L15: Diffusion in solid-liquid phase transformations

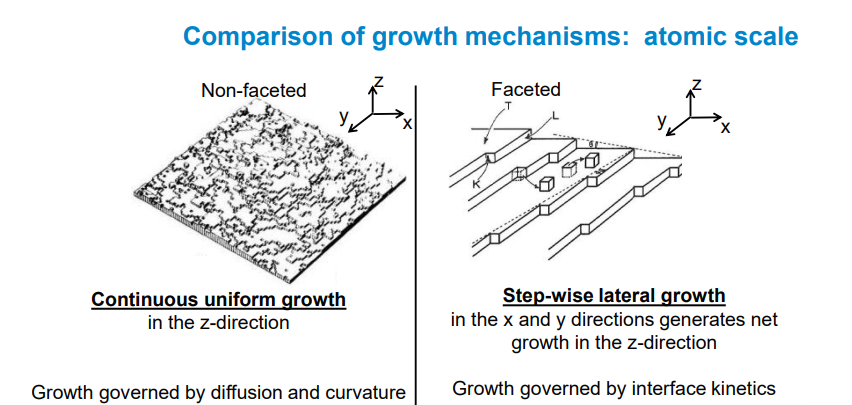
1. Heat and Solute Diffusion in Solidification
Heat Diffusion:
Latent Heat Release: During solidification, latent heat is released at the S-L interface. This heat must be removed to keep the interface moving.
Heat Flow Direction: Heat flows down the temperature gradient, either into the liquid or the solid, depending on which is colder.
Governing Equation: The temperature profile in the mould can be described by:

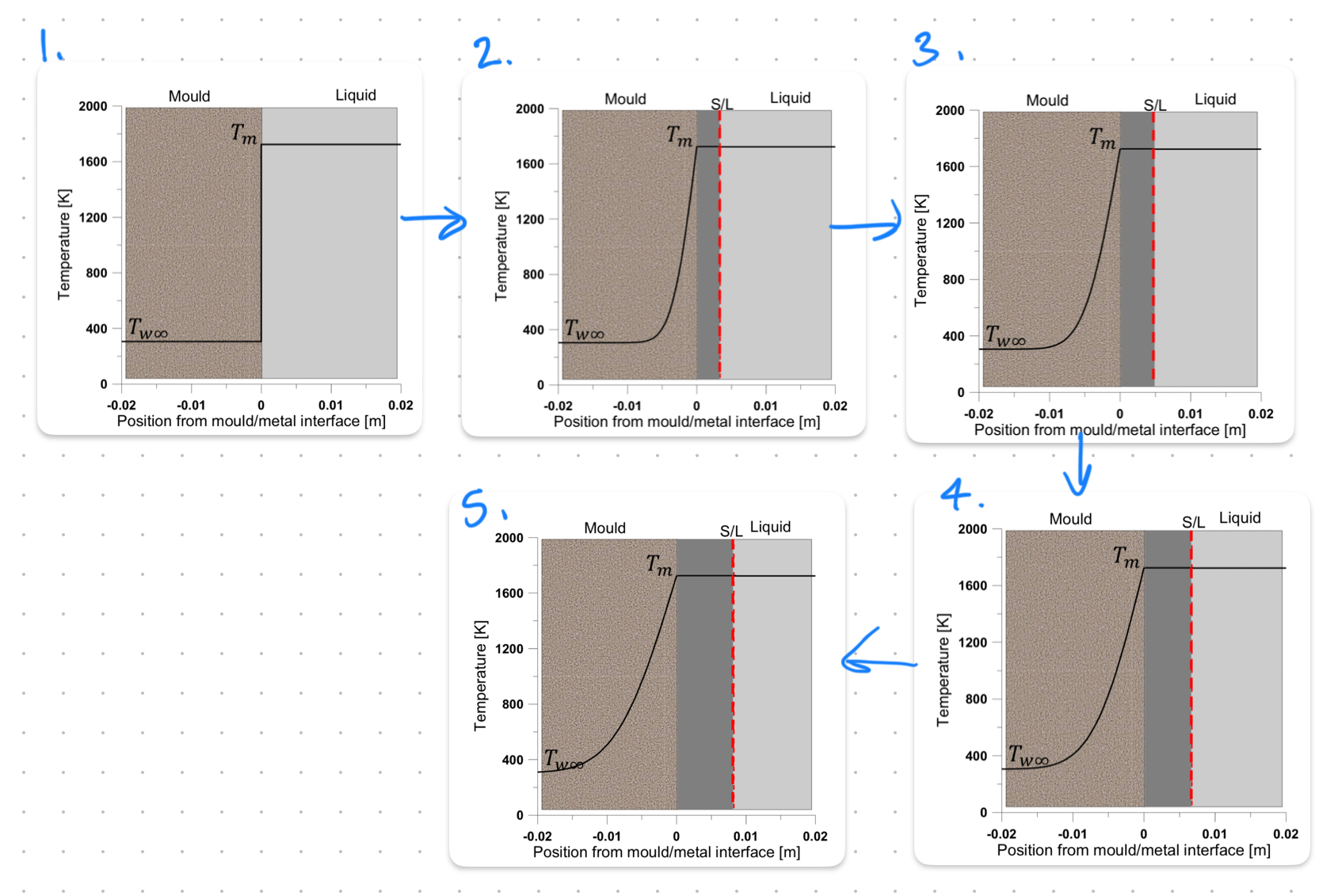
Initial State: Liquid metal at melting temperature (Tm); mould at lower temperature (TW∞).
Nucleation Start: S/L interface forms; steep temperature gradient develops.
Solidification Progress: S/L front moves into the liquid; heat conduction continues.
Solid Growth: Solid region expands; temperature gradient stabilizes in the mould.
Cooling Completion: More solid forms; system cools toward thermal equilibrium.
Solute Diffusion:
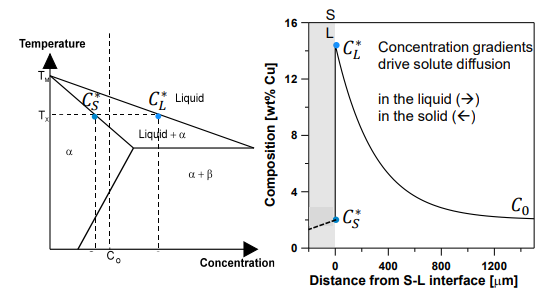
Solute Partitioning: Solute is partitioned between the solid and liquid at the S-L interface. The liquid at the interface has a higher solute content than the bulk liquid.
Concentration Gradients: These gradients drive solute diffusion in the liquid and solid.
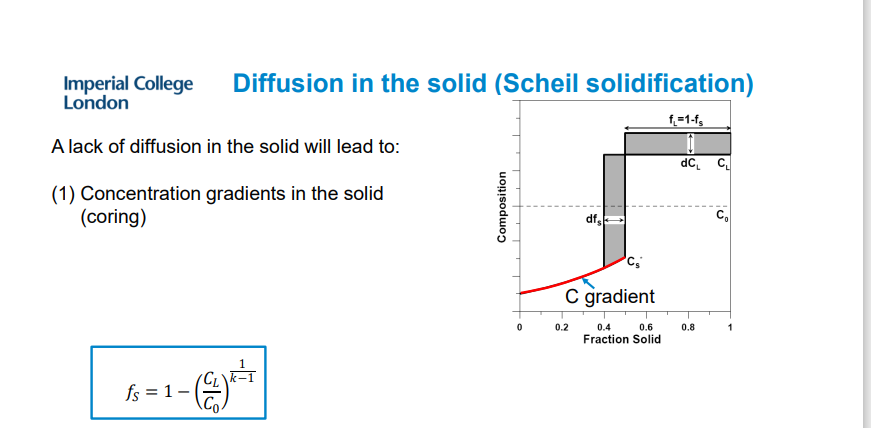
Scheil Equation: Describes the composition of the solid when there is no diffusion in the solid:

2. Constitutional Supercooling
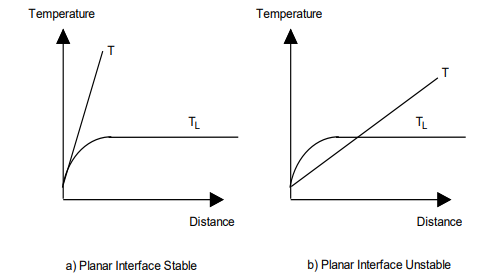
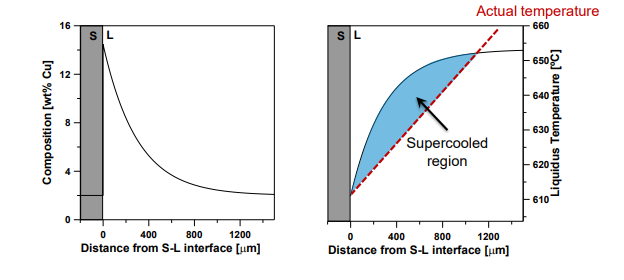
What is Constitutional Supercooling?:
Occurs when the actual temperature in the liquid is below the liquidus temperature, creating a supercooled region ahead of the interface.
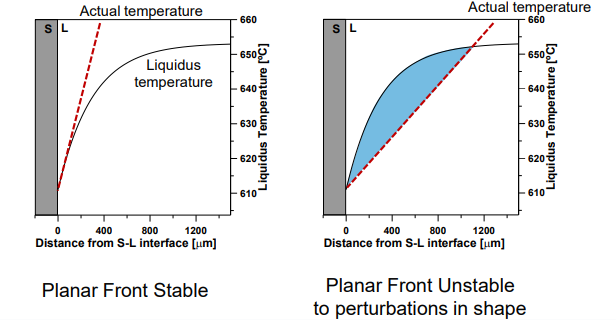
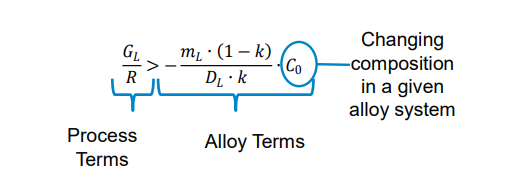
Condition for Stability: A planar interface is stable if:

Gradient in Liquidus Temperature:
The gradient in liquidus temperature at the interface is given by:

Instability and Growth Forms:
If GL is too shallow, the planar interface becomes unstable, leading to:
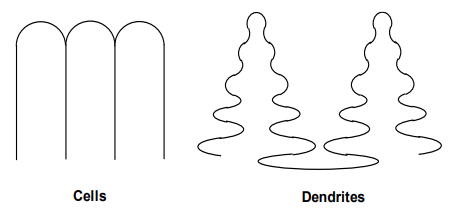
Cellular Growth: Intermediate GL/R.
Dendritic Growth: Low GL/R.
3. Effect of Process Parameters and Alloy Composition
Process Parameters:
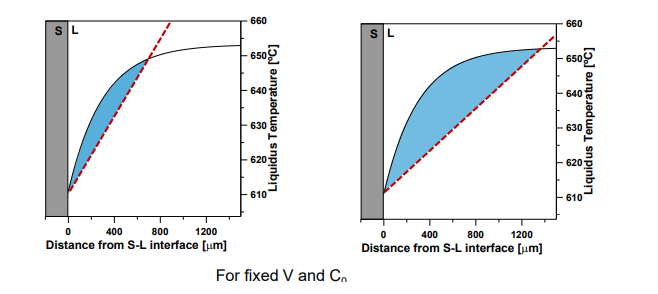
Decreasing GL(Temp grad in liq):
Increases constitutional supercooling.
Increases the distance over which supercooling occurs.
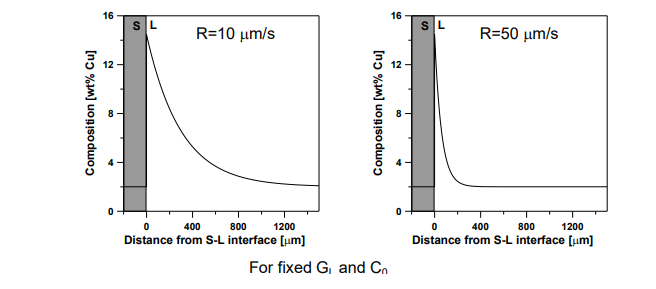
Increasing Growth Rate (R):
Leads to steeper solute gradients.
Increases constitutional supercooling.
Alloy Composition:
Increasing C0:
Increases solute rejection at the interface.
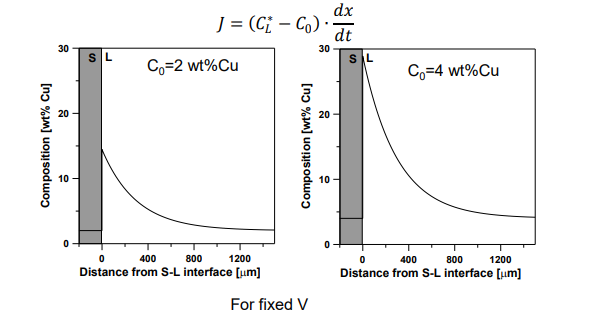
Leads to more constitutional supercooling.
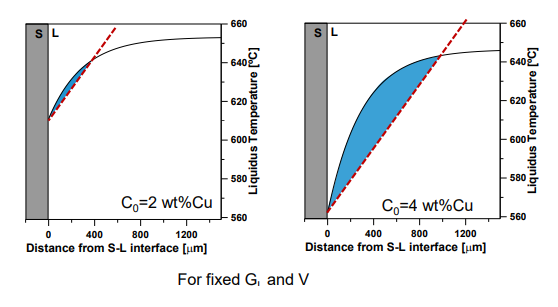
4. Steady-State Solute Diffusion
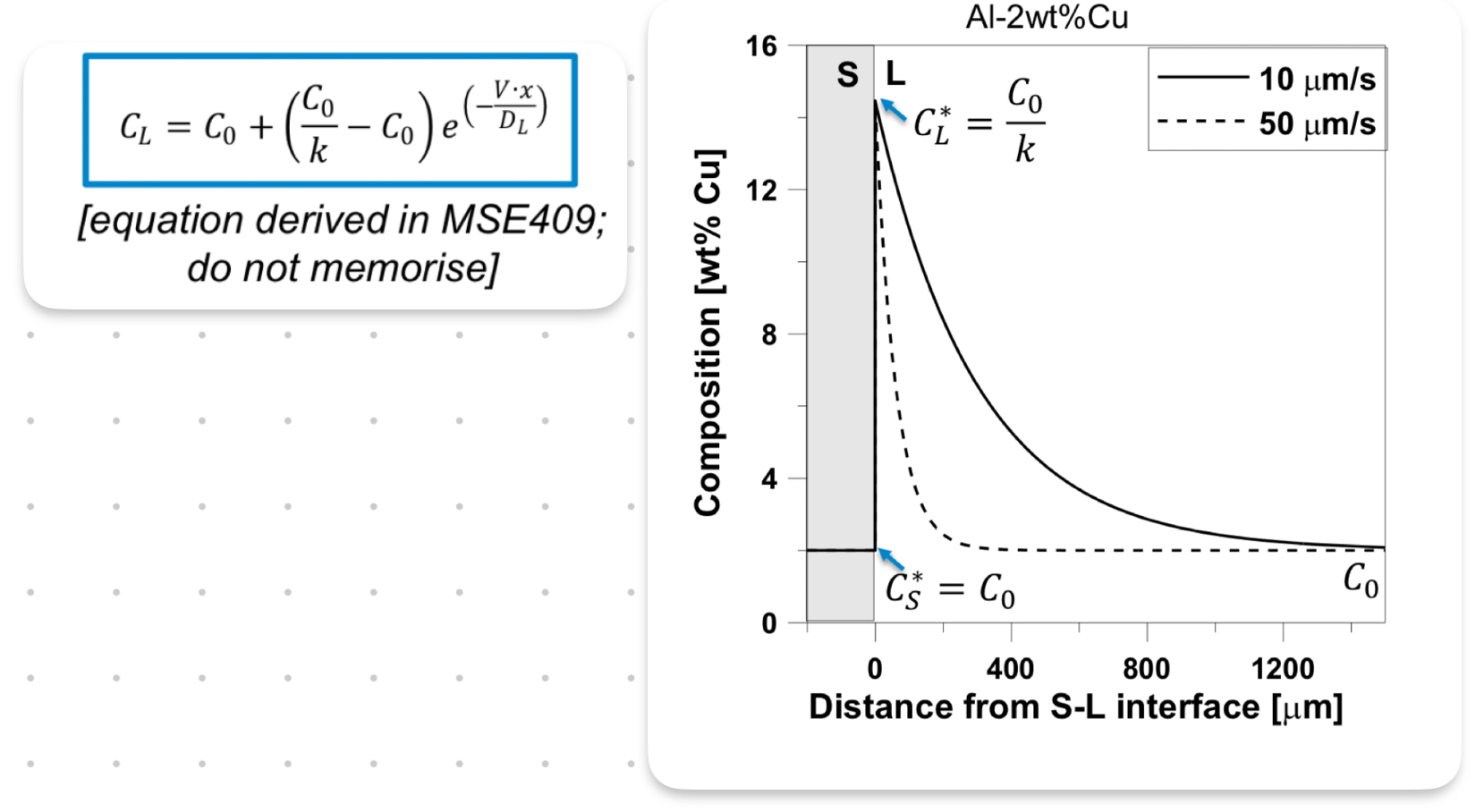
Solute Diffusion Profile:
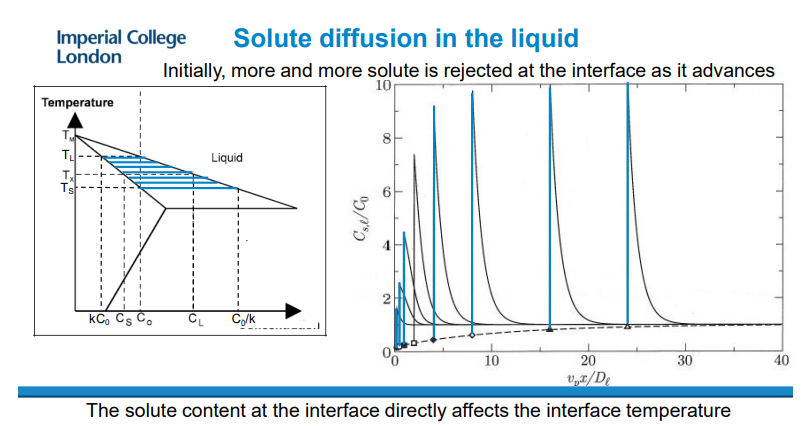
Solute diffusion in a liquid assumes that the interface is at equilibrium and that the composition of the liquid becomes increasingly concentrated.
As the concentration increases, the temperature decreases, meaning the interface must be cooling.
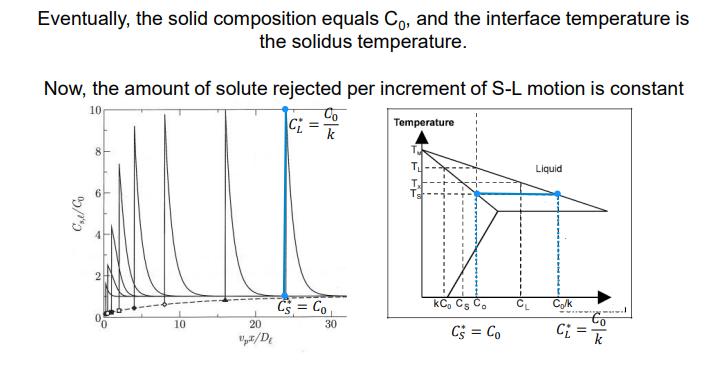
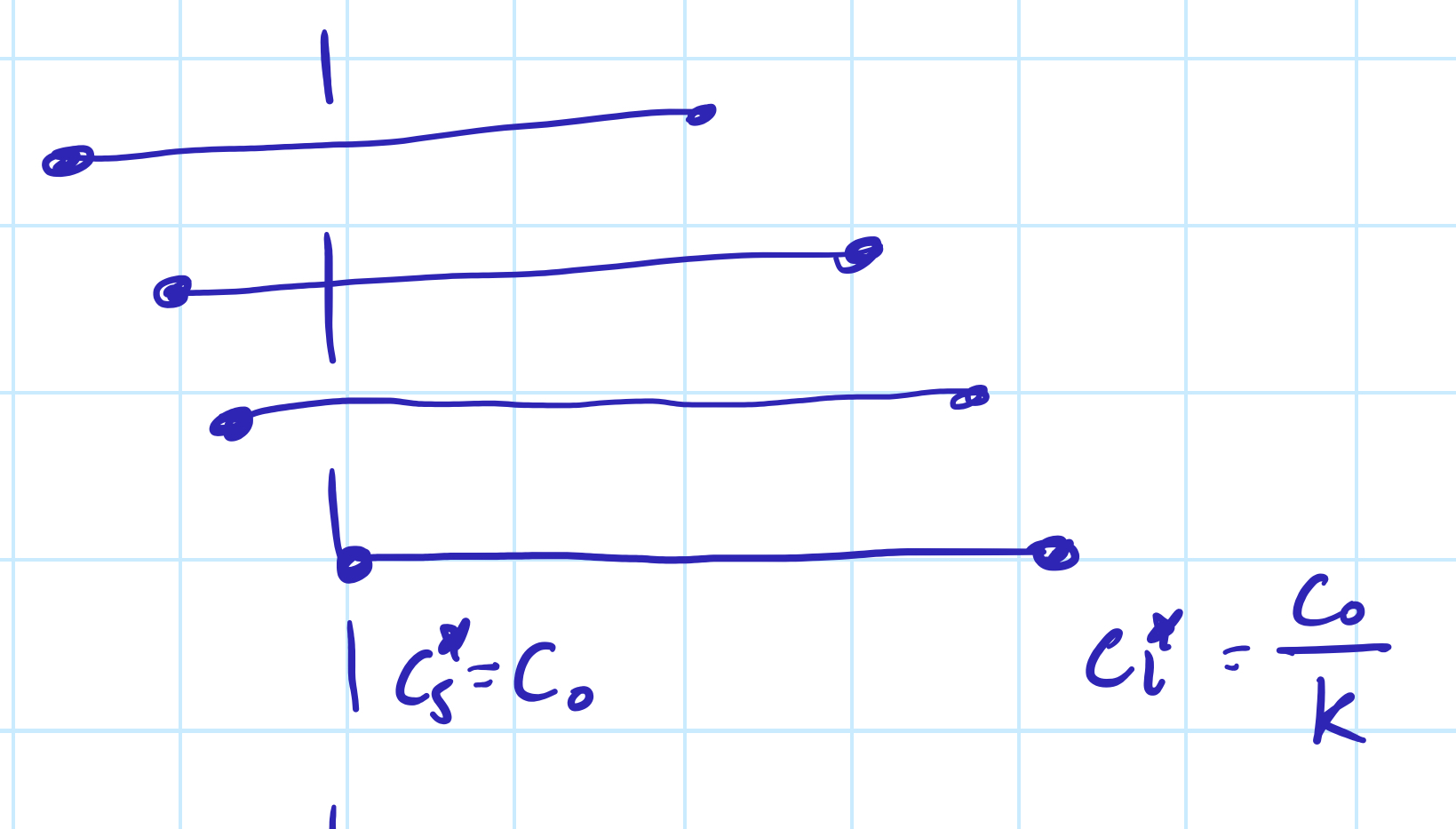
k = Cs*/Cl*

Interface Flux Equation:
The rate of solute diffusion at the interface must balance the rate of solute rejection:

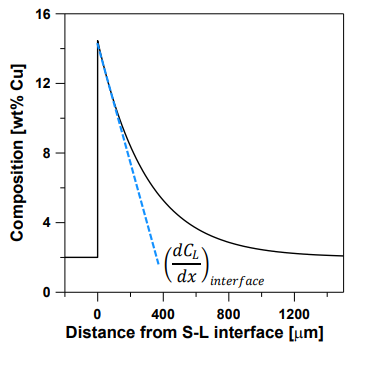
The interface tip is where solute diffusion away equals solute rejection by the solid. As the interface moves, composition stays constant. Flux is negative as solute moves from high to low concentration.
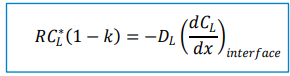
5. Growth Forms
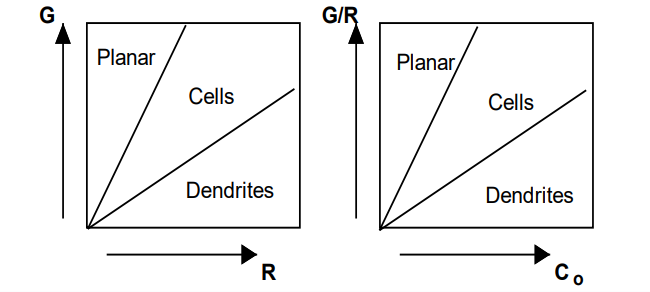
Planar Growth:
Stable at high GL/R.
Occurs when the temperature gradient is steep enough to prevent constitutional supercooling.
Cellular Growth:
Forms at intermediate GL/R
Occurs when the temperature gradient is not steep enough to maintain a planar interface.
Dendritic Growth:
Forms at low GL/R.
Occurs when the temperature gradient is very shallow, leading to tree-like structures