Groups in the Periodic Table
The periodic table: a list of elements arranged in order of increasing atomic number.
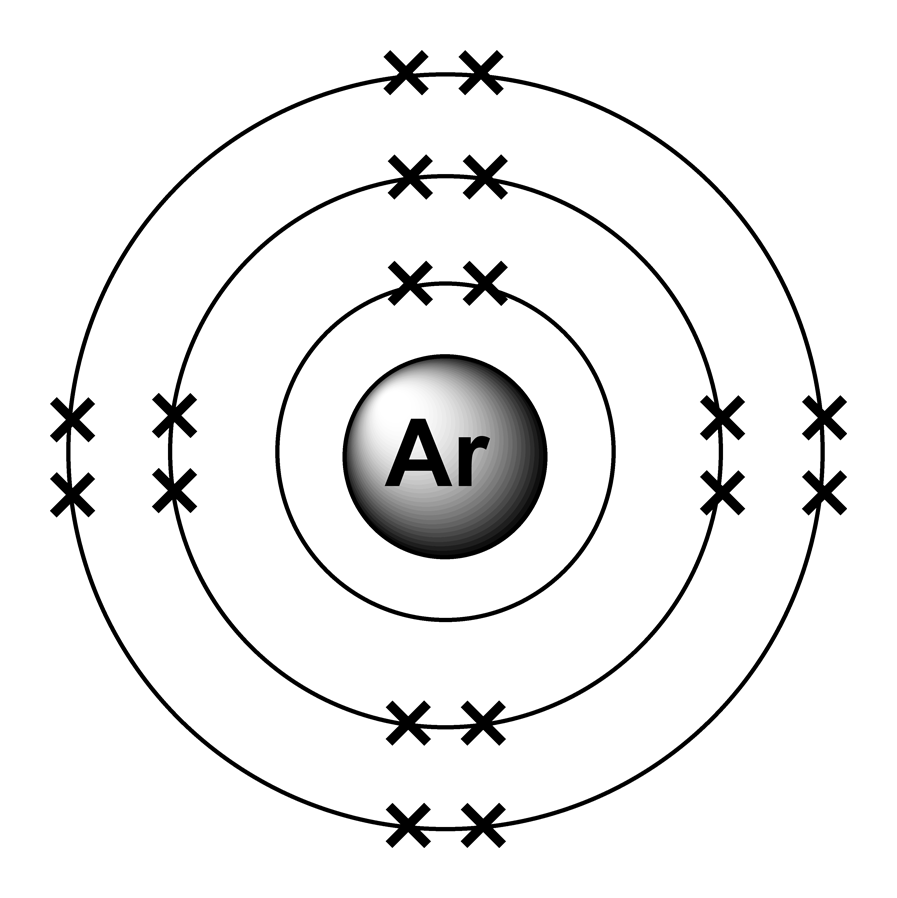
The electronic configuration of the first 20 Elements is shown below:
For groups 1-7 the group number tells us the number of electrons in the outer most shell, as it is the outer or valence electrons that determine an elements chemical reactivity, it follows that elements in the same group have similar chemical properties as they have same number of electrons in their outer shell or same valency
The period number tells us the total number of shells containing electrons.
Metal and Non-Metals
The periodic table can be split into metals and non-metals by the zig-zag line that starts between group 2 and 3 in period 3.
You may wonder who decided where to draw the line. There are numerous ways to classify elements as metals and non-metals and you may previously have used physical properties such as appearance to distinguish between them. E.g. metals are often shiny when freshly cut, metals are malleable etc.
Elements can also be classified by their chemical properties, in this case the acid/base nature of their oxides.
Oxides of metals form bases e.g. magnesium oxide, MgO
Oxides of non-metals form acids e.g. sulfur dioxide, SO2
8.2 – Group 1, The Alkali Metals
The alkali metals can be found in group 1 of the periodic table.
Physical Properties
Silvery/shiny when freshly cut
Soft (easily cut with a knife) – become softer as you move down the group
Low density – float on water
Conduct electricity
Chemical Properties
They react with water to give a metal hydroxide and hydrogen
E.g. sodium + water → sodium hydroxide + hydrogen
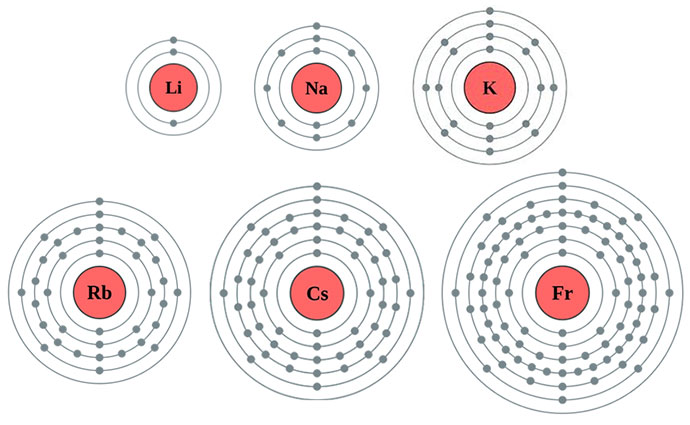
They become more reactive with increasing atomic number (as you go down the group)
The reaction of Alkali Metals with Water
The alkali metals all react rapidly with water producing hydrogen gas and the corresponding metal hydroxide. This similarity in reactivity creates a family of elements referred to as a group. The similarity in chemical properties within a group is due to all the members having the same number of electrons in the outer shell.
Observation for Li
Li is stored in oil to prevent it reacting with air
Li is soft enough to cut with a knife
Li floats on water (its density must be less than that of water)
Li moves slowly around the surface of the water
Li fizzes when in reacting with water (hydrogen gas is given off)
The piece of Li disappears once it has all reacted
The water has become alkaline due to the formation of lithium hydroxide
2Li(s) + 2H2O(l) → 2LiOH(aq) + H2(g)
Observations for Na
Na is stored in oil to prevent it reacting with air
Na is softer than Li
Na floats on water (its density must be less than that of water)
The heat of the reaction melts the Na into ball
Na moves quickly around the surface of the water
Na fizzes rapidly when in reacting with water (hydrogen gas is given off)
The piece of Na disappears once it has all reacted
The water has become alkaline due to the formation of sodium hydroxide
2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
Observations for K
K is stored in oil to prevent it reacting with air
K is softer than Na
K floats on water (its density must be less than that of water)
The heat of the reaction melts the K into ball
K moves very quickly around the surface of the water
K fizzes violently when in reacting with water (hydrogen gas is given off)
The K ignites and burns with a lilac flame
The piece of K disappears once it has all reacted
The water has become alkaline due to the formation of potassium hydroxide
2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)
Summary
Alkali metal | ||||
Observation | Li | Na | K | |
Physical properties | Cut with a knife | soft | softer | softest |
Floats on water | ✓ | ✓ | ✓ | |
Chemical properties | Stored under oil | ✓ | ✓ | ✓ |
Disappears | ✓ | ✓ | ✓ | |
Melts into a ball | 🗶 | ✓ | ✓ | |
Flame | 🗶 | 🗶 | ✓ (lilac) | |
Moves across the water | slowly | quickly | very quickly | |
Fizzing | steady | rapid | violent | |
Explaining the trend in reactivity of the alkali metals
It is clear from the reactions of alkali metals with water that they become more reactive as you go down the group.
When alkali metals react they lose their outer electron to for positive ions, e.g.
Na → Na+ + e-
As we go down the group, the outer electron is further away from the nucleus and there are more shells of electrons between the nucleus and outer electron increasing the amount of shielding. Accordingly, the outer electron is held less tightly and lost more easily so the reactivity increases.
8.3 – Group 7, The Halogens and Redox
The halogens are all non-metals and found in group 7 of the periodic table. As elements they all occur as diatomic molecules. There are trends in some of the physical properties as you go down the group:
They become darker in colour
Their mpt/bpt increases (hence the gradual change in state at room temperature from gas to liquid to solid)
Key physical properties that you must learn have been highlighted in bold type. The other you can predict based on the trends listed above.
Name | Formula | Colour | Physical State at rtp | Colour in solution |
Fluorine | F2 | Yellow | Gas | |
Chlorine | Cl2 | Green | Gas | Pale yellow |
Bromine | Br2 | Red/Brown | Liquid | Orange |
Iodine | I2 | Dark Grey | Solid | Dark reddish brown |
Astatine | At2 | Black | Solid |
Test for Chlorine gas
Put in a piece of damp blue litmus paper
The paper goes red and then white (bleached) if chlorine gas is present
Relative reactivities of the Halogens
The halogens become less reactive as you go down the group. This can be illustrated by comparing their reactions with iron or hydrogen. As a result a more reactive halogen will displace a less reactive halogen from a solution of one of its salts.
Eg. If you add colourless chlorine water to a colourless solution of potassium bromide the chlorine will displace the bromine from the potassium bromide as chlorine is more reactive. The solution turns orange as bromine is formed.
Cl2(aq) + 2KBr(aq) → 2KCl(aq) + Br2(aq)
Orange solution
Colourless solutions
To form the ionic equation we re-write the equation splitting ionic compounds into their ions
Cl2(aq) + 2K+(aq) +2Br-(aq) → 2K+(aq) +2Cl-(aq) + Br2(aq)
Remove any ions that appear on both sides of the arrow, these are called spectator ions
Cl2(aq) + 2K+(aq) +2Br-(aq) → 2K+(aq) +2Cl-(aq) + Br2(aq)
What remains is the ionic equation
Reduction
Cl2(aq) +2Br-(aq) → 2Cl-(aq) + Br2(aq)
Oxidation
From the ionic equation we see that these displacement reactions are also redox reactions.
In the above example:
Cl2 has gained electrons and been reduced to form Cl- ions
Br- ions have lost electrons and been oxidised to form Br2
Explaining the trend on reactivity of the halogens
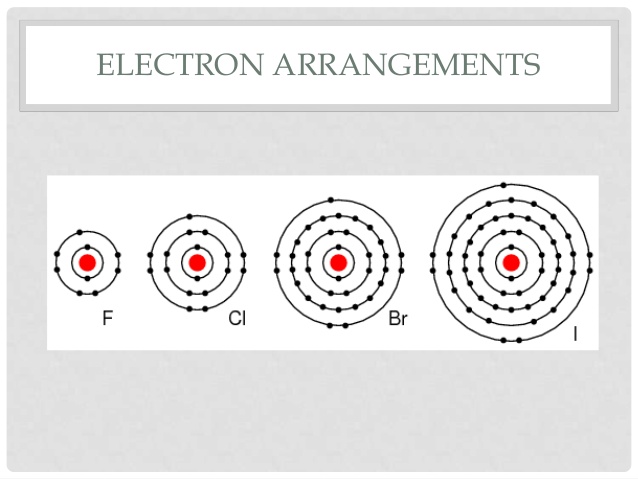
Halogens become less reactive as you go down the group this is because going down the group there are more shells and the outer shell gets further away from the nucleus. The electron gained will join the outer shell, as this get further from the nucleus the electrostatic attraction between the incoming electron and the nucleus decreases. Furthermore, as the number of shells increases, the amount of shielding also increases.
This can be seen in the electron configurations below:
An incoming electron will much further away from the nucleus and there are 4 inner shells shielding the outer electrons in iodine
8.4 – Group 0, The Noble Gases
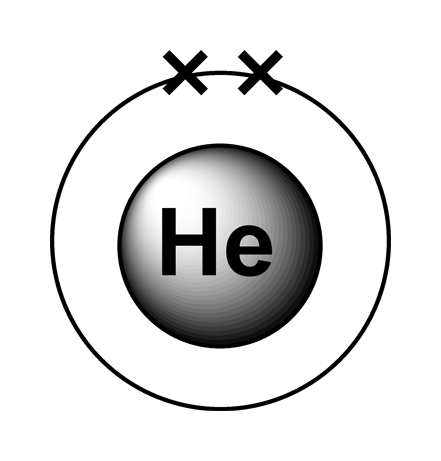
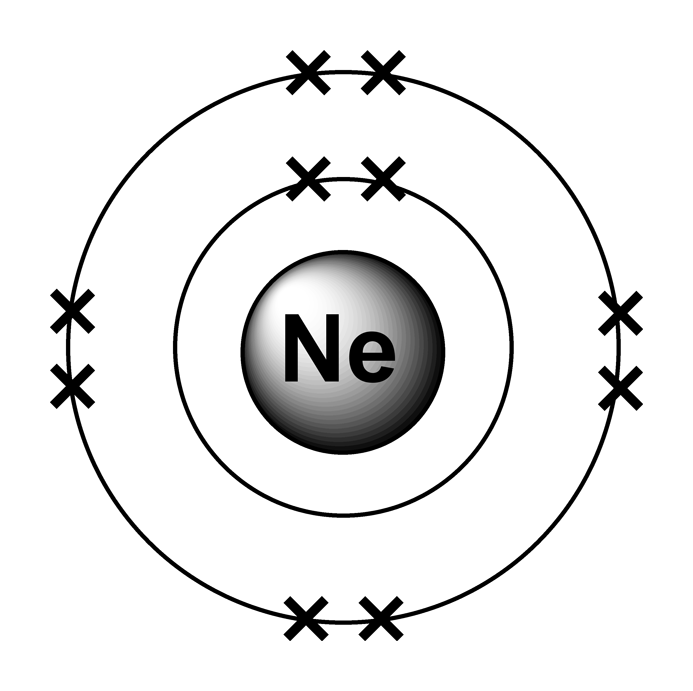
The noble gases are a family of inert gases found in group 0 of the periodic table. They are very unreactive because they all have full valence shells and hence have a valency of 0.
The atoms hold onto their electrons too tightly to form covalent bonds or +ve ions. They don’t gain electrons to form –ve ions because the electrons would have to go into the next shell but as this is further away from the nucleus the nuclear attraction would be insufficient.
A group is a vertical column in the periodic table. Hydrogen is special and is often put on its own.
Valence electrons are outermost electrons
Valency can be simply thought of as the number of hydrogen atoms an element can combine with assuming all bonding is covalent (this a simplification)
A period is a horizontal row in the periodic table
Inner shells of electrons repel the outer electrons (reducing the effective nuclear charge i.e. the net positive charge that the outer electron is exposed to), this effect is called shielding or screening
mpt = melting point; bpt = boiling point
rtp = room temperature and pressure
At low concentration this may appear as colourless
Bromine produces a red/brown vapour
Iodine produces a violet vapour when heated
Iodine has a very low solubility so you may also see a dark grey precipitate of iodine
This experiment can also be done by bubbling chlorine gas through the potassium bromide solution
Halogens are not very soluble in water but are more soluble in an organic solvent called cyclohexane which is often added and shaken with the mixture. Cyclohexane and water are immiscible so two layers form with most of the halogen dissolved in the cyclohexane layer. As cyclohexane is less dense than water it will be the top layer. The colours of the halogen solutions when dissolved in cyclohexane are: Chlorine gives a colourless solution, Bromine gives an orange solution and Iodine gives a violet solution.
A redox reaction is a reaction in which both oxidation and reduction occur
The noble gases are so unreactive it was once thought that they didn’t react with anything, hence they were called inert. It was discovered in the 1960s that under certain conditions they could be made to react and so they are now referred to as the noble gases.