Chapter 3 Earth's Minerals
Chapter 3 Outline
3.1 Matter Matters
3.2 Minerals and Mineral Groups
3.3 Mineral Identification
3.4 Mineral Formation
3.5 Mining and Mineral Use
3.1 Matter Matters
Lesson Objectives
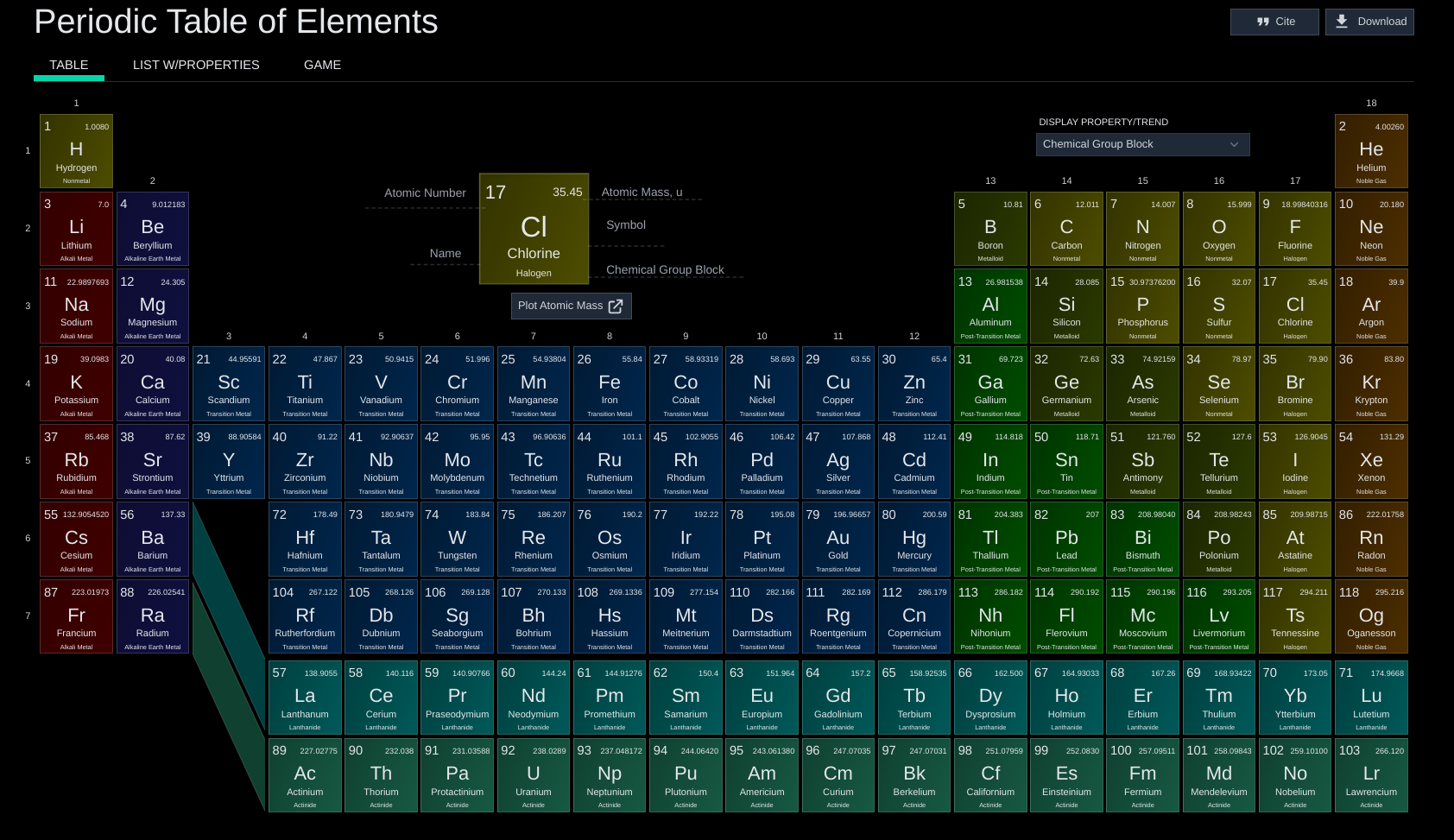
Review basic chemistry concepts including the structure of atoms, the distinction between elements and compounds, and the role of ions and molecules in chemical interactions.
Understand the types of chemical bonding (ionic, covalent, and hydrogen bonds) and how these bonds result in the formation of molecules and compounds.
Vocabulary
Atom: Basic unit of matter, consisting of a nucleus surrounded by electrons.
Atomic mass: Average mass of an atom, reflecting the number of protons and neutrons in the nucleus.
Chemical bond: Force that holds atoms together in molecules, influencing properties and reactivity.
Covalent bond: Bonds formed by the sharing of electrons between atoms, leading to the formation of molecules such as water (H2O).
Electron: Negatively charged particle in an atom that orbits the nucleus.
Element: Pure substance that cannot be broken down into simpler substances by chemical means; represented on the periodic table.
Hydrogen bond: A weak bond between polar molecules, significant in biological molecules like DNA.
Ion: A charged atom that has gained or lost electrons, resulting in a positive (cation) or negative (anion) charge.
Ionic bond: Bond formed by the transfer of electrons from one atom to another, exemplified by table salt (NaCl).
Isotope: Variants of the same element that have the same number of protons but different mass numbers due to differing numbers of neutrons.
Molecular mass: The sum of the atomic masses of all atoms in a given molecule, crucial for stoichiometric calculations in chemical reactions.
Molecule: Smallest unit of a chemical compound, formed by the chemical bonding of two or more atoms.
Neutron: Uncharged particle within the nucleus of an atom, contributing to its mass.
Nucleus: Center of an atom, composed of protons and neutrons.
Polar molecule: A molecule with an uneven distribution of charge, leading to distinct positive and negative ends (e.g., water).
Proton: Positively charged particle in an atom, defining the identity of the element.
3.2 Minerals and Mineral Groups
Lesson Objectives
Describe the distinctive characteristics that all minerals share, including their physical and chemical properties.
Identify and classify minerals into groups based on their common properties and chemical compositions.
Vocabulary
Chemical compound: A substance formed when two or more chemical elements are chemically bonded together.
Crystal: A solid material whose constituents are arranged in a highly ordered structure, forming a lattice that extends in all directions.
Inorganic: Substances not derived from living organisms; typically, they do not contain carbon-hydrogen bonds.
Mineral: Naturally occurring inorganic solid with a definite chemical composition and a crystalline structure.
Silicates: A group of minerals composed mostly of silicon and oxygen; they form the largest class of minerals found in Earth's crust.
Characteristics of Minerals
Minerals are categorized based on their chemical composition and structure; they can be classified through physical observation and chemical testing.
They are inorganic crystalline solids formed through natural geological processes, where each mineral is defined by a specific chemical formula (e.g. Quartz is composed of Silicon Dioxide, SiO₂).
Over 4,000 distinct minerals have been identified in Earth's crust, each with unique characteristics.
Crystalline Solid
A solid in which atoms are arranged in a highly ordered repeating pattern, contributing to the mineral's definite geometrical shape.
Inorganic Substances
Inorganic: Refers to minerals and substances that do not possess the characteristics of living organisms. Organic matter typically consists of carbon-based compositions, while substances like coal are classified as sedimentary rocks, not minerals.
Chemical Composition
Approximately 98.5% of Earth’s crust is composed of eight elements: oxygen, silicon, aluminum, iron, calcium, sodium, potassium, and magnesium, from which most minerals are formed.
Physical Properties of Minerals
Color: May vary due to impurities; not a reliable identifier on its own.
Streak: Color of the mineral in powdered form, more consistent than raw color.
Luster: Indicates how light reflects off the surface of the mineral (e.g., shiny vs. dull).
Specific Gravity: Density comparison to water, providing insight into the mineral's composition.
Cleavage: Tendency of a mineral to break along flat, even planes.
Fracture: The way a mineral breaks irregularly; different from cleavage.
Hardness: Resistance to scratching, measured using Mohs scale, ranking minerals from 1 (talc) to 10 (diamond).
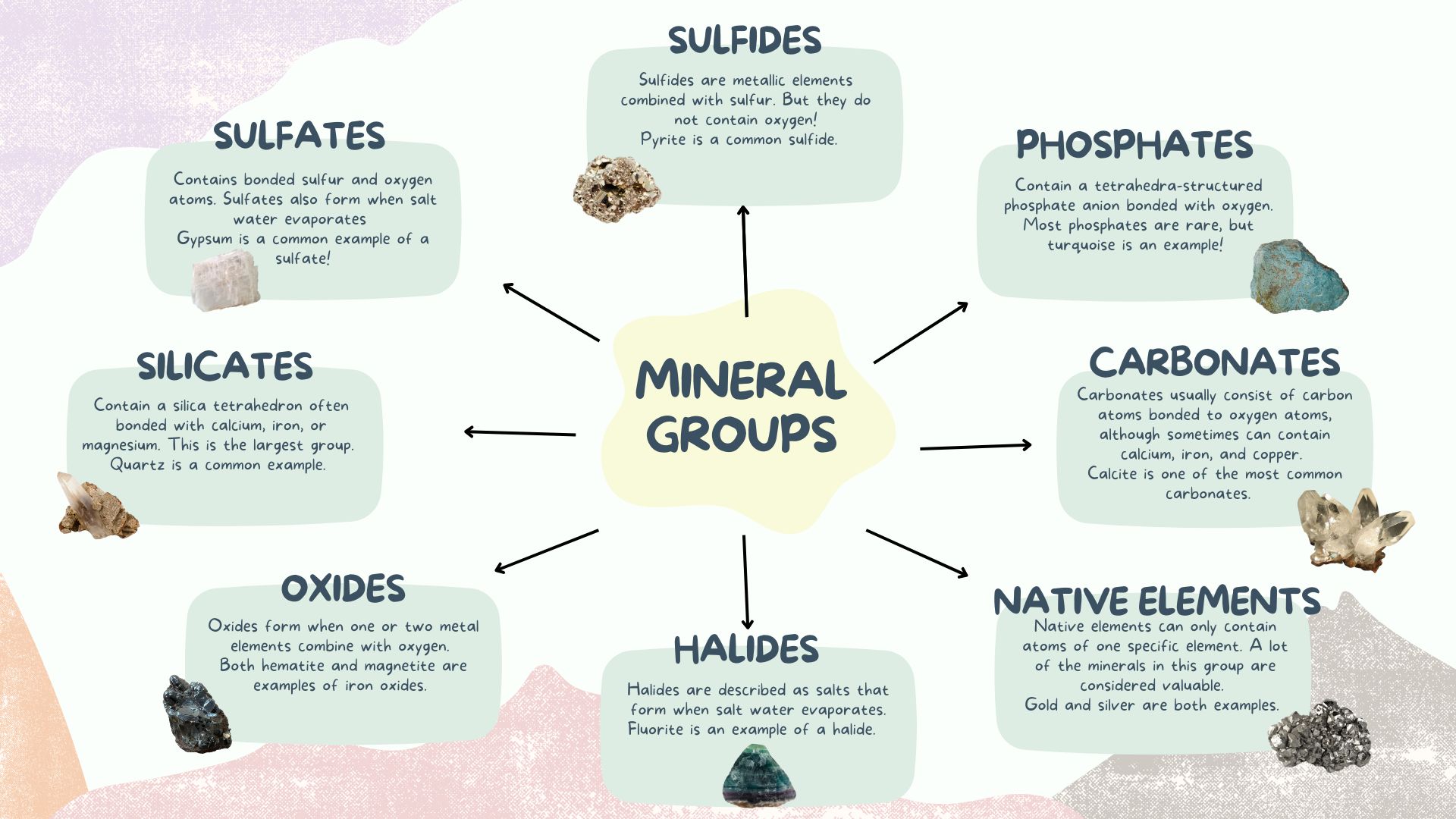
Mineral Groups
Silicate Minerals: Make up 90% of Earth’s crust; examples include feldspar and quartz, fundamental to igneous rocks.
Native Elements: Rare minerals composed of a single element, such as gold (Au) and silver (Ag).
Carbonates: Contain carbon and oxygen; a common example is calcite (CaCO₃).
Halides: Formed through the evaporation of saltwater, such as halite (table salt).
Oxides: Composed of metals bonded with oxygen, such as hematite (Fe₂O₃).
Phosphates: Minerals containing phosphorus combined with oxygen; important in fertilizers.
Sulfates: Contain sulfur bonded to oxygen, e.g., gypsum (CaSO₄·2H₂O).
Sulfides: Composed of metals combined with sulfur; examples include pyrite (FeS₂).
Mineral Group | Composition of Group | Examples |
Silicate Minerals | Composed mainly of silicon and oxygen; forms the largest class of minerals. | Feldspar, Quartz |
Native Elements | Rare minerals made of a single element. | Gold (Au), Silver (Ag) |
Carbonates | Contain carbon and oxygen. | Calcite (CaCO₃) |
Halides | Form through the evaporation of saltwater. | Halite (table salt) |
Oxides | Composed of metals bonded with oxygen. | Hematite (Fe₂O₃) |
Phosphates | Contain phosphorus combined with oxygen. | Apatite (Ca₅(PO₄)₃(F,Cl,OH)) |
Sulfates | Contain sulfur bonded to oxygen. | Gypsum (CaSO₄·2H₂O) |
Sulfides | Composed of metals combined with sulfur. | Pyrite (FeS₂) |
3.3 Mineral Identification
Lesson Objectives
Explain the methodologies utilized in identifying minerals, including assessment of various physical characteristics.
Describe important characteristics like color, luster, streak, specific gravity, hardness, and fracture in detail.
Vocabulary
Cleavage: The tendency of a mineral to break along specific planes of weakness in its structure.
Density: Measures the mass of a mineral compared to its volume.
Fracture: Characteristic of breaking along uneven surfaces.
Hardness: Indicates how resistant a mineral is to scratching.
Luster: Describes how a mineral surface reflects light.
Mineralogist: A scientist who studies minerals, their distribution, identification, and classification.
Streak: The color of the powdered form of the mineral, useful for identification.
Identifying Minerals
Minerals are identified through a systematic evaluation of their physical and chemical properties; this includes observing the mineral's color, streak, and luster, but ultimately relies on more reliable characteristics such as hardness, cleavage, and specific gravity.
Mohs Hardness Scale

A scale of 1 to 10 that indicates the softness or hardness of a mineral where talc is the softest (1) and diamond is the hardest (10).
3.4 Mineral Formation
Lesson Objectives
Explain how various geological processes such as the cooling of magma or evaporation of solutions contribute to mineral formation.
Vocabulary
Lava: Molten rock that erupts onto the Earth's surface.
Magma: Molten rock located beneath the Earth's surface.
Rock vein: A distinct mineral-rich area within a larger rock formation.
Methods of Mineral Formation
Minerals can form through various geological processes including:
Cooling magma: Slowly cooling magma underground can create large crystals (e.g., granite).
Evaporating solutions: As water evaporates, the dissolved minerals precipitate, forming sedimentary deposits.
Hot underground waters: These can transport minerals, which precipitate out when the water cools or loses pressure, leading to formations like geodes where large crystals grow in cavities.
3.5 Mining and Mineral Use
Lesson Objectives
Explain the various methods by which minerals are extracted from the earth.
Describe the processes involved in extracting metals from ores and the various uses of gemstones.
Identify major useful minerals in industry and everyday life.
Vocabulary
Gemstone: A precious or semi-precious stone that has been cut and polished for use in jewelry.
Ore: A type of rock that contains sufficient minerals with economically important elements, typically metals, that can be extracted profitably.
Ore deposit: A locality where a particular mineral resource is concentrated and can be mined.
Placer: Deposits of minerals that have been eroded from their parent rock, concentrated by water action.
Reclamation: The process of restoring land disturbed by mining activities to a natural or economically usable state.
Minerals and Mining
Ore: Rock containing useful elements; crucial for sourcing metals and other valuable materials.
Mining methods: Encompass surface mining, where minerals are extracted from the near-surface, and underground mining, which reaches deeper deposits.
Mining Methods
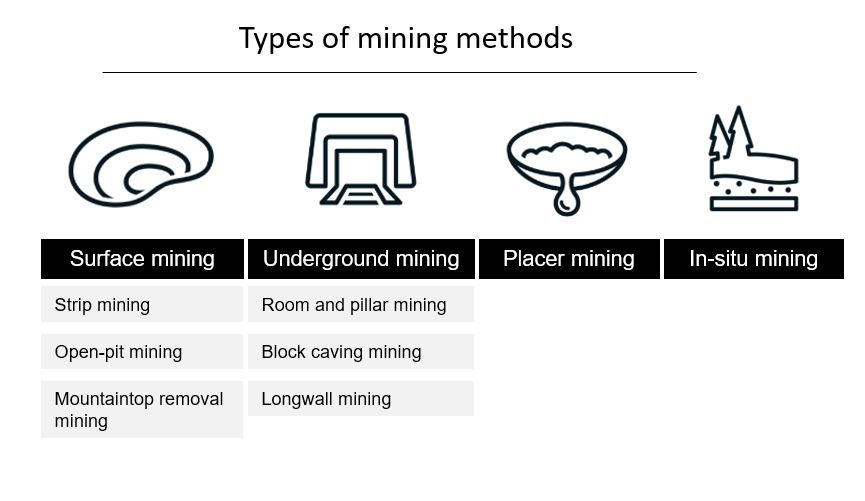
Surface Mining: Techniques include open-pit mining and strip mining, involving blasting and removal of vast soil layers.
Underground Mining: Involves digging tunnels that require ventilation for safety and lighting; presents various risks and challenges.
Extraction techniques: Include processes like heap leaching, flotation, and smelting, each tailored for different mineral types and desired end products.
Environmental Impact of Mining
Mining activities can lead to habitat destruction, soil erosion, and water pollution, significantly affecting local ecosystems.
Contamination of water sources often occurs from the runoff of toxic chemicals used in extraction processes, posing risks to both wildlife and human populations.
Mitigation strategies, such as reforestation and rehabilitation of mined areas, are essential to minimize the long-term ecological footprint of mining.
Mining can lead to significant environmental degradation, impacting both terrestrial and aquatic ecosystems. Key issues include:
Deforestation: Large areas of forest are often cleared to make way for mining operations, which disrupts the local vegetation and wildlife. This not only contributes to loss of biodiversity but also affects carbon storage, exacerbating climate change.
Habitat Loss: The extraction of minerals can lead to the destruction of habitats for many species, leading to endangerment or extinction. As natural habitats are removed, wildlife is displaced, which can disrupt local ecosystems.
Soil Erosion: Mining activities disturb the soil and remove protective vegetation, making the soil prone to erosion. This loss of topsoil decreases the land's agricultural viability and can lead to sediment runoff into waterways.
Contamination of Water Systems: Mining operations can result in toxic substances, including heavy metals and chemical runoff, entering rivers and lakes. This pollution can significantly affect aquatic life and reduce water quality for local communities.
Because of these severe environmental impacts, reclamation efforts are mandated by legislation. Reclamation includes:
Restoring Native Vegetation: This involves planting native species that are well-suited to the local environment to help restore the ecosystem.
Preventing Further Erosion: Techniques such as contour plowing and building terraces may be applied to stabilize the soil and prevent future erosion.
Water Quality Management: Efforts must be made to monitor and improve water quality post-mining, which may include managing drainage and treating polluted water before it re-enters natural systems.
Overall, effective reclamation requires careful planning and implementation to ensure that the landscape is not only restored but that it can support biodiversity and ecosystem services after mining has ceased.