Coulombic Attraction (Forces)
Attraction between oppositely charges particles (p+ & e-)
amount of charge (magnitude of charge) + distance between charged particles
periodic - repeating over a given interval
Rules
When you increase the number of shells/distance, the attractive force will decrease.
When you increase the number of protons attractive force will increase.
As you move from smallest to largest atom, the attractive force will become weaker.
Each electron gets the full attractive force of the nucleus.
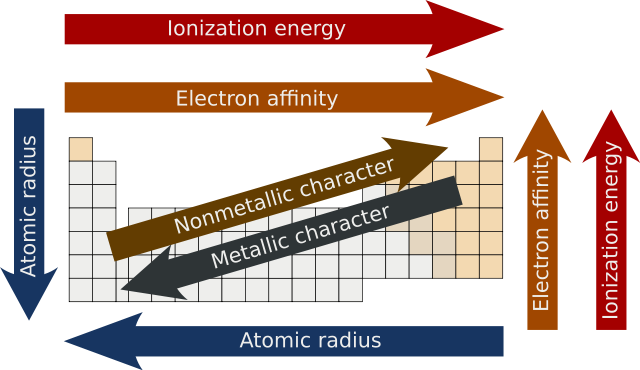
5 Different Trends