Transamination & Glycogenesis
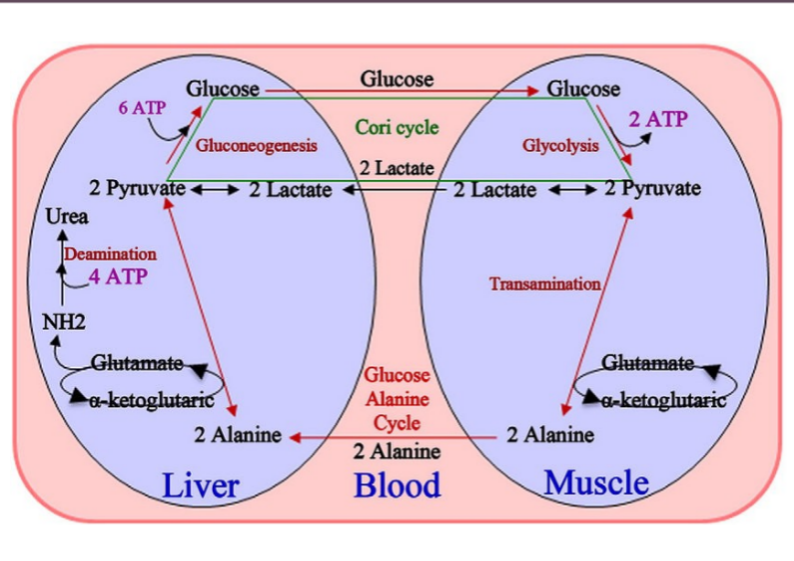
alanine aminotransferase/glutamine synthetase: liver metabolism test
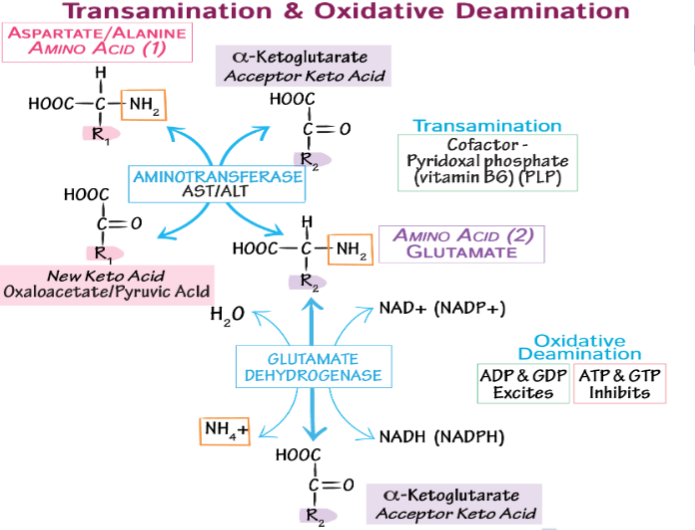
glutaminase in kidney: control pH
kidney failure: dialysis for 3-5 years
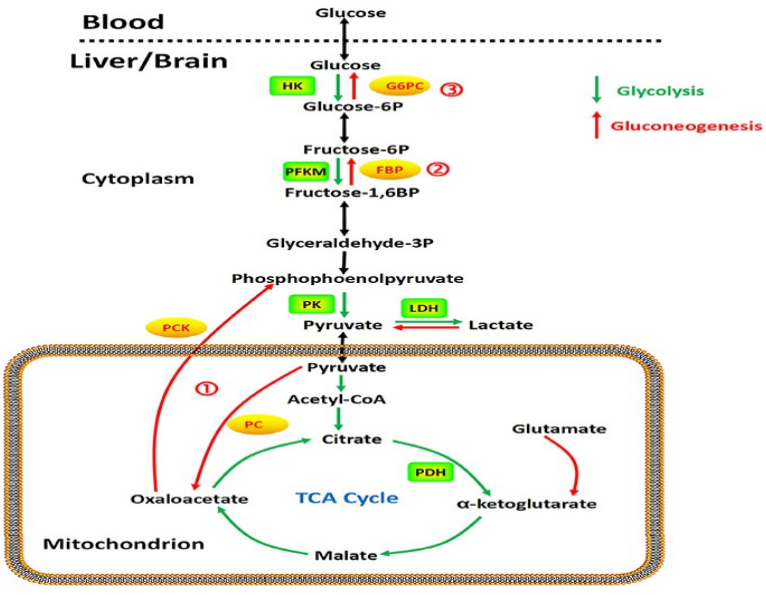
Central Metabolism Pathways of Glucose
synthesis of structural polymers: extracellular matrix and cell wall polysaccharides
storage: glycogen, starch, sucrose
oxidation via glycolysis: pyruvate
oxidation via pentose phosphate pathway: ribose 5-phosphate
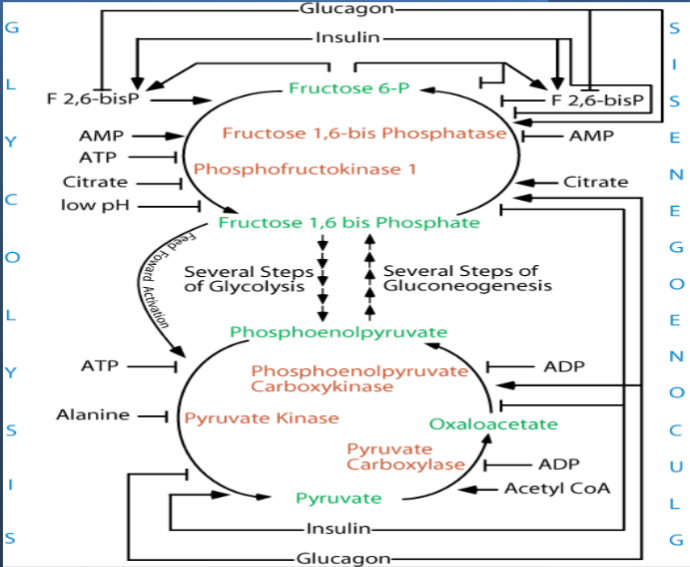
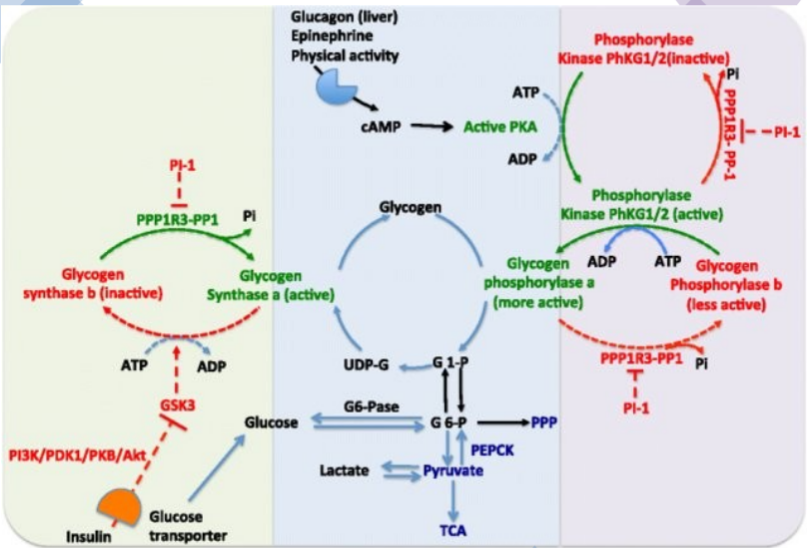
F-2,6-BP promotes PFK1
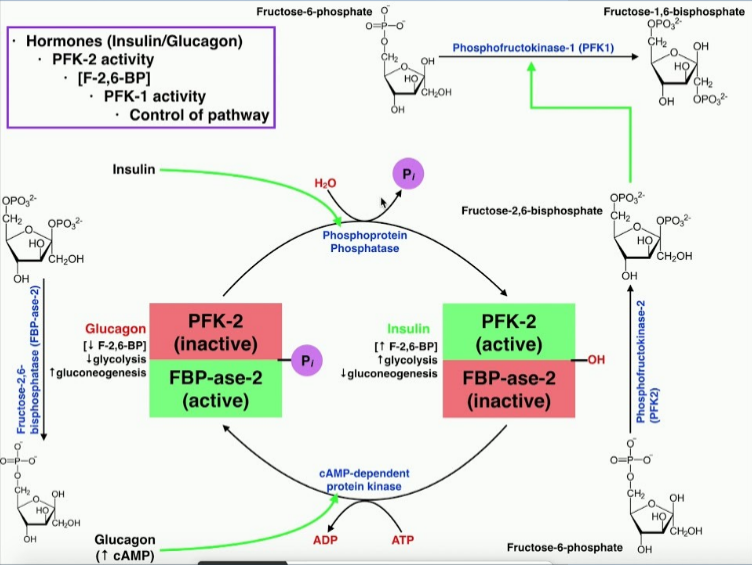
FBPase2 - phosphatase of F-2,6-BP
PFK2 - kinase of F-6-P
Liver cell:
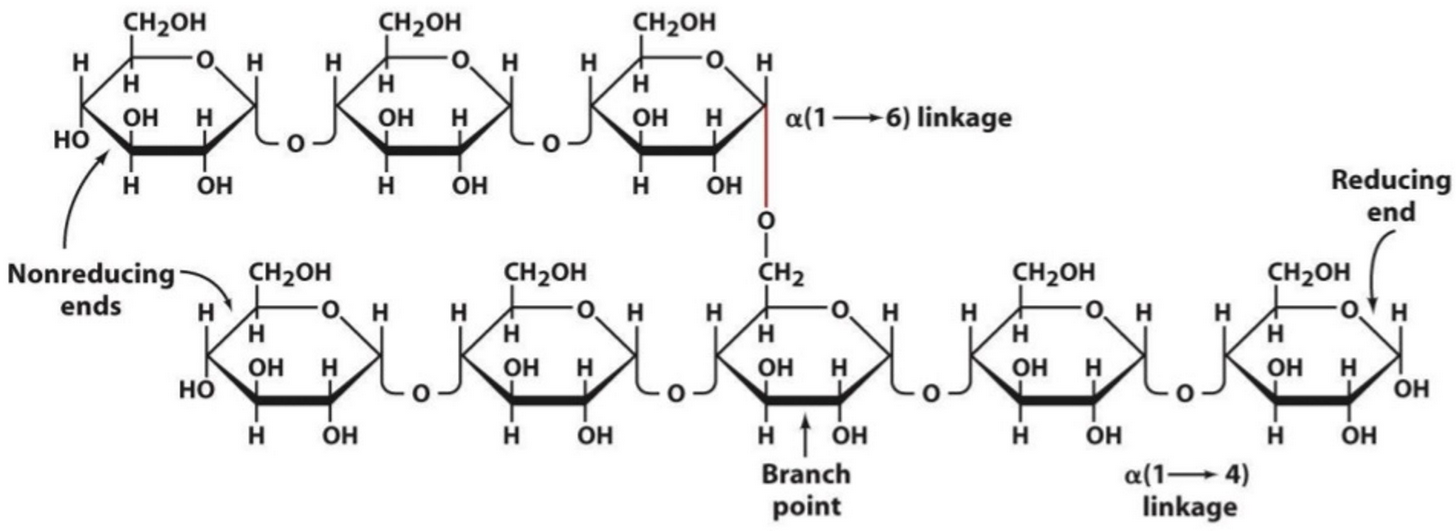
black spots: glucose residues from glycogen “rosettes”
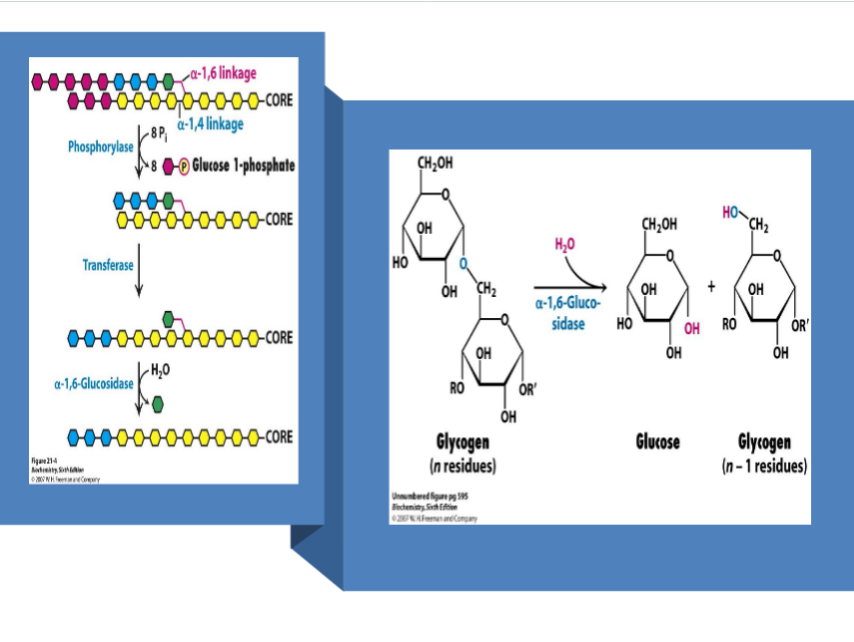
highly branched
largest organ inside the body
20-25% of liver weight
one of the most aerobic tissues: 2K to 2.5K mitochondria (compare to myocardial (same range) and brain (3K))
muscle cell:
1% weight glycogen
3D structure: alpha helix
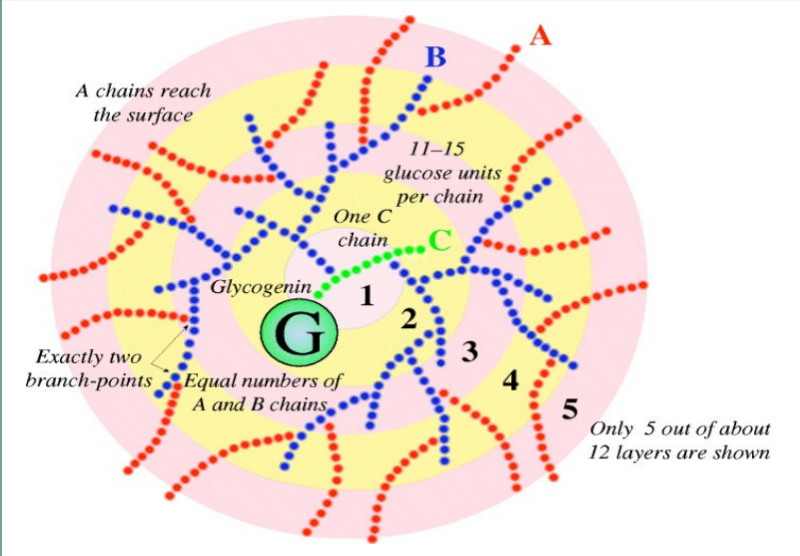
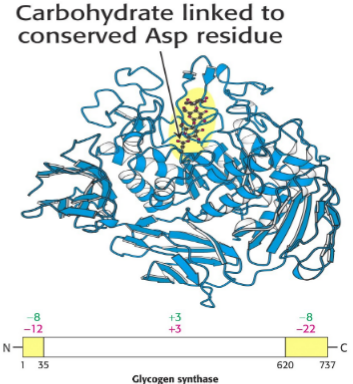
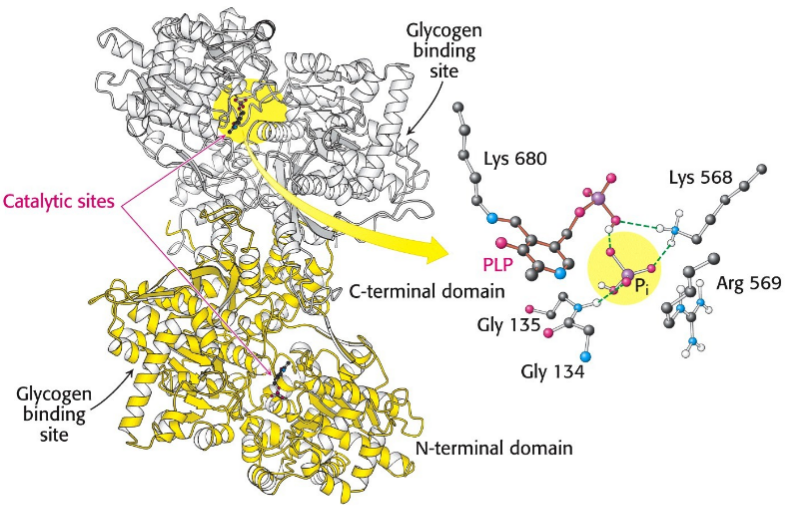
Glycogenin -”primase” of glycogen; initiates synthesis of the main branch; dimer
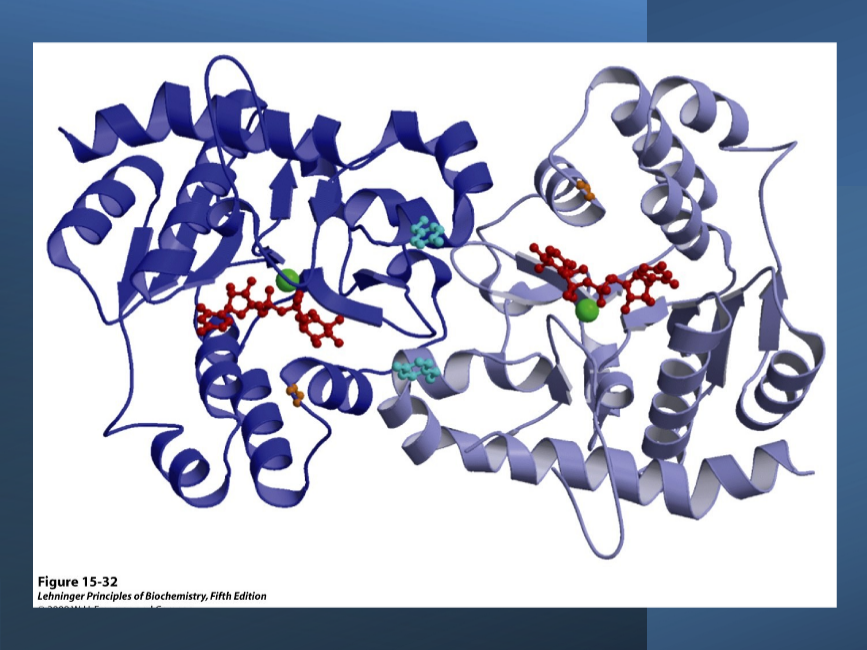
Glycogen phosphorylase - dimer
Thermodynamics
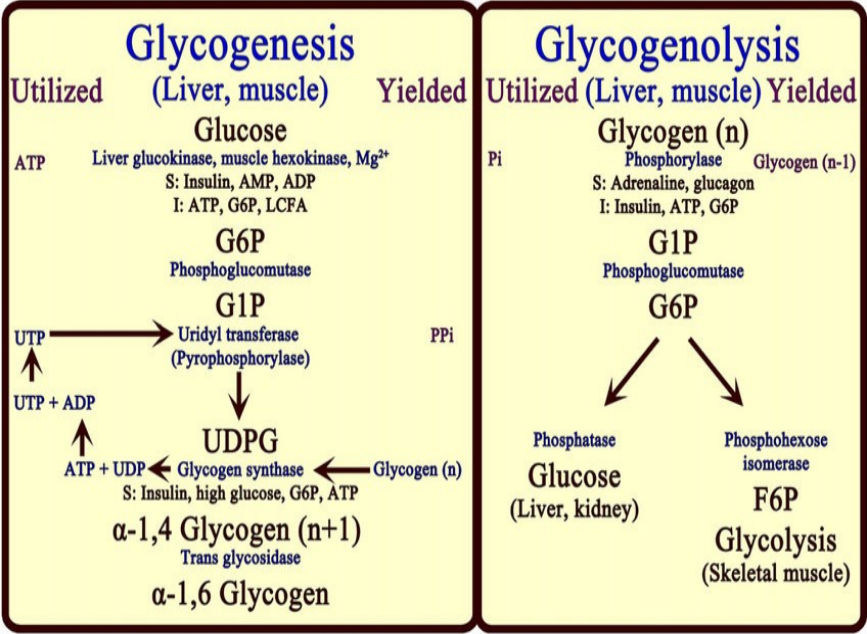
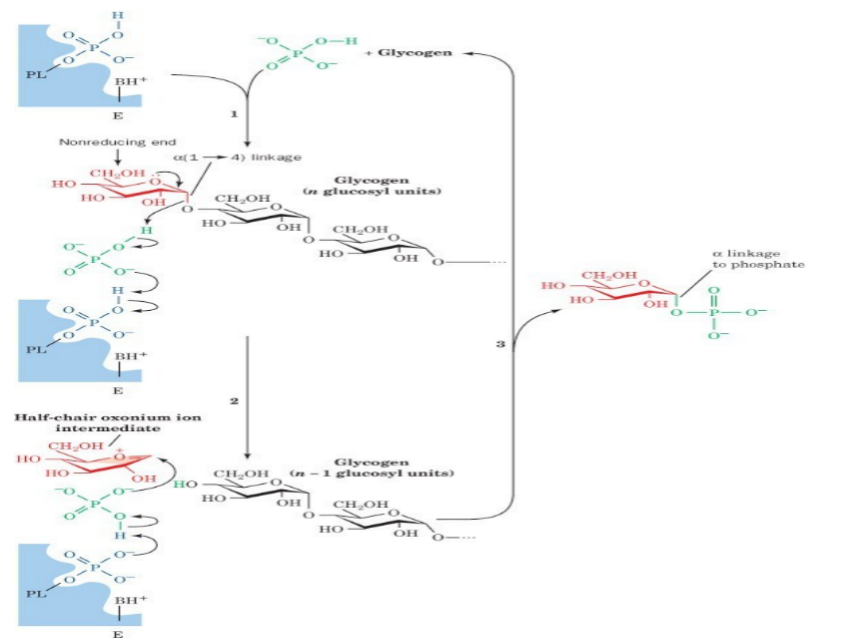
Under physiological conditions, phosphorolysis of glycogen is exergonic, -5 to -8 KJ/mol
The formation of G1P under physiological condition is unfavorable, requiring free energy input.
Consequently, breakdown and synthesis must be separate pathways.
This allows reciprocal controls and independent regulation of each pathway.
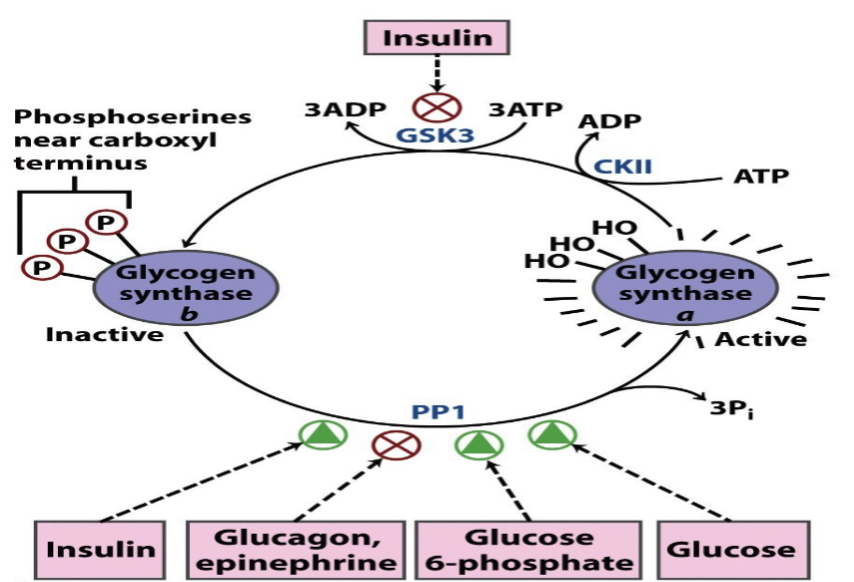
Since the synthesis of glycogen from G1P is thermodynamically unfavorable, it requires a supply of energy.
Glycogen synthesis requires an additional exergonic step, formation of UDP-glucose.
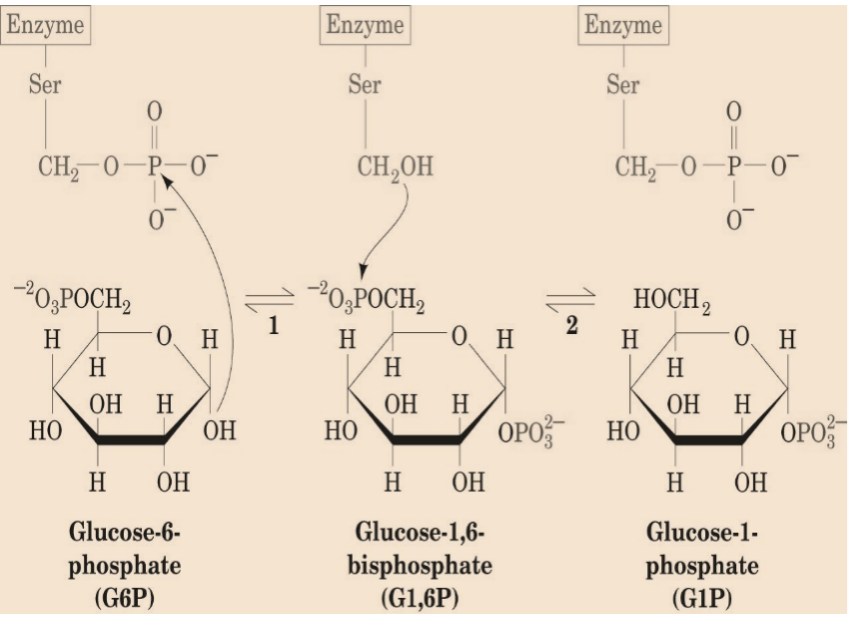
Three enzymes catalyse the formation of glycogen; UDP-glucose pyrophosphorylase, glycogen synthase & glycogen branching enzyme.
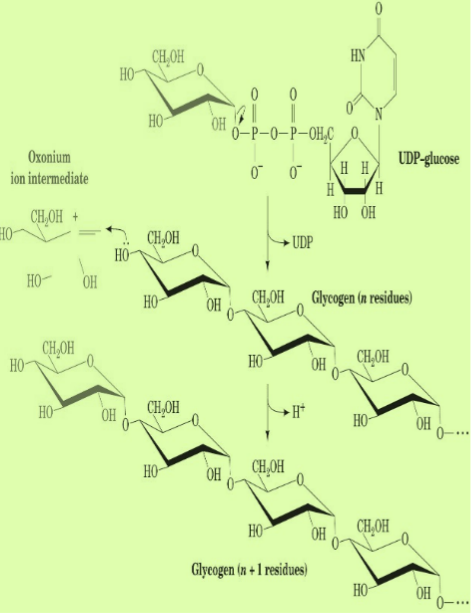
G1P +UTP → UDPG + PPi ≈0 kJ/mol
H2O + PPi → 2Pi -33.5 kJ/mol
Cycle
GPCR
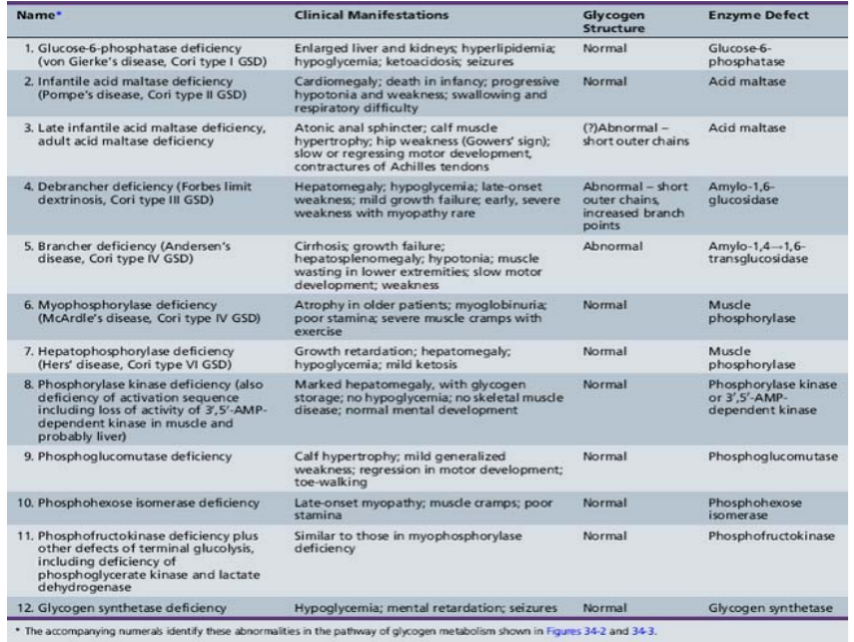
diabetic blood sugar 8mm - rest removed in urine “maple syrup urine”
constant amino acids → amine → urea cycle
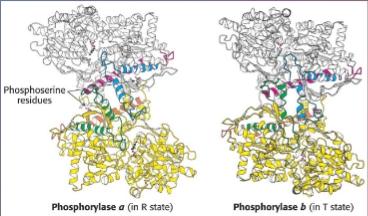
in the T state, the enzyme active site is buried, hence the low affinity for the substrate
in the R state, the enzyme has an accessible catalytic site and high affinity phosphate binding site.
AMP promotes T(inactive)→R(active) conformational shift.
ATP binds to the allosteric effector site in the T conformation and inhibits the T(inactive)→R(active)
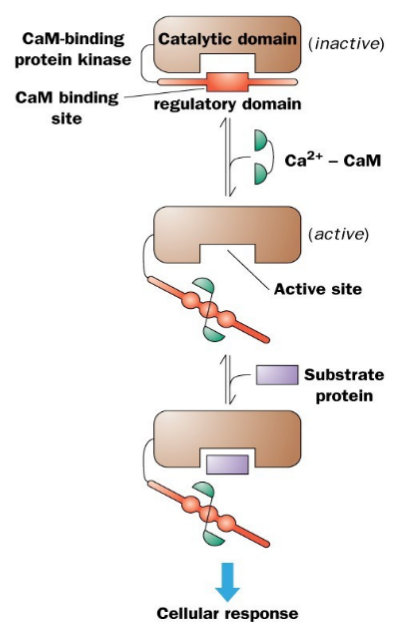
Auto-inhibited kinases have an N or C terminal pseudo-substrate sequence. It make the active site inaccessible to substrate. The CaM subunit binds near the auto- inhibitory sequence and the activation of CaM by binding Ca+2, binds to the auto- inhibitory sequence and it opens up the access to the active site. It can now bind to proteins and phosphorylates them. The activity is dependent on the Ca+2 availability.
phosphorolypase C/I3P receptor/opens calcium 10^-11 to 10^-9
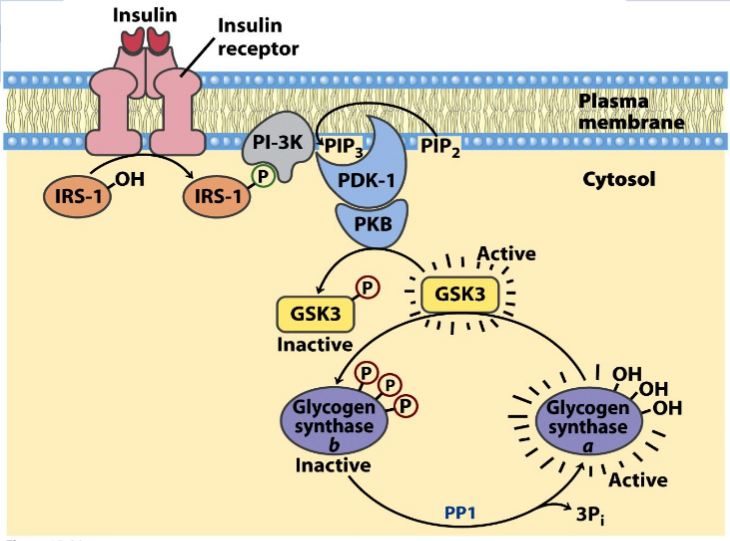
Stress
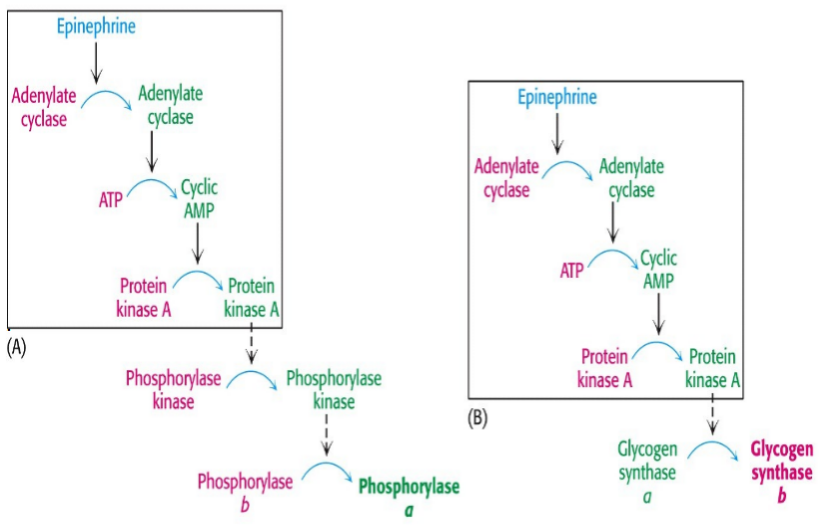
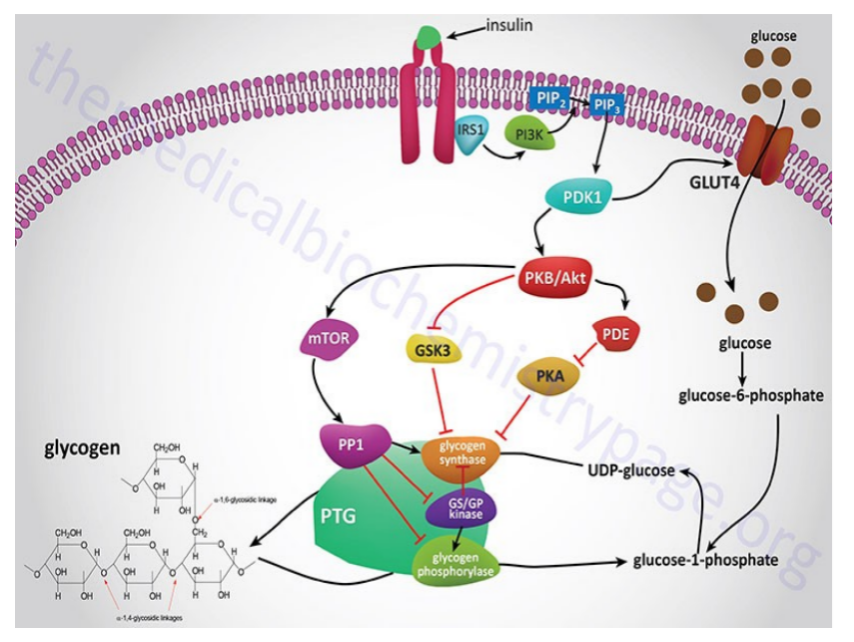
Stimulation of both the alpha- and beta-adrenoreceptors by epinephrine. Epi activates phospholipase C to hydrolyze PIP2 to IP3 and DAG. Both of these lead to rapid increases in [cAMP] & Ca+2. The release of Ca reinforces the effects of cAMP. PhK which activates glycogen phosphorylase and inactivates glycogen synthase, isonly fully active when it is phosphorylated and in the presence of Ca+2. Glycogen synthase is phosphorylated and inactivated by several other enzymes. In the presence of Ca+2 & DAG PKC is activated and it will also phosphorylate the synthase.
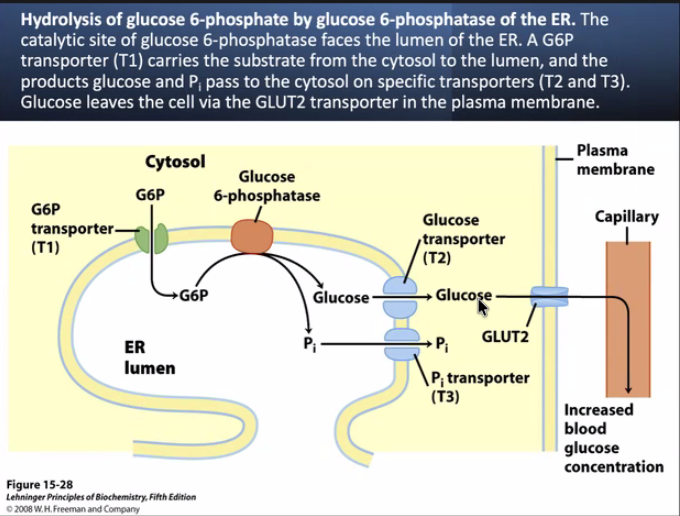
Stimulation of the a adrenoreceptor by epi activates phosphlipase C to hydrolyse PIP2 to IP3 & DAG. The participation of two second message systems: the cAMP mediated glycogenolysis and inhibition of glycogen synthesis triggered by glucagon and the adrenoreceptor activation; & IP3, DAG and Ca+2 mediated stimulation of glycogenolysis as well as inhibition of glycogen synthesis. DAG & Ca+2 activate PKC that PO4 glycogen synthase causing inactivation. G6Pase is an ER transmembr protein. T1 G6P translocase(T1) bring in the G6P, G6Pase metabolizes it to glucose + Pi and T2 & T3 transport glucose & Pi repectively to cyotosol. GLUT2 transports glucose into blood.
mTOR - target of Rifampicin - inhibits it