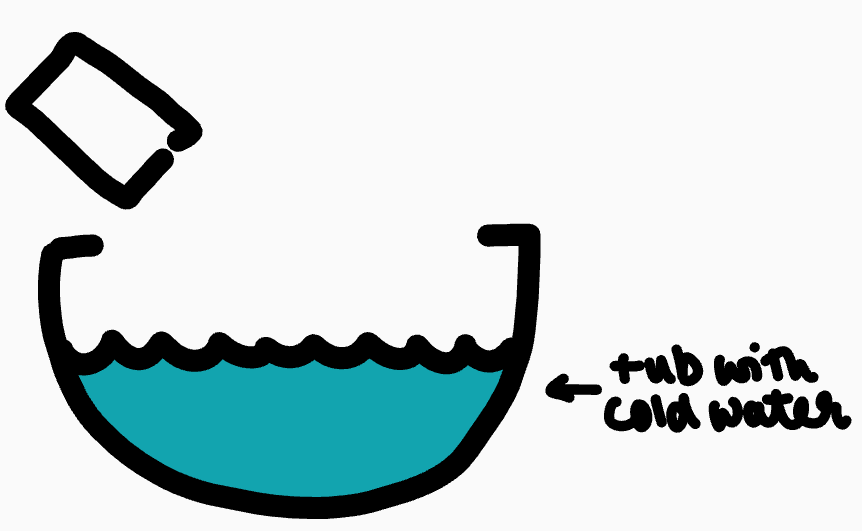
The can collapses
Steam cools suddenly and cannot escape (condenses into water)
Pressure decreases suddenly (atmospheric pressure is higher) and so the can crushes