Alkanes
Alkanes
General Formula - CnH2n+2
Only contains C and H atoms
Each C atom has 4 bonds
Saturated - only contain single bonds
Cycloalkanes - when the C atoms form a ring structure (CnH2n)
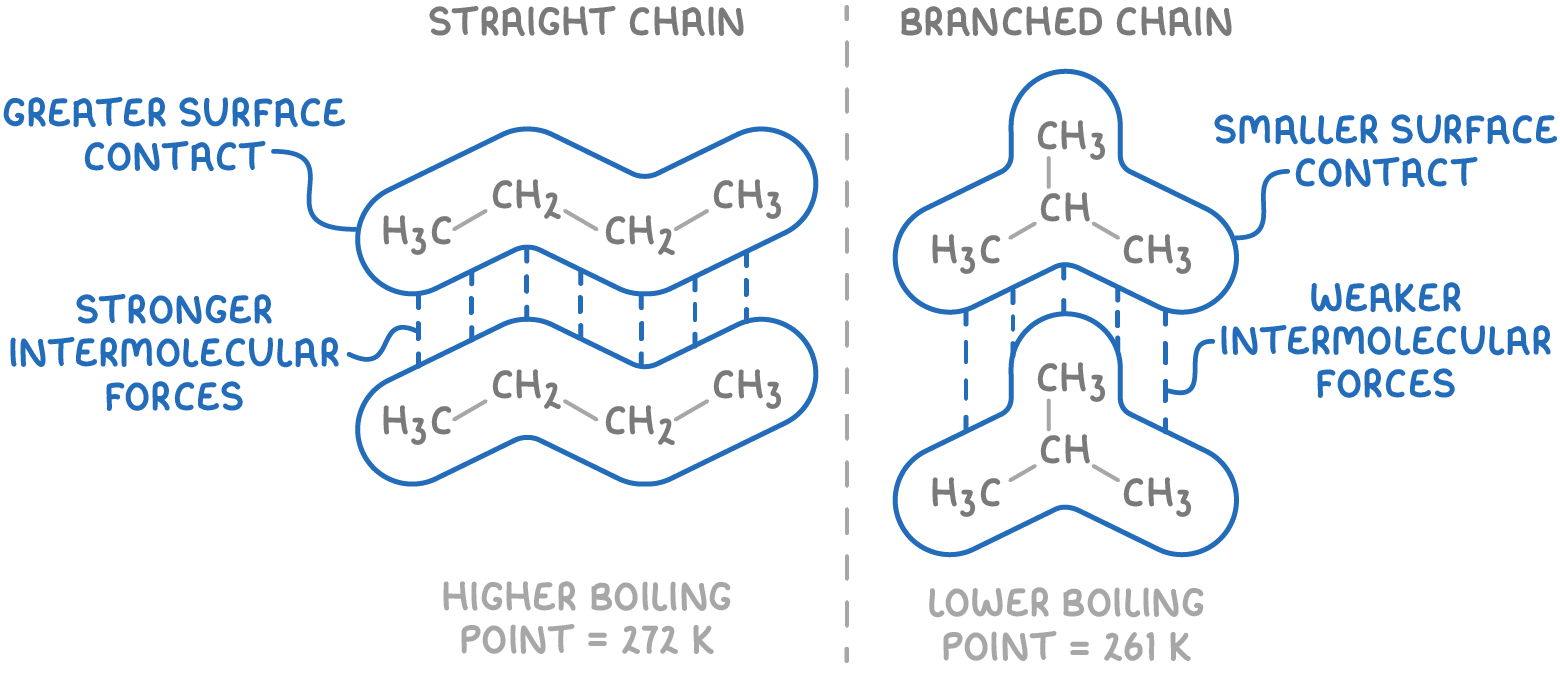
Shapes of alkanes
4 bonding pairs of electrons
Arranged in a tetrahedral shape due to the equal repulsion of electron pairs
Bond angle = 109.5°
e.g, Methane (CH4)
How structure affects boiling points
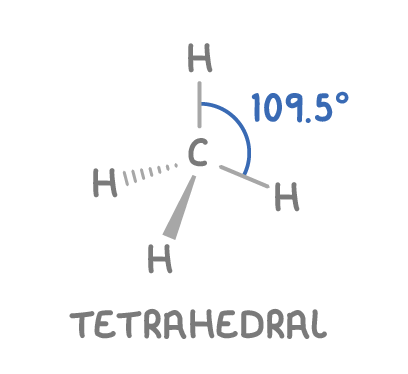
Boiling point of an alkane is influences by the strength of its intermolecular induced dipole-dipole forces, which vary based on length of the carbon chain and the extent of branch
Carbon chain length
Longer C chain means more electrons creating stronger temporary induced dipole
These stronger dipoles result in stronger induced dipole-dipole forces between molecules
More energy is needed to overcome these forces and boil the alkane
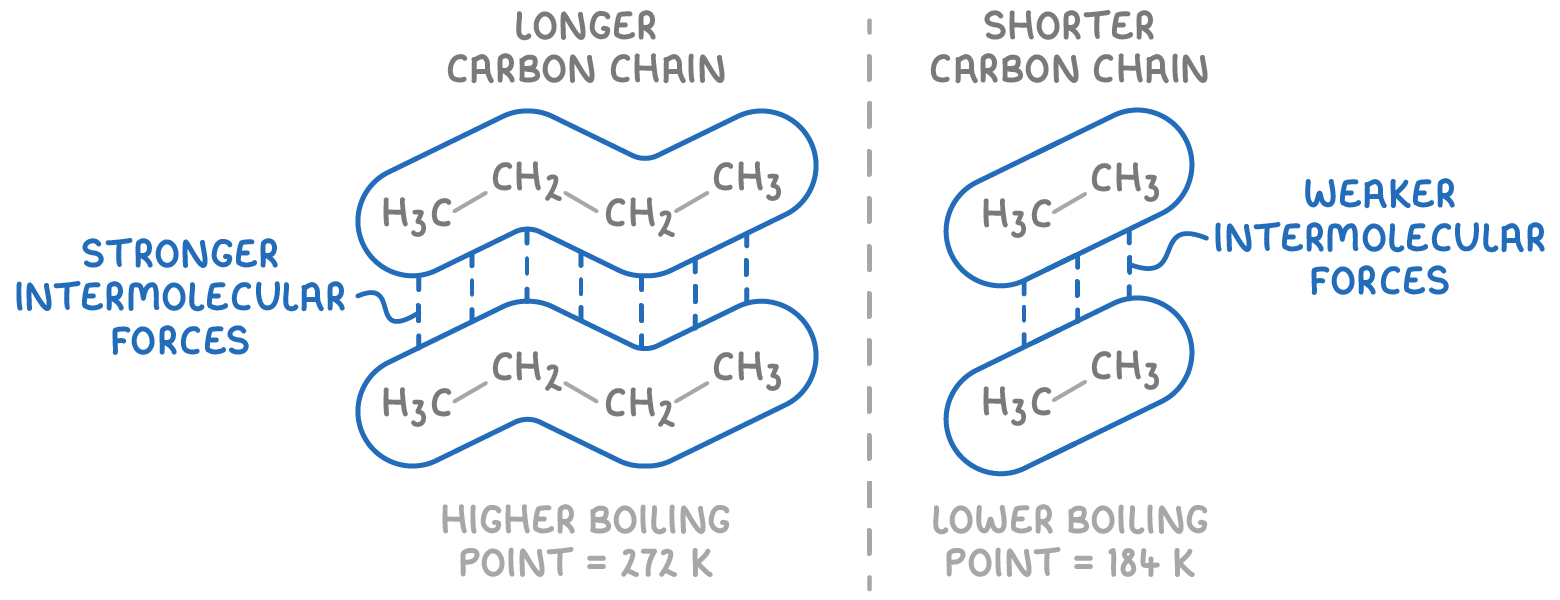
Branching
Straight chain alkanes can pack together more closely, maximising interaction between molecules.
This leads to stronger induced dipole-dipole forces in straight chain alkanes
Therefore, more energy is required to seperate these molecules