Chp 2 Atoms, Molecules & Ions
2.1 Early Ideas in Atomic Theory
Dalton’s Atomic Theory
matter made out of atoms
elements are only made out of one type of atom
atoms from one element differ from atoms from all other elements
compounds. two or more atoms. the ratio is always consistent. (ratio is whole number)
Atoms not destoryed nor created in chem change just rearranged
Law of definite proportions/constant composition
#s of atoms of a given element always exist in the same ratio
Law of multiple proportions
when two elements form more than one compound →will react with masses of the other element in a ratio of small, whole numbers
2.2 Evolution of Atomic Theory
electron - negatively charged subatomic particle
alpha particles - 2 protons + 2 neutrons
Erenest Rutherford findings
atoms have a large amount of empty space
atoms have nucleus. small (relatively heavy) positive charge at their center.
proton - positively charged subatomic particle @ nucleus
isotopes - atoms @ same element that differ in mass
What did James Chadwick find?
neutrons - uncharged subatomic particles mass roughly the same as a proton
→ neutrons explain isotopes bc they change the mass but not the charges
2.3 Atomic Structure & Symbolism
what do we use to measure atoms?
unified atomic mass unit (u) = dalton (Da)
fundamental unit of charge (e)
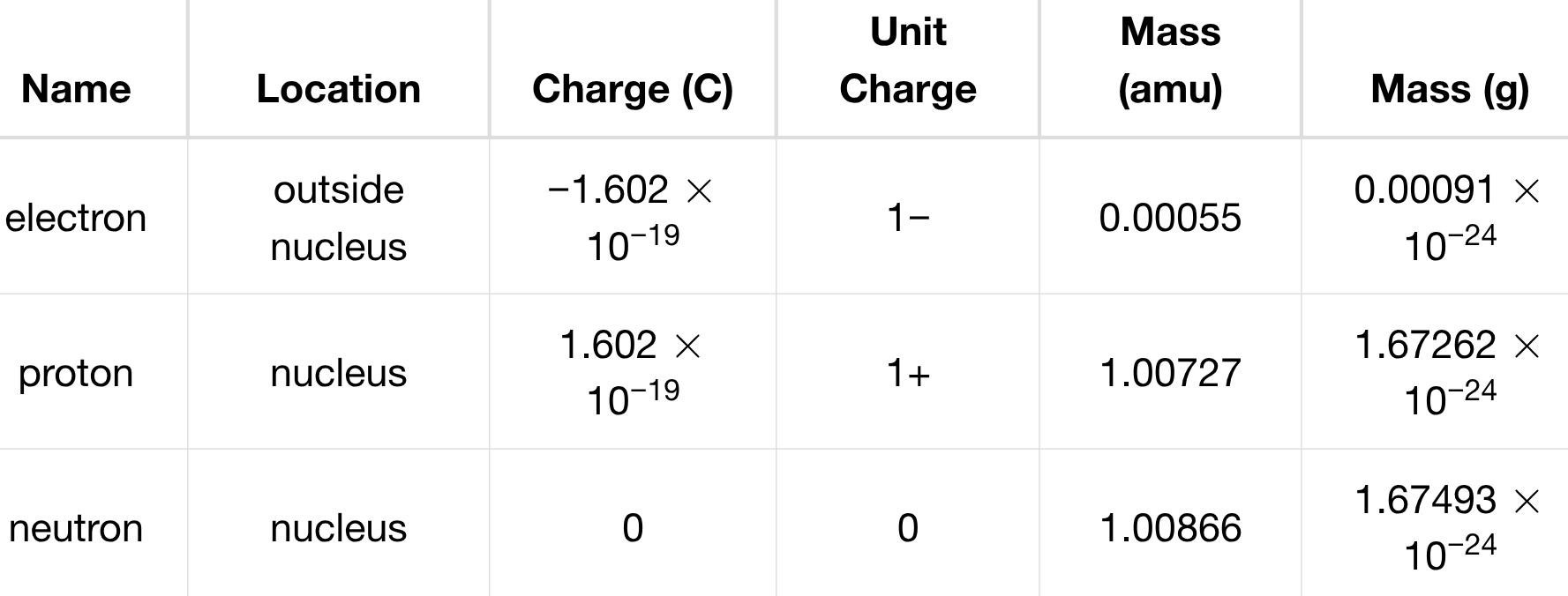
atomic number (Z)
# of protons in the nucleus @ atom
→ defining trait. determines atom’s identity
atomic number shows → mass # (A)
total # of protons & neutrons
ion - when subatomic particles are not equal
anion - gains 1 or more electrons → negative charge
cations - losses 1 or more electrons → positive charge
Chemical symbols - abbreviation for element/atom of element
microscopic → Hg one atom of mercury
macroscopic →Hg conatiner of mercury
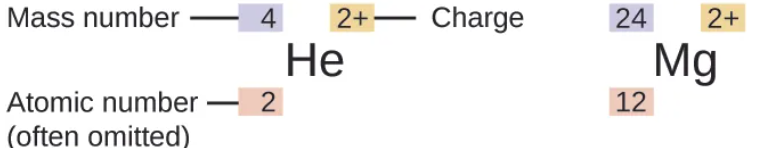
atomic mass - it’s amu. the amount of protons & neutrons in an atom
not a single atom weighs that much it is the average of all the atoms
2.4 Chemical Formulas
molecular formula - rep @ molecule uses chemical symbols to indicate the types of atoms, followed by subscripts to show the number of atoms of each type in the molecule.

structural formula - same as molecular formula EXCEPT shows how atoms are connected @ molecule
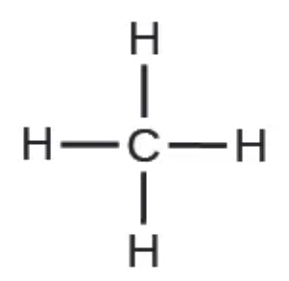
What’s the rule for subscripts & for # before the chem symbol?
before the chem symbol then it is two separate atoms
if subscript it is connected
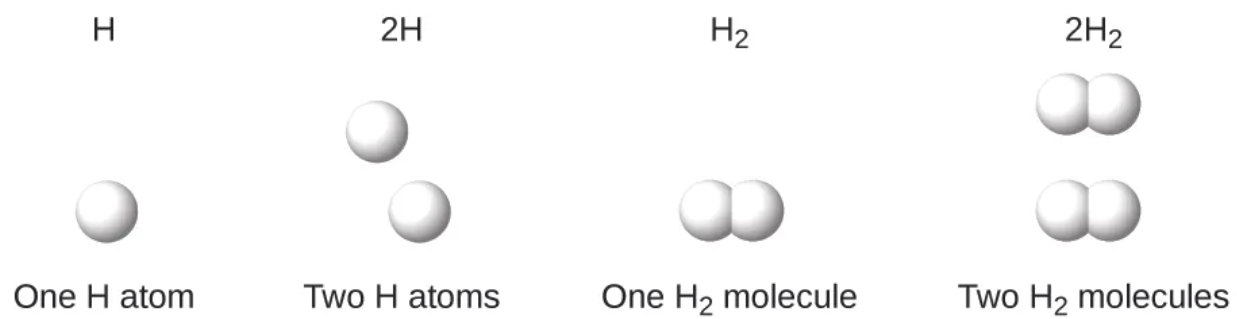
empirical formula - simplest whole # ratio of atoms (or ions) in a compound

isomers - compounds with the same chemical formula but different molecular structure
structural isomers
spatial isomers - relative orientations of the atoms in space can be different.
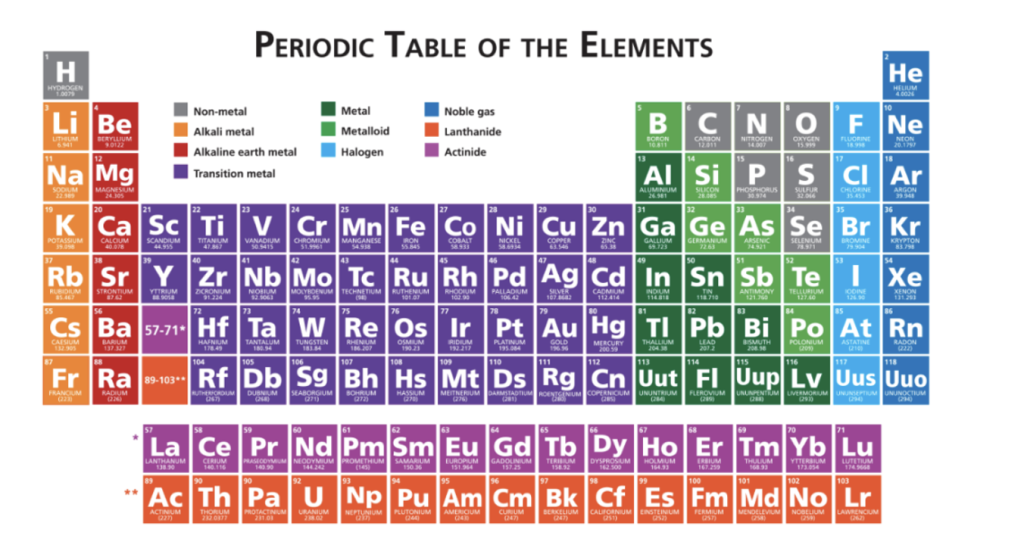
2.5 The Periodic Table
periodic law - properties of elements are in periodic functions of their atomic #
Overarching classes of elements:
metals - shiny, malleable, good conductors of heat and electricity
nonmetals - dull, poor conductors of heat and electricity
metalloids - conduct heat and electricity moderately well (between metals & nonmetals)
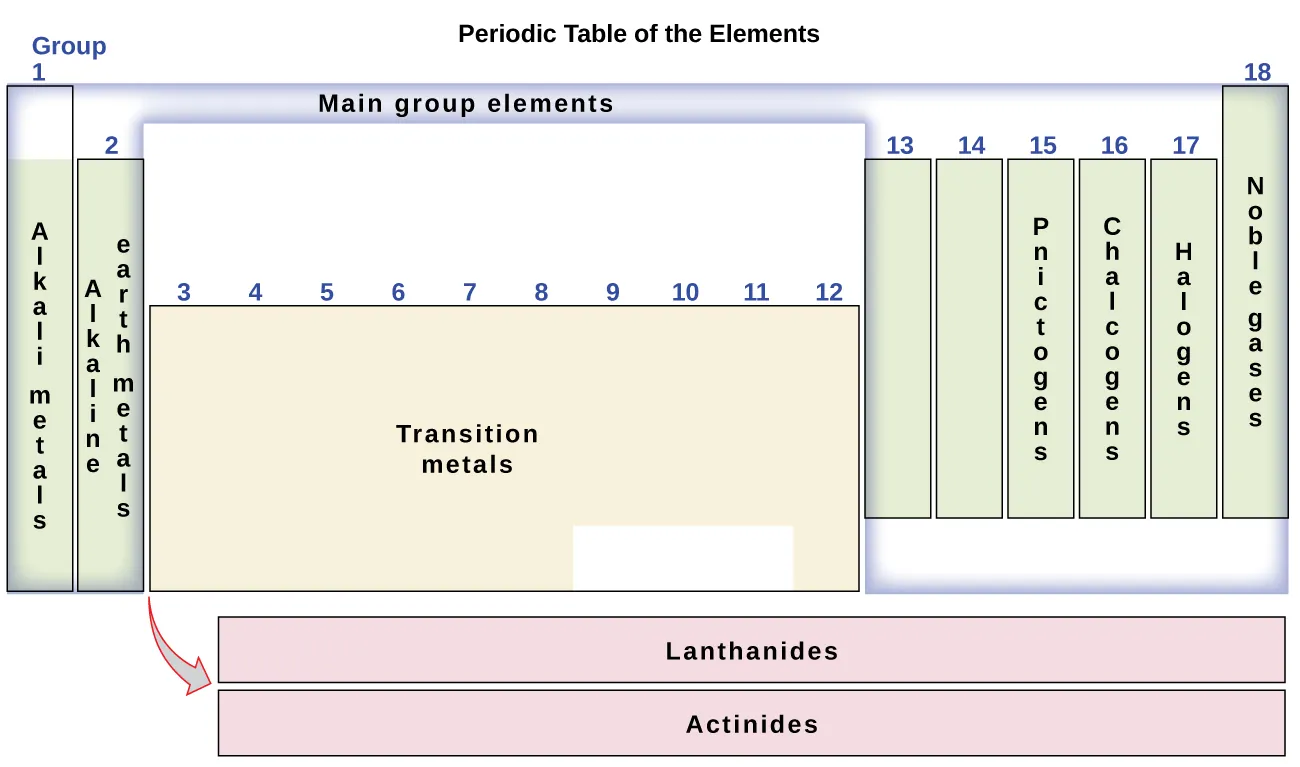
group 1 - representative/main group elements
group 2 - representative/main group elements. alkali earth metals
group 3 - transition metals
group 4 - transition metals
group 5 - transition metals
group 6 - transition metals
group 7 - transition metals
group 8 - transition metals
group 9 - transition metals
group 10 - transition metals
group 11 - transition metals
group 12 - transition metals
group 13 - representative/main group elements
group 14 - representative/main group elements
group 15 - representative/main group elements. pnictogens
group 16 - representative/main group elements. chalcogens
group 17 - representative/main group elements. halogens.
group 18 - representative/main group elements. noble gases/inert gases
inner transition metals in the two rows at the bottom of the table (the top-row elements are called lanthanides and the bottom-row elements are actinides
2.6 Ions & Molecular Compunds
when electrons transferred & ions formed → ionic bonds
electrons shared → covalent bonds
ionic compound
contains ions & held together by ionic bonds
solids, melt at high temps, boils at higher temps
does not conduct electricity
formula is ratio of ions → positive & negative charges equal
why is the formula important? * do practice problems of this & predicting bonds
EX: Al³+ and O²-
ionic compounds MUST BE electrically neutral so these are equal… with 6 charges on each side
final formula:
molecular compounds (aka covalent compounds)
often gases
low boiling & low melting solids (there are exceptions though)
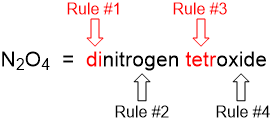
2.7 Chemical Nomenclature
nomenclature - collection of rules about naming things
binary compounds - only consisting of two elements
ionic compounds
kind of compound | rules | EXs |
only monatomic ions | cation then anion. suffix “-ide”
| Na2O, sodium oxide |
polyatomic ions | cation then anion | Al2(CO3)3, aluminum carbonate |
compounds containing metal ion w/ variable charge
named cation then anion
metal ion’s charge is shown in roman numerals
EX: iron (II) chloride, the iron has a charge of 2+
ionic hydrates
hydrates - ionic compounds that contain water molecules as integral components of their crystals
# of water molecules + suffix hydrate

Molecular (Covalent) Compounds
names indicate the ratio
Compounds Composed of Two Elements
1st name of the more metallic element (the one farther to the left and/or bottom of the periodic table)
2nd nonmetallatic element with “-ide” ending
Binary Acids
what is a binary acid? →compound comprised of hydrogen & one other nonmetallic element)
Naming:
hydrogen →prefix “hydro-”
nonmetallic element suffix “-ic”
Add acid at the end!
EX: Hydrochloric acid, HCl
Oxyacids
What is an oxyacid? → compounds that contain hydrogen, oxygen & one other element
hydrogen, oxygen & one other element
naming: no “hydrogen”, root of anion,
naming: –ate →–ic, or –ite → –ous
EX: , nitrous acid