Aerobic Metabolism
Aerobic Metabolism: Overview
Substrates and Main Pathways
Two primary pathways discussed: Krebs cycle (citric acid cycle) and Electron Transport Chain (ETC) with oxidative phosphorylation (the process that links them).
Oxidative phosphorylation = oxidative respiration = mitochondria “breathing,” using oxygen to add a phosphate to ADP to form ATP.
Substrates feeding aerobic metabolism:
Glucose (from glycolysis) continues through glycolysis → pyruvate → acetyl-CoA (via pyruvate dehydrogenase).
Free fatty acids (FFAs) are transported to muscle and enter beta-oxidation to become acetyl-CoA to then enter the mitochondria and the Kreb cycle.
Amino acids contribute very minimally to ATP production in this context; generally considered negligible under normal conditions because their contribution is slow and limited.
Important caveat: transport of substrates into mitochondria involves membrane transport steps (two membranes) and the carnitine shuttle for fatty acids.
Glycolysis and Pyruvate Dehydrogenase (PDH) Bridge to Aerobic Metabolism
Glycolysis overview (revisited): glucose → pyruvate via 10 steps; net production includes ATP and NADH. The key idea is that glycolysis provides ATP rapidly but also produces NADH; under aerobic conditions, pyruvate is further oxidized rather than building up lactate.
Pyruvate to acetyl-CoA:
Pyruvate + CoA + NAD⁺ → acetyl-CoA + CO₂ + NADH
This step feeds acetyl-CoA into the Krebs cycle.
Important interconnections:
NAD⁺ is required for glycolysis; later, NADH produced feeds the ETC, linking glycolysis to oxidative phosphorylation.
CO₂ is a byproduct of PDH and Krebs cycle, explaining why we breathe out CO₂ during exercise.
Krebs Cycle (Citric Acid Cycle)
Core concept: the cycle requires oxaloacetate and acetyl-CoA to proceed; if one is limiting, the cycle slows or stalls.
Entry point and key enzymes:
Acetyl-CoA combines with oxaloacetate via citrate synthase to form citrate.
Isocitrate is converted through several steps (aconitase, isocitrate dehydrogenase) with NADH and CO₂ production.
Alpha-ketoglutarate dehydrogenase produces NADH and CO₂; a key regulatory step is alpha-ketoglutarate dehydrogenase.
Succinyl-CoA synthetase produces GTP (or ATP in some tissues) and succinate.
Succinate dehydrogenase (a part of the ETC) generates FADH₂.
Fumarase adds H₂O; malate dehydrogenase produces NADH and regenerates oxaloacetate.
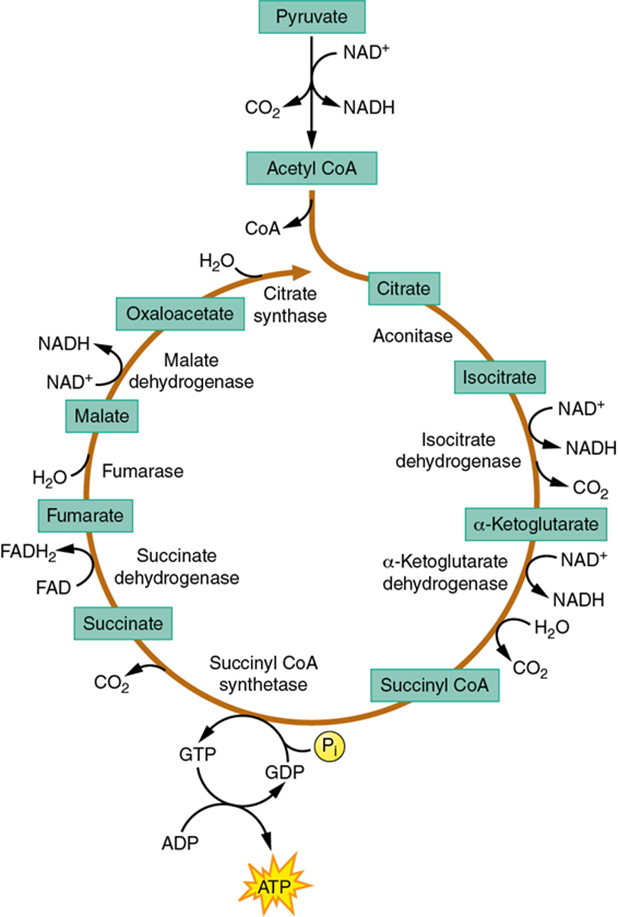
Per-acetyl-CoA yield (typical simplification):
4 NADH + 1 FADH₂ + 1 GTP (ATP) + 4 CO₂
For a glucose molecule, the cycle runs twice (two acetyl-CoA molecules enter per glucose).
Therefore, per glucose: 8 NADH, 2 FADH₂, 2 GTP/ATP, and 8 CO₂ (before ETC contributions).
Regulatory notes discussed:
Isocitrate dehydrogenase = rate-limiting step in the Krebs cycle.
Oxaloacetate must be available to accept acetyl-CoA; otherwise, acetyl-CoA accumulates and the cycle slows.
The phrase “fat burned in the flame of carbohydrate” highlights the need for some carbohydrate-derived oxaloacetate to keep the cycle going when fat oxidation is high.
Carbohydrate dependence for fat oxidation:
If there is not enough oxaloacetate (i.e., insufficient carbohydrate intake), acetyl-CoA cannot enter the cycle efficiently, limiting fat oxidation.
Lipid Metabolism: Lipolysis and Beta-Oxidation
Lipolysis (lipid breakdown):
Triglycerides (TAGs) are hydrolyzed by lipases to yield a glycerol backbone and three free fatty acids (FFAs).
The main rate-limiting enzyme for lipolysis is hormone-sensitive lipase.
Glycerol can be used for gluconeogenesis in the liver but not as readily in muscle cells; FFAs are the primary energy source for muscle during aerobic metabolism.
The three FFAs are released into circulation and bound to albumin for transport in the bloodstream.
The glycerol backbone is largely redirected to liver for glucose synthesis; FFAs are transported to muscle for oxidation.
Carnitine shuttle and mitochondrial entry:
Fatty acids need carnitine to cross the outer mitochondrial membrane; acetyl-carnitine is an intermediate step for inner mitochondrial entry.
Once inside, beta-oxidation shortens the fatty acid by two-carbon units to form acetyl-CoA, which then enters the Krebs cycle.
Beta-oxidation details:
Longer fatty acid chains yield more acetyl-CoA and thus more ATP overall than shorter chains; the energy yield scales with chain length.
Example given: a 16-carbon fatty acid can yield about
which then enter the Krebs cycle and ETC to yield a large amount of ATP (noted as about 129 ATP per 16-carbon FFAs in the lecture; multiply by the number of fatty acids in a triacylglycerol for full TAG yield).
Glycerol metabolism:
Glycerol from TAGs can be converted to glucose in the liver; this glucose can then be used by muscle, but this process is relatively slow.
Most FFAs enter directly into muscle metabolism and fatty acid oxidation is a major energy source during prolonged exercise.
Fatty acids vs. carbohydrate energy balance:
FFAs provide a high-energy yield, contributing to sustained aerobic energy production.
The liver and adipose tissue are key sources of circulating FFAs during prolonged exercise or fasting.
Ketone Bodies and Ketogenesis
Ketogenesis occurs in the liver when carbohydrate availability is very low and acetyl-CoA accumulates from beta-oxidation.
Ketone bodies produced:
Acetoacetate, beta-hydroxybutyrate, and acetone (the latter often detected in breath).
Ketone use:
Ketones can be used by the brain and other tissues as an alternative energy source when glucose is scarce.
Ketone energy vs. carbohydrate energy:
Ketones are a lower-energy substrate compared to glucose-derived ATP, but they provide essential energy when carbs are low and help spare glucose for essential tissues.
Ketosis vs. diabetic ketoacidosis (DKA):
Nutritional ketosis (e.g., ketogenic diet) is a controlled state with elevated ketones but not pathological acidosis.
DKA is a dangerous clinical state (often in type 1 diabetes) with high glucose, high ketones, acidosis, dehydration, and potential for cerebral edema if fluids/insulin are not managed carefully.
Ketone Metabolism in Health and Disease (Practical Context)
Ketone production as a response to low carbohydrate intake or impaired carbohydrate metabolism.
Ketosis can be therapeutically relevant in some disease states, but can cause side effects (dehydration, insomnia, constipation, potential osteoporosis with prolonged high protein/low carb regimens).
Ketone breath (acetone) is a clinical sign sometimes observed in ketosis.
Ketosis can be dangerous when accompanied by insulin deficiency, leading to DKA.
Diabetic Ketoacidosis (DKA) Case Study: Illustration and Management
Presentation features in a typical pediatric case:
Hyperglycemia (e.g., capillary glucose around 400 mg/dL).
Elevated ketone bodies (e.g., 3.2 mmol/L).
Metabolic acidosis (low pH, e.g., pH ~7.21) and anion gap metabolic acidosis.
Signs of volume depletion: dehydration; poor urine output.
Kussmaul respiration (compensatory breathing pattern).
Triggers can include infection (e.g., urinary tract infection).
Laboratory and diagnostic steps:
Glucose measurement with glucometer; ketone measurement (serum ketone meter); blood gas analysis to assess acidosis; urine ketones testing.
Anion gap considerations and evaluation of pH to quantify severity.
Initial treatment protocol:
Fluids: IV normal saline bolus followed by maintenance IV fluids (e.g., 20 mL/kg/h) with gradual adjustment.
Insulin therapy: regular insulin infusion at 0.1 units/kg/h after initial fluid bolus to correct hyperglycemia and acidosis.
Potassium management: monitor serum potassium; start K⁺ supplementation (KCl) as needed due to shifts with insulin and improved insulin sensitivity.
Glucose management: once plasma glucose approaches ~200 mg/dL, switch to IV fluids with dextrose (e.g., 5% dextrose in normal saline) to prevent hypoglycemia and cerebral edema during correction.
Monitoring and safety: careful monitoring of electrolyte balance, hydration status, and neurological status to guard against cerebral edema during rehydration/insulin therapy.
Outcome considerations:
DKA is often triggered by infection (e.g., UTI) and can be managed successfully with IV fluids, insulin, and electrolyte correction in an ICU setting.
The case study emphasizes the need to measure glucose, ketones, pH, and electrolytes, and to manage fluids and insulin carefully to avoid complications.
Aerobic Exercise: Role, Adaptations, and Practical Implications
Aerobic dominance under rest and prolonged, low-to-moderate intensity exercise:
The aerobic system is most utilized at rest and during long-duration, low-to-moderate intensity exercise.
Intensity zones: typically 60–70% VO₂ max for building a strong aerobic base; higher athletes may reach 80–90% in extreme cases.
Recovery and post-exercise dynamics:
Excess post-exercise oxygen consumption (EPOC) reflects the restoration processes after exercise (phosphocreatine resynthesis, glycogen replenishment, heart rate normalization).
Time course of adaptation and training considerations:
Aerobic metabolism adapts slowly to changes in exercise intensity; initial minutes of high intensity recruit anaerobic metabolism before stabilizing into steady-state aerobic metabolism.
Training focuses on increasing mitochondrial density and metabolic efficiency for longer, lower-intensity work.
Training prescription and energy system focus:
Long-duration, low-intensity workouts aim to maximize mitochondrial density and oxidative capacity.
Moderate-to-long endurance workouts (e.g., marathon-like volumes) increase mitochondrial quantity and improve oxidative enzyme activity.
Physiological adaptations (systemic and muscle-level):
Systemic adaptations:
Cardiac adaptations: increased stroke volume, allowing higher cardiac output with lower heart rate for the same work, improved blood flow distribution.
Blood adaptations: potential increase in hematocrit with training; initial body water expansion can mask hematocrit changes early on.
Vascular adaptations: improved vasodilation and blood flow to working muscles; more efficient oxygen transport.
Muscle-specific adaptations:
Increased mitochondrial density (more mitochondria per muscle fiber).
Increased aerobic enzyme activity (e.g., isocitrate dehydrogenase) to accelerate the Krebs cycle.
Increased intramuscular triglyceride storage (IMTG) and utilization (minor but notable adaptation).
Improved glycogen sparing (better ability to sustain high-intensity effort by conserving muscle glycogen).
Hormonal adaptations (brief overview):
Hormonal responses support increased fat oxidation and red blood cell production under hypoxic or altitude-like conditions (erythropoietin, EPO).
Chronic adaptations to aerobic training can include improved insulin sensitivity and metabolic flexibility.
Practical application examples and student discussion:
Alternative activities: swimming, cycling, pickleball, and other moderate-to-long duration activities emphasize aerobic metabolism depending on pace and effort.
Pros and cons of relying primarily on aerobic glycolysis (oxidative metabolism):
Pros:
Very energy-dense: ATP yield per glucose is high when fully oxidized; Supports sustained, moderate-intensity activity with large energy stores (liver glycogen, intramuscular triglycerides).
Does not acutely acidify as much as anaerobic metabolism; can sustain work for longer periods.
Cons:
Slower to produce energy; limited capacity for high-intensity efforts due to oxygen delivery and utilization limits.
Fat metabolism requires more oxygen; at very high intensities, carbohydrate metabolism is favored.
Respiratory implications and RER:
Respiratory Exchange Ratio (RER) indicates substrate use: closer to 0.7 indicates fat oxidation; closer to 1.0 indicates carbohydrate oxidation.
Practical clinical and sports science tie-ins:
Understanding substrate use informs dietary recommendations (carbohydrate availability, fat adaptation strategies) and training programs.
Isocitrate dehydrogenase as a key regulatory enzyme in the Krebs cycle highlights potential control points for metabolic modulation.
Conceptual takeaway: the body can burn fat and carbohydrate in a coordinated manner; efficient fat oxidation depends on having some carbohydrate to supply oxaloacetate for the Krebs cycle, enabling continued acetyl-CoA entry and sustained energy production.
Key Formulas and Numerical References (LaTeX)
Pyruvate oxidation to acetyl-CoA:
Krebs cycle per acetyl-CoA (simplified):
Per glucose (two acetyl-CoA enter the Krebs cycle):
Electron Transport Chain: general ATP yield per NADH and FADH₂ (illustrative values from lecture):
NADH → approximately 2.5–3 ATP (depending on shuttle);
FADH₂ → approximately 1.5 ATP;
Overall, about 36 ATP per glucose in brain/muscle could be cited in some contexts; the lecture notes reference 36 ATP with the gradient-driven mechanism and 6 H⁺ → 3 ATP for a given step in ATP synthase; NADH enters earlier (more protons pumped) vs FADH₂ entering later (fewer protons pumped).
ATP yield from beta-oxidation (example):
For a 16-carbon fatty acid: approximately (summed from beta-oxidation cycles, Krebs cycle turns, and ETC).
Gas exchange and substrate utilization:
Fat oxidation is associated with an RER near ; carbohydrate oxidation is near .
Ketone bodies and energy considerations:
Ketogenic energy sources provide energy with acetyl-CoA derived from fatty acid oxidation when carbohydrate supply is limited; ketones are lower energy than glucose but sustain cells when glucose is scarce.
Connections, Implications, and Takeaways
“Fat burnt in the flame of carbohydrate” underlines the necessity of a baseline carbohydrate availability to keep oxaloacetate present for acetyl-CoA to enter the Krebs cycle during fat oxidation.
Aerobic metabolism is the dominant energy system for endurance and recovery, powering long-duration activities and rebuilding ATP between bouts via oxidative phosphorylation and phosphocreatine replenishment.
The efficiency of energy systems is affected by substrate availability, mitochondrial density, enzyme activities (e.g., isocitrate dehydrogenase), hormonal milieu, blood flow, and training adaptations.
Clinical relevance: understanding the metabolic pathways helps explain conditions like DKA, the role of insulin deficiency, and how interventions with fluids and insulin correct metabolic derangements while avoiding cerebral edema.
Real-world relevance: athletes can optimize training and nutrition to enhance mitochondrial density, substrate utilization, and recovery (e.g., through long, low-intensity training, adequate carbohydrate intake to sustain oxaloacetate, and fat-adaptation strategies with caution).
Quick Recap (Top Takeaways)
Aerobic metabolism relies on glucose and fatty acids to produce ATP via glycolysis, the Krebs cycle, and the ETC; fatty acids contribute significantly during prolonged activity through beta-oxidation, producing acetyl-CoA for the Krebs cycle.
The Krebs cycle requires acetyl-CoA and oxaloacetate; fat oxidation can overwhelm the cycle without sufficient oxaloacetate (carbohydrate-derived) and thus carbohydrate availability is critical for sustained fat metabolism.
The ETC uses NADH and FADH₂ to pump protons and generate ATP via ATP synthase; NADH yields more ATP than FADH₂ due to earlier entry points in the chain.
Ketone bodies provide an alternative energy source during low carbohydrate availability; DKA is a dangerous condition requiring urgent medical management.
Aerobic adaptations include increased mitochondrial density, enhanced oxidative enzyme activity, improved cardiovascular function (stroke volume, hematocrit), and better substrate utilization with training.