Carbon and Its Compounds
Introduction to Carbon
Carbon is a non-metal with the symbol "C" and an electronic configuration of 2, 4.
It is one of the most versatile elements, forming the backbone of all living organisms.
Its compounds are widely used in daily life, such as in clothes, medicines, books, food, fertilizers, and fuels.
Abundance of Carbon
Carbon is present in limited quantities:
Earth's crust: 0.02% (as carbonates, hydrogen carbonates, coal, and petroleum).
Atmosphere: 0.03% as carbon dioxide.
Despite this, carbon is significant due to its ability to form diverse compounds like hydrocarbons and other compounds containing elements such as oxygen, nitrogen, sulfur, halogens, and phosphorus.
Daily Use of Carbon Compounds
Most everyday items (soaps, detergents, books, furniture, and food) are carbon-based, except for water and utensils made of glass, metals, or ceramics.
Bonding in Carbon Compounds
Covalent Bonding:
Carbon forms covalent bonds by sharing electrons to achieve stability (octet rule).
Tetravalency allows carbon to form bonds with four atoms of the same or different elements.
Types of Covalent Bonds:
Single Bond: Sharing one pair of electrons (e.g., H-H, H-Cl).
Double Bond: Sharing two pairs of electrons (e.g., O=O, CO₂).
Triple Bond: Sharing three pairs of electrons (e.g., N≡N, C₂H₂).
Why Carbon Forms Covalent Bonds:
Gaining four electrons to form C⁴⁻ is difficult due to repulsion among extra electrons.
Losing four electrons to form C⁴⁺ requires excessive energy.
Carbon shares electrons instead, forming stable covalent bonds.
Gaining or losing four electrons is impractical due to energy requirements, so carbon shares electrons to achieve stability.
Allotropes of Carbon
Allotropy: It is the phenomenon of existence of an element in two or more forms which have different physical properties but identical chemical properties. The different forms are called allotropes.
Carbon exists in three main crystalline forms:
Diamond:
Each carbon atom is bonded tetrahedrally to four others, forming a rigid 3D structure.
Properties: Transparent, colorless, hard, high refractive index (2.45).
Use: Cutting tools, jewelry.
Preparation of synthetic diamond:
Diamond can be synthesized by subjecting pure carbon to very high pressure and temperature. These synthetic diamonds are smaller than natural diamonds.
Graphite:
Graphite is also known as black lead as it marks paper black.
In a crystal of graphite the carbon atoms are arranged in a hexagonal pattern in parallel planes. In a layer of graphite each carbon atom is strongly bonded to three other carbon atoms by covalent bonds. Thus, one valence electron of each carbon atom is free in every layer of graphite crystal.
Carbon atoms are arranged in hexagonal layers with weak forces between layers, allowing them to slide over each other.
Properties: Opaque, slippery, conducts electricity (due to free electrons).
Use: Pencils, lubricants.
Fullerenes:
Spherical or tubular carbon structures.
The molecule containing sixty atoms of carbon has been named as Buckminster fullerene.
C₆₀ is the most stable and most studied form of fullerenes.
C₆₀ has a symmetrical structure with 20 hexagons and 12 pentagons.
Versatile Nature of Carbon
Catenation:
"The property of forming bonds with atoms of the same element is caled catenation".
Carbon can bond with itself to form long chains (straight, branched, or ringed).
No other element shows catenation to the same extent due to the strong carbon-carbon bond.
Silicon forms compounds with hydrogen which have chains of upto seven or eight atoms, but these compounds are very reactive. The carbon-carbon bond is very strong and hence stable.
Types of chains:
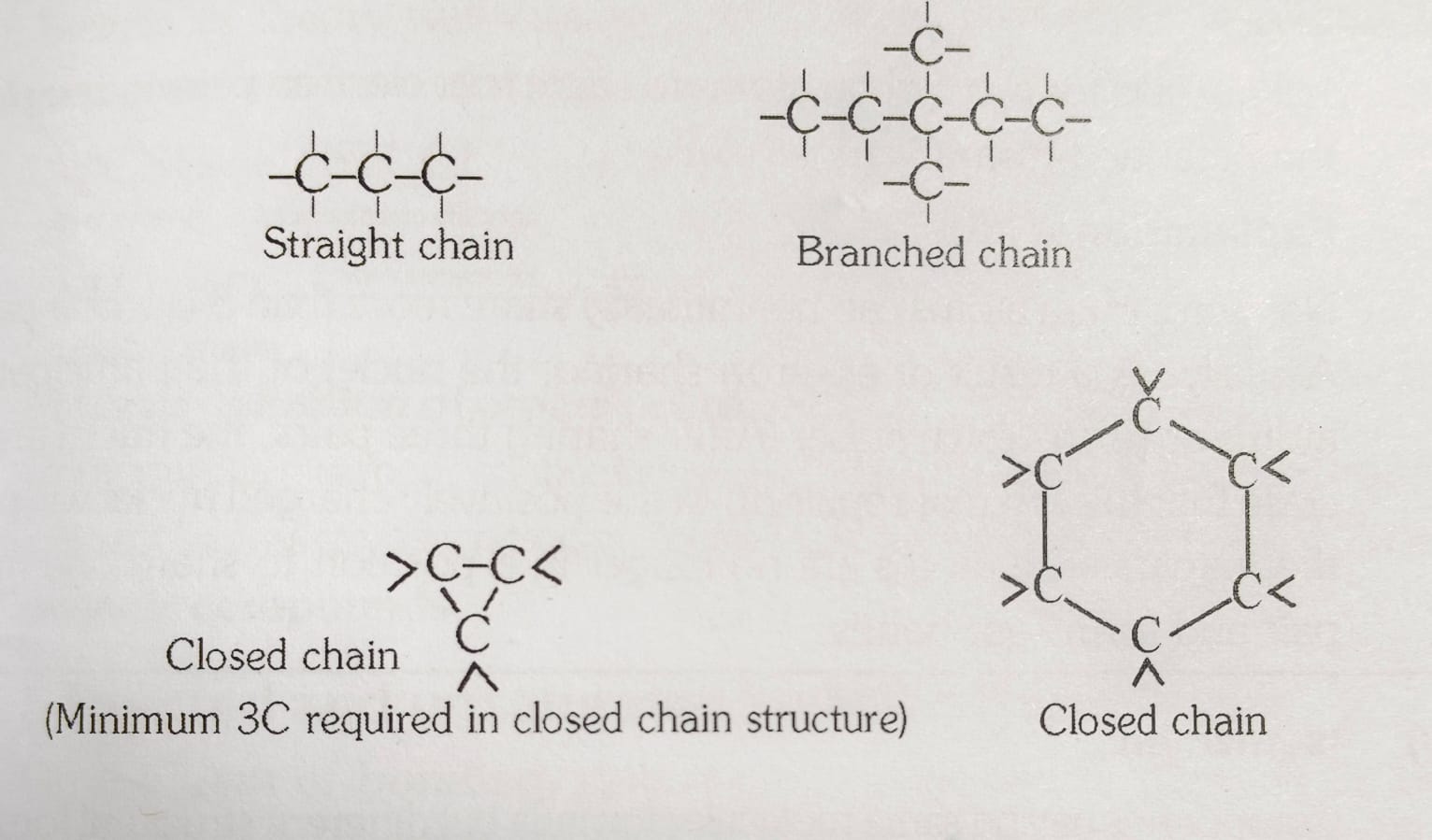
CONCEPT 2
Q.Why carbon shows maximum catenation among all elements?
-Causes of maximum catenation in carbon are:-
(1) Small size.
(ii) Unique electronic configuration (2, 4).
(iii) Great strength of carbon-carbon bonds (355 kJ/mol).
Tetravalency:
Carbon forms stable covalent bonds with various elements like hydrogen, oxygen, nitrogen, and sulfur.
Multiple Bonds:
Carbon easily forms double or triple bonds due to its small size and strong bonding ability.
CONCEPT 3
Q.Is it possible for two carbon atoms to share four electron pairs to complete the octet and form C₂ molecule?
-No, two carbon atoms can not mutually share more than 3 electron pairs. Actually, as a result of electron sharing, the nuclei of the participating atoms are also drawn closer. After sharing three pairs, the nuclei are so close that the force of repulsion in the positively charged nuclei will push them apart. The atoms are no longer in a position to share one more pair and form four bonds.
Isomerism:
Compounds having same molecular formula but different structural formulae are known as isomers and the phenomenon of existence of isomers is termed as isomerism.
Explames-
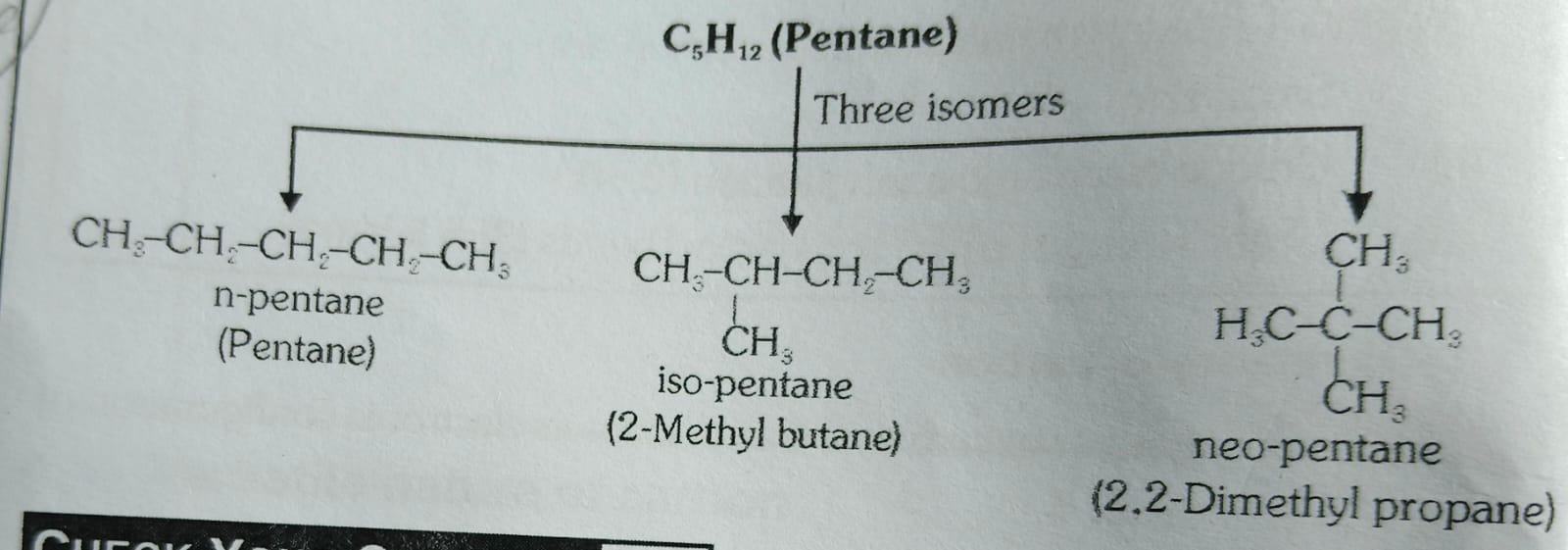
Organic Compounds
1. Versatility of Carbon
Due to its versatile nature, carbon forms a vast array of compounds. In the 18th century, compounds were classified into two categories:
Organic Compounds: Derived from living organisms, such as urea, sugar, oils, fats, and dyes.
Inorganic Compounds: Sourced from non-living materials like rocks and minerals, e.g., marble, common salt, alum, and copper sulfate.
2. Vital Force Theory
Proposed by Berzelius in 1815, this theory stated that organic compounds could only be produced by living organisms due to a "vital force."
Disproval by Wohler's Synthesis: In 1828, Friedrich Wohler synthesized urea in a laboratory from ammonium cyanate, disproving the vital force theory.
3. Modern Definition of Organic Compounds
Organic compounds are defined as carbon compounds containing hydrogen and other elements like oxygen, nitrogen, sulfur, halogens, and phosphorus.
4. Hydrocarbons
Carbon compounds are classified based on bonding into two main types:
Saturated Hydrocarbons (Alkanes): Contain only single carbon-carbon bonds.
General formula: CnH2n+2C_nH_{2n+2}CnH2n+2.
Examples: Methane (CH4CH_4CH4), Ethane (C2H6C_2H_6C2H6), Propane (C3H8C_3H_8C3H8).
Unsaturated Hydrocarbons: Contain double or triple bonds between carbon atoms.
Alkenes: Contain one or more double bonds.
General formula: CnH2nC_nH_{2n}CnH2n.
Examples: Ethene (C2H4C_2H_4C2H4), Propene (C3H6C_3H_6C3H6).
Alkynes: Contain one or more triple bonds.
General formula: CnH2n−2C_nH_{2n-2}CnH2n−2.
Examples: Ethyne (C2H2C_2H_2C2H2), Propyne (C3H4C_3H_4C3H4).
5. Structure-Based Classification
Organic compounds are categorized by their structural arrangement:
(a) Straight Chain Compounds
Consist of carbon atoms linked in a linear fashion.
Examples:
Alkanes: Methane (CH4CH_4CH4), Butane (C4H10C_4H_{10}C4H10).
Alkenes: Ethene (C2H4C_2H_4C2H4), Butene (C4H8C_4H_8C4H8).
Alkynes: Ethyne (C2H2C_2H_2C2H2), Butyne (C4H6C_4H_6C4H6).
(b) Branched Chain Compounds
Contain carbon atoms linked in branching patterns.
Examples:
Alkanes with more than three carbons: Iso-butane (C4H10).
Unsaturated compounds like butene (C4H8) can also have branched chains.
(c) Closed Chains or Ring Compounds (Cyclic Hydrocarbons)
Contain carbon atoms arranged in a closed loop or ring structure.
Types:
Alicyclic Hydrocarbons (Cycloalkanes): Saturated cyclic compounds, named with the prefix "cyclo."
Example: Cyclopropane (C3H6).
Aromatic Hydrocarbons: Contain at least one benzene ring, characterized by alternating double bonds.
Example: Benzene (C6H6).