5. Cytokine therapeutics
Cytokine and its role
- Cytokines are small proteins that act as signalling molecules in the immune system.
- They are secreted by cells of the innate and adaptive immune system.
- Their effect is mediated by binding to specific receptors on the surface of target cells.
- Cytokines play a central role in regulating the immune response. They mediate communication with immune cells, influencing the movement of immune cells to sites of infection or injury.
- There is no “one cytokine – one cell” rule:
- Made by many cell types
- Same cytokine made by different cell types
- Cytokine receptors are found on many cell types
Examples of cytokines: interleukins, interferons, tumor necrosis factors (TNFs), and chemokines.
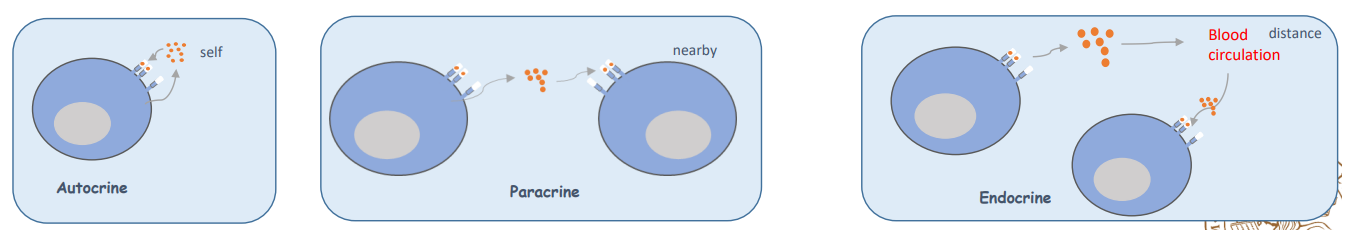
Cytokines’ mechanism of actions- Three different ways that cytokines can act in response to an activating stimulus and upon binding to their receptor:
1) Autocrine manner: acting on cells that produce it (helps them to get activated)
2) Paracrine manner: acting on cells nearby those that produce it (they can act on other cells nearby)
3) Endocrine manner: Secreted by one tissue into the blood, and acting on a different tissue. This depends on their ability to enter the blood circulation and their half-life
Cytokine traits- They have different traits
• Pleiotropy: Many different effects e.g. activation, differentiation, proliferation on tissue on different cell types by the same cytokine
• Redundancy: Different cytokines can have the same effect on one particular cell type
• Synergy: The effect of two cytokines together on one cell type is greater than the effect
of one cytokine alone
• Antagonism: The effects of one cytokine inhibit those of another
Cytokines and the immune system
• Cytokines produced as a result of activation of the innate immune system
•Typical cytokines: Type I interferon, interleukin (IL-) 1, IL-6, tumor necrosis factor (TNF)
• Cytokines produced as a result of activation of the adaptive immune system
• Interferon-γ, IL-2, IL-4, IL-5
• Cytokines that stimulate immature leukocyte growth and differentiation
• IL-3, colony-stimulating factors
All interleukins are cytokines, but not all cytokines are called interleukins
Role of cytokines inflammation-
Inflammation: Detected some damaged or abnormal activity
Inflammatory response initiated within hours of infection/wounding!
A state of inflammation is induced in the tissue by cytokines
1) Destruct invading microorganisms
2) Induce local blood clotting
3) Repair injured tissue
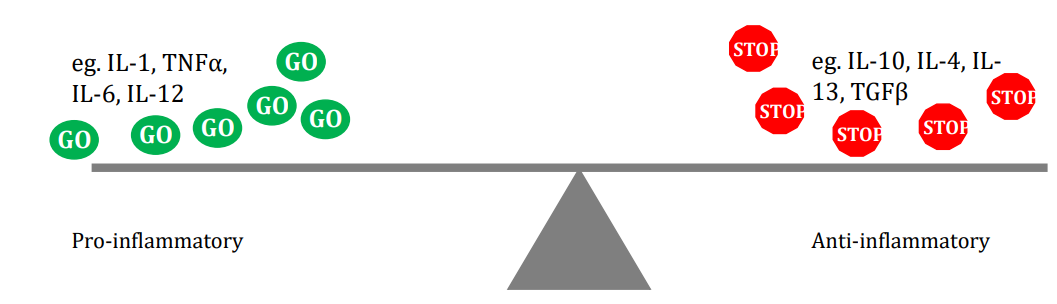
Pro-inflammatory vs anti-inflammatory cytokines-
Pro-inflammatory cytokines:
- Set-up/initiate an immune response against infection/injury.
- Eg. IL-1, TNFα, IL-6, IL-12
Anti-inflammatory cytokines:
- Control and inhibit the release of pro-inflammatory mediators (to keep a balance)
- Limit the inflammatory response
- eg. IL-10, IL-4, IL-13, TGFβ
There needs to be a balance between pro-inflammatory and anti-inflammatory cytokines in a healthy immune response.
Too many pro-inflammatory cytokines → sepsis
Too many anti-inflammatory cytokines → Immunosuppression
IL-1 family
- 11 members (incl. IL-1α, IL-1β, IL-18, IL-33), both pro- and anti-inflammatory
- Two main members: IL-1α and IL-1β (pro-inflammatory).
- Promotes the activity of cells of the innate immune system (e.g. neutrophils, eosinophils, basophils etc.)
- Activates and reinforces T cell function
- T helper 17 (TH17) cells require IL-1 to differentiate from naive T cells
- TH1 cells are mainly affected by IL-18
- TH2 cells are mainly affected by IL-33
IL-1 dysregulation is implicated in autoimmune diseases (e.g. psoriasis and lupus), autoinflammatory diseases (e.g. Chron’s disease and UC) and asthma.
IL-1 family
• Comprised of 11 members (incl. IL-1α, IL-1β, IL-18, IL-33), both pro- and anti-inflammatory members
• Promote the activity of cells of the innate immune system (e.g. neutrophils, eosinophils, basophils etc)
• Activate and reinforce T cell function
• T helper 17 (TH17) cells require IL-1 to differentiate from naïve T cells
• TH17 cells mediate autoimmune and chronic inflammatory diseases
• TH1 cells are mainly affected by IL-18
• TH2 cells are mainly affected by IL-33
• Closely related receptors – signalling!
IL-6
- Pro-inflammatory cytokine
- Expressed by mononuclear phagocytes, T cells, B cells, fibroblasts, endothelial cells and many more
- It has pleiotropic activity involved in:
- Haematopoiesis (blood cell formation)
- Maturation of B cells into antibody-producing plasma cells
- Activation of T cells
- plays a role in the differentiation and regulation of helper T cells type 2 and Treg phenotype.
- Signals through a specific receptor complex. It binds to the IL-6 receptor α chain and the signal transducing component gp130, which triggers intracellular signaling events that mediate the biological effects of IL-6 on target cells
- Systemic and local inflammatory effects
- Anti-IL-6 antibodies as treatment for many diseases (e.g. SLE, rheumatoid arthritis)
TNFα-
- Pro-inflammatory cytokine
- Mainly secreted from activated macrophages, monocytes, NK cells, T cells, neurons and others.
- Pleiotropic cytokine
- Stimulates cell proliferation
- Exerts cytolytic and cytostatic activity against tumour cells
- plays a role in the body’s defense against cancer
- Has antiviral effects and plays a role in regulating the immune system
- Linked to various physiological processes, including blood coagulation, insulin resistance and endothelial function
IL-2
- Pleiotropic cytokine
- Mainly produced b Secreted predominantly by antigen-stimulated CD4+ T cells
- Produced also by CD8 T cells, NK cells, activated DCs
- Stimulate differentiation of naïve CD8 T cells, into memory T cells
Cytokine release syndrome
• Massive T cell stimulation → overproduction of cytokine (it was called cytokine storm)
• One of the most frequent adverse events of such therapies
• Systemic inflammatory response, triggered by infections, drugs (antibody, proptein and non-protein based) and other factors
• Observed in the setting of transplantation and GVHD
• The severity of CRS can vary from mild to severe/life-threatening
A long way to go..
- Cytokines’ half life is small – limits their efficacy (excert their function)
- Systemic administration
- Limited efficacy
- More toxicities!
- Why are many immunotherapies given at a late disease stage? Risk of systemic, not a high response. Their immune system is tired → they can’t respond as you wish
- Why are they given to specific subcategories of diseased patients?
Know at least two examples of cytokine therapeutics used for the treatment of diseases.
anti-TNFα therapy-
- Monoclonal antibodies that target TNFα (anti-TNF therapy) to treat autoimmune diseases such as rheumatoid arthritis.
- By inhibiting TNFα, the overactive immune response is regulated.
- An example of Anti-TNF Therapy is Humira
IL-2 therapy
- IL-2 stimulates the proliferation and maturation of T cells.
- Aldesleukin (a recombinant form of IL-2) can be used as a treatment for metastatic melanoma and renal cell carcinoma.
- This stimulates T cells to target and attack cancer cells.
- Often provided as a systemic injection (short half-life), which can lead to toxicities (e.g. cytokine release syndrome).