B. The particulate nature of matter
The Kinetic Theory of Matter: Matter is made up of a large number of tiny particles. The microscopic (small) behavior of the particles determines the macroscopic (large) behavior of the material.
Temperature (T) is a measurement of the average kinetic energy per particle in a material (C/K)
Internal Energy or Thermal Energy (Eint) is a measurement of the total kinetic energy of particles in a substance plus the total potential energy of the bonds between the particles (J)
Heat (Q) is the total change in thermal energy, or final thermal energy minus beginning thermal energy (J). heat gained by one is lost by the other
change in temp is the same in K and C
absolute zero = the temperature when there is no kinetic energy
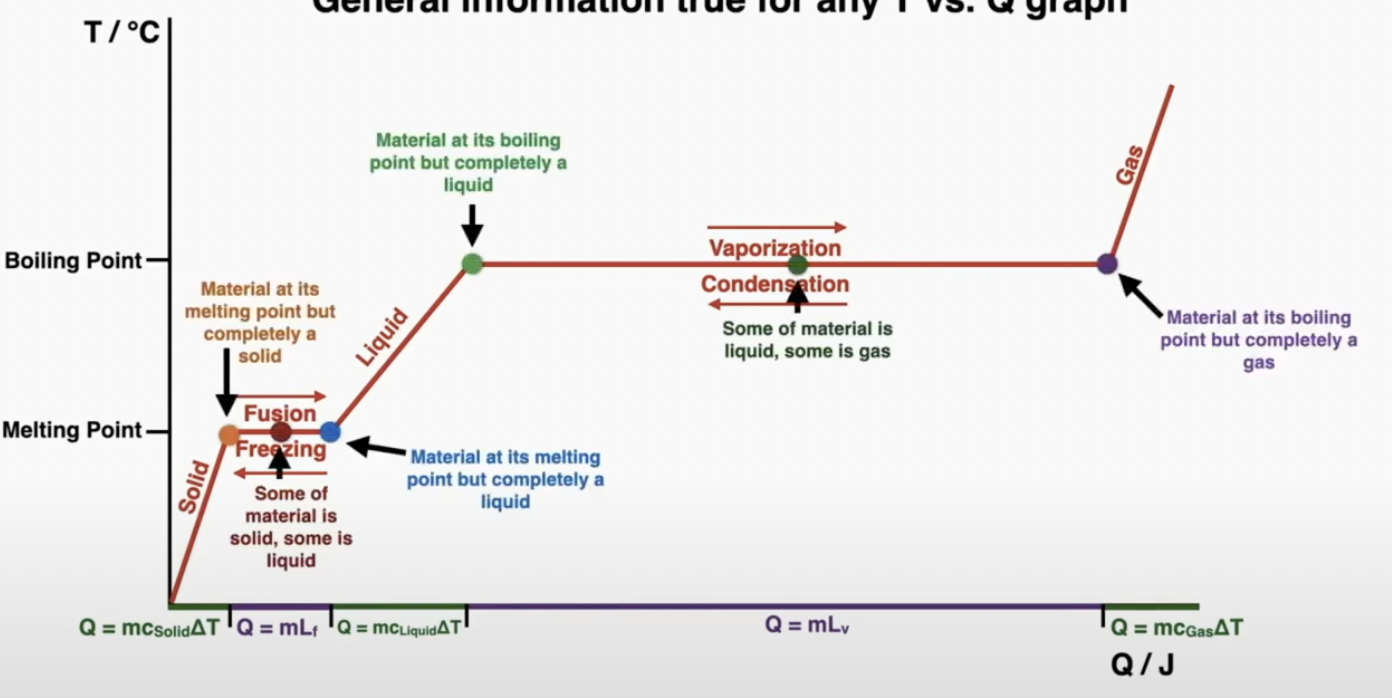
phase changes: Temperature increases
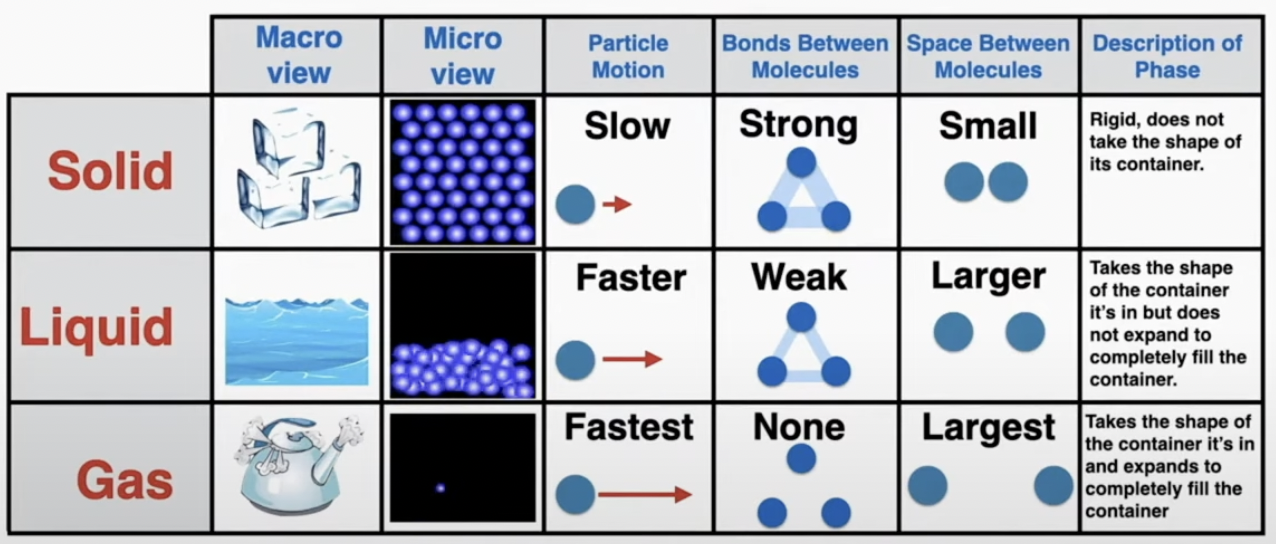
during a phase change only the potential energy changes (the energy that holds the particles together). no temperature change
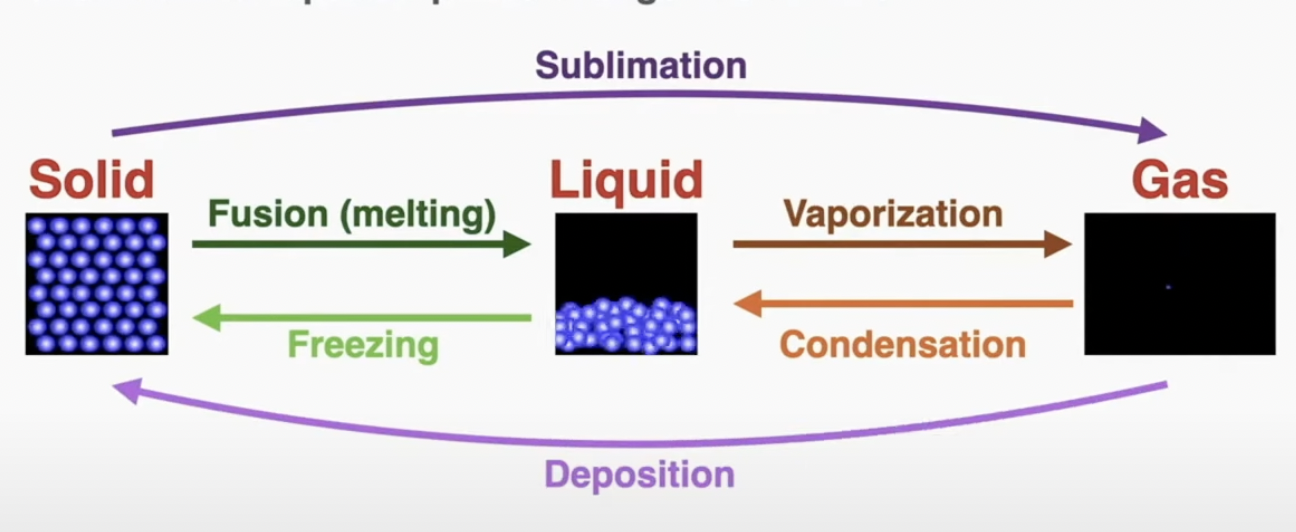
The melting point of a material is the temperature at which the phase changes from solid to liquid or vice versa.
The boiling point of a material is the temperature at which the phase changes from liquid to gas or vice versa.
When a material is not at its melting or boiling point, energy added to it changes its temperature (the kinetic energy of the particles). When a material is at its melting or boiling point, energy added to it changes its phase (the potential energy in the bonds of the particles).
specific heat is a measurement of how many joules of heat are requited to change the temperature of 1kg of mass by 1 degree
C= Q/mΔT or Q = mcΔT
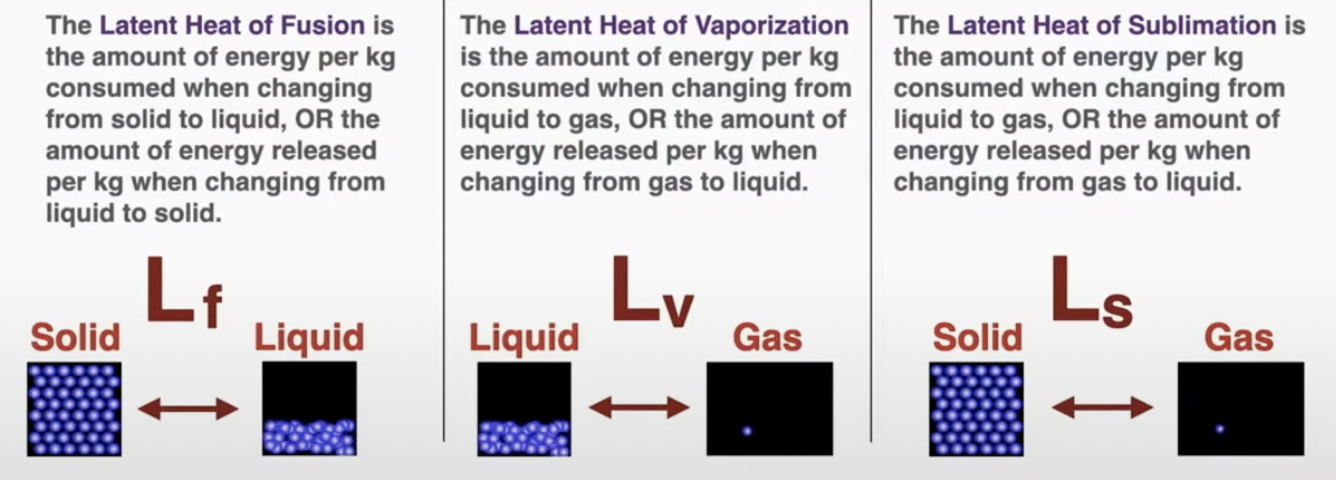
Latent Heat is a measurement of how much energy it will take to change 1 kilogram of material from one phase to another when the material is at the correct temperature to change its phase.
Q = mL
Power in heat equations:
P = Q/t so Q= mL and Q=mcΔT can be put in
Temperature heat graph: slope = 1/mc (mass stays constant)
Thermal equilibrium: Two objects are In Thermal Equilibrium if they have the same temperature
The 0th Law of Thermodynamics:
"If A and B are in thermal equilibrium, and B and C are in thermal equilibrium, A and C are also in thermal equilibrium"
Thermal contact is when two objects are usually physically touching so that heat can move between them
When two objects are in thermal contact, heat will move from the higher temperature to the lower temperature object until the two objects are in thermal equilibrium. Once they reach thermal equilibrium, heat stops moving between them.
Tfinal = Tfinal
-Q1 = +Q2
Greenhouse Gas: A gas that absorbs and emits radiant energy from the sun. We call them greenhouse gases because they store energy in the Earth's atmosphere like the roof of a greenhouse. If we significantly change the amount of greenhouse gases in the atmosphere, we will change the energy in the atmosphere. This causes climate change.
Climate Change: A change in the overall average temperature of a region or of the Earth. It is caused by a change in the greenhouse gas makeup of the atmosphere. Climate change is a threat to our current existence because it threatens to change how and where food can be grown, and where people can live.
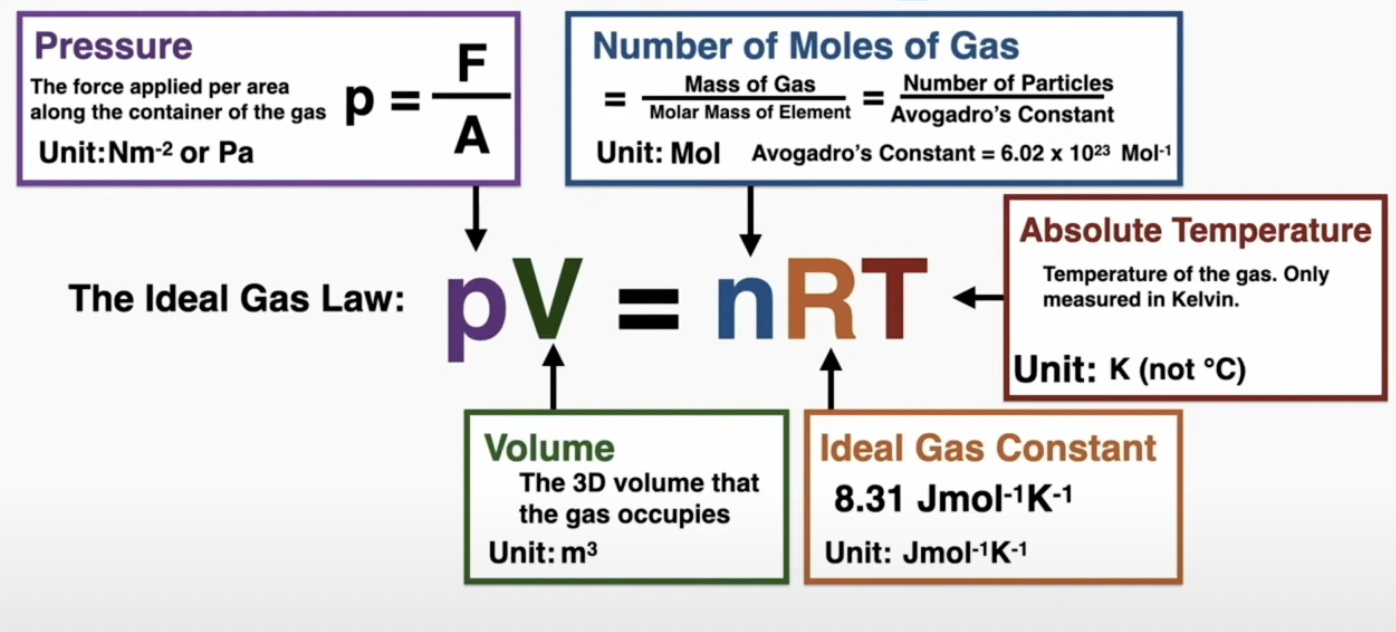
Pressure (p) is the fore applied per unit area (Pa), scalar
p = F/A
gas always flow form a highe pressure to a lower pressure region until the two pressures are equal
density ρ as given by ρ = mV
ideal gas: all internal energy added becomes kinetic energy
The gas is made up of a very large number of small identical molecules in random motion.
There can be no intermolecular forces or bonds between the particles.
All collisions between particles, and between particles and the barrier, must be perfectly elastic.
There are just as many molecules moving in one direction as any other direction (the gas as a whole doesn't travel in any one direction)
The individual forces from collisions between molecules average out to a uniform pressure throughout the gas.
more kinetic = more like ideal]
less potential = more like ideal
lower pressure (less interactions = less intermolecular forces) = more like ideal
higher temps (more kinetic) = more like ideal
lower density (less interactions) = more like ideal
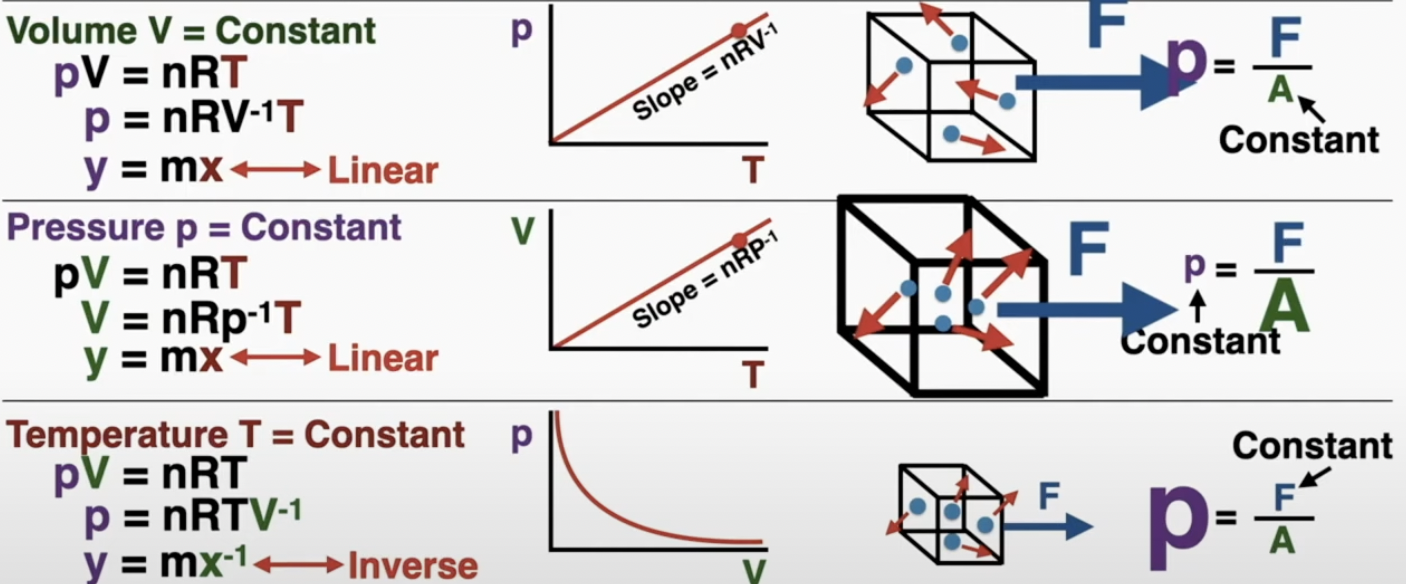
ideal gas law: pV = nRT
pressure comes from the impulse of the particles on the container
volume (sphere: 4/3π(r)³, cylinder: h2π(r)²)
n = N/NA

total kinetic energy = total internal energy
Ek = Eint/N

finding the velocity of the particles use m = molar mass/avogadro’s constant → into kg *10^-3 and then put into Ek = ½ mv² where Ek = 3/2KbT
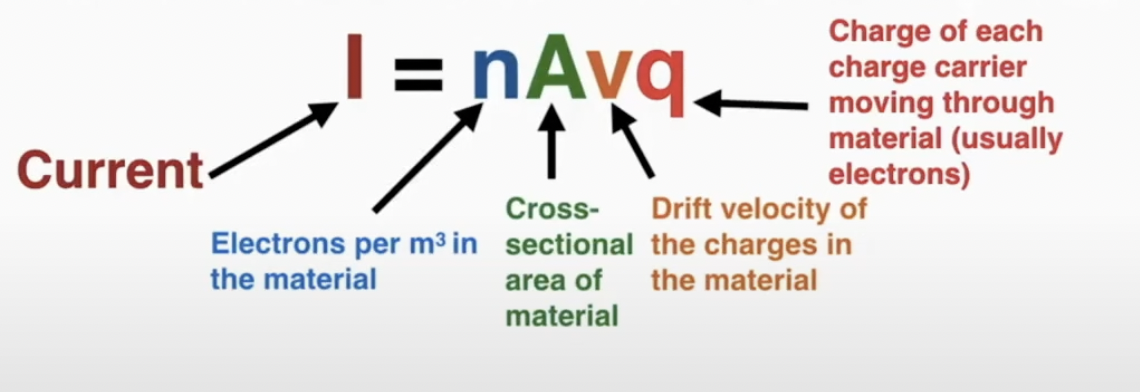
Current (I) is a measurement of the amount of charge passing by a point per unit of time, (A) scalar
I = q/t
Voltage (V) is a measurement of the electrical potential energy per unit charge between two points in an electric field. It is a scalar. Voltage tells you how much energy will be added to a charge as it moves a given distance in an electric field based on the amount of charge. (V)
V = Work/q
V = Ed

electon-volt is a unit of energy (eV)
#of electron volts = (# of electrons) * voltage
1 eV = 1.6×10^-19 J
Resistance (R) is the amount of opposition current will experience to its flow when moving through an object. It can be thought of as the amount of electrical energy per charge (voltage) lost per current moving through the material. (Ω)
R = V/I
Resistivity (ρ) is the amount of resistance a given cross-sectional area of a material will experience per unit of length of the material. (Ωm)
ρ = RA/L
a circuit is a path along which an electrical current can be carried
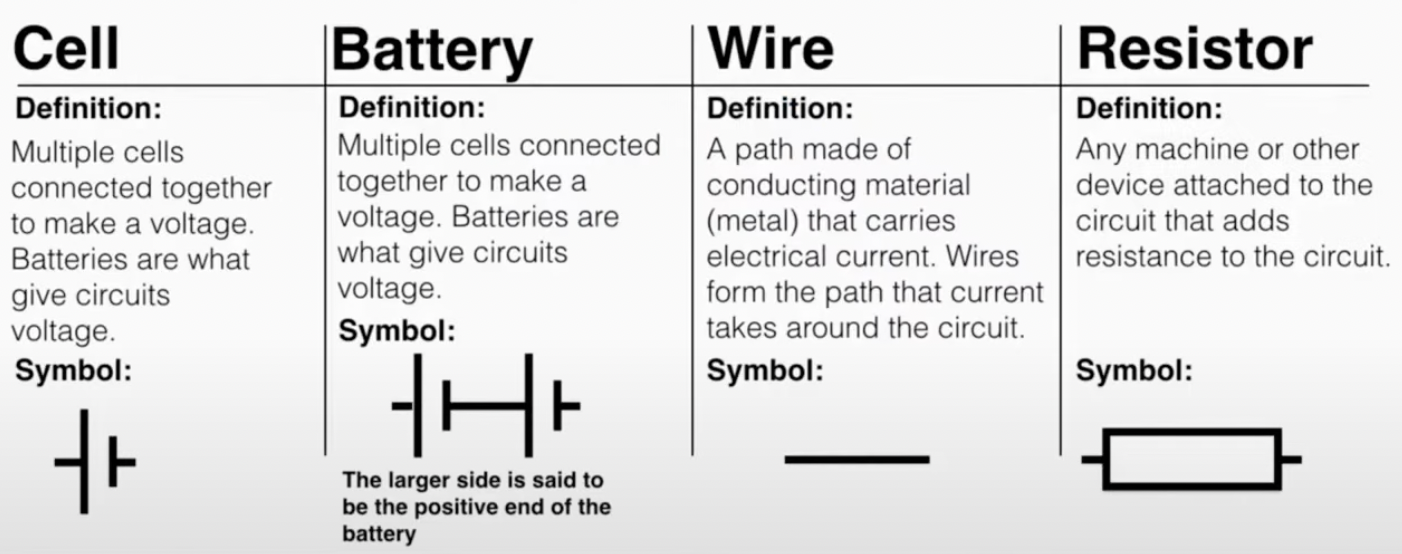
battery pushes the electrons already in the circuit around. the battery provides a voltage that adds energy to the electrons which allows them to push the electrons in front of them (as they are repulsed by each other). when the electrons first leave the battery they have a lot of electrical potential but when they have completed the circuit they have no more.
closed circuits: a path from one end of the battery back to the other end. if the path is broken the electrons stop working
the direction of conventional current is the opposite of the direction of the electrons (from the positive to the negative end)
if there is more than one path back to the battery the current will split adn take both paths. if the path does not lead back to the battery no current will flow into that split because the electrons cannot be pushed through the resistance in the open part of the path.
Circuits always include at least one resistor. In a resistor, the electrons lose some of the energy they are gaining from the battery in the process of "pushing through" the resistance. The total voltage lost by the current in the circuit is equal to the voltage added by the battery.
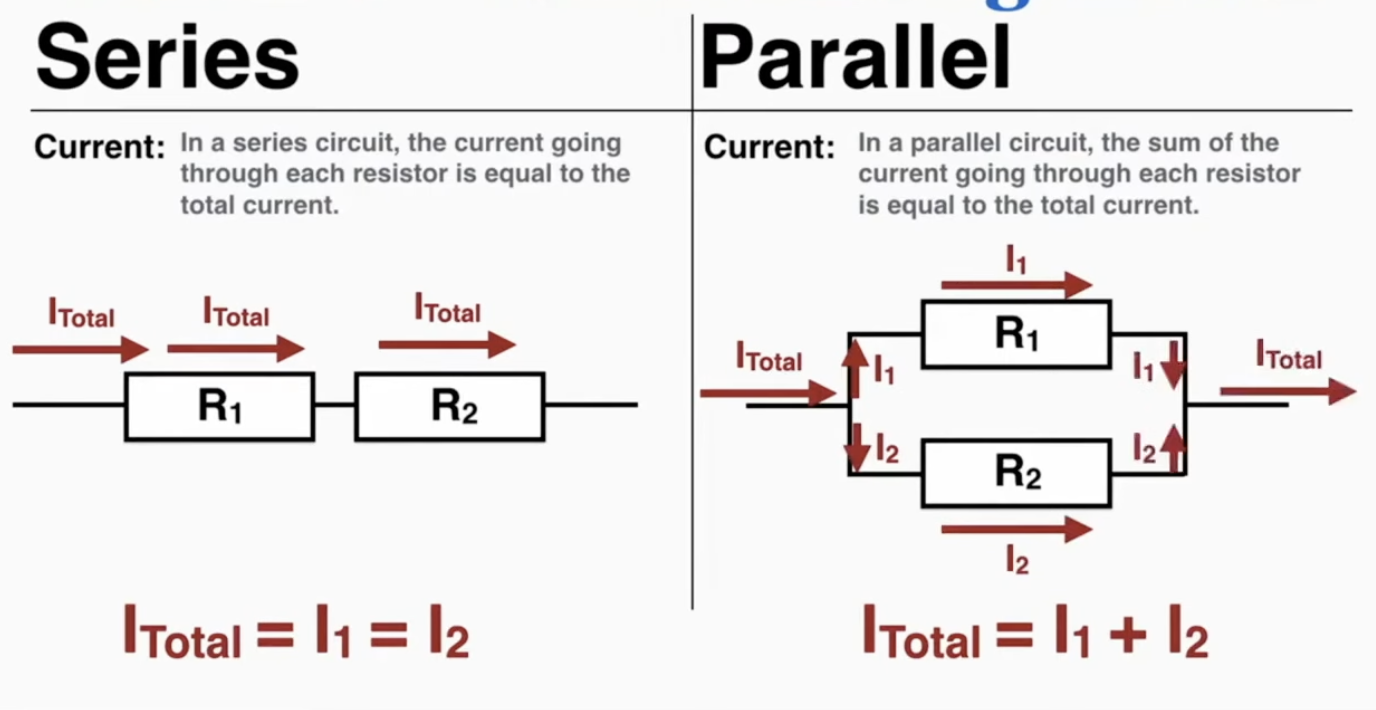
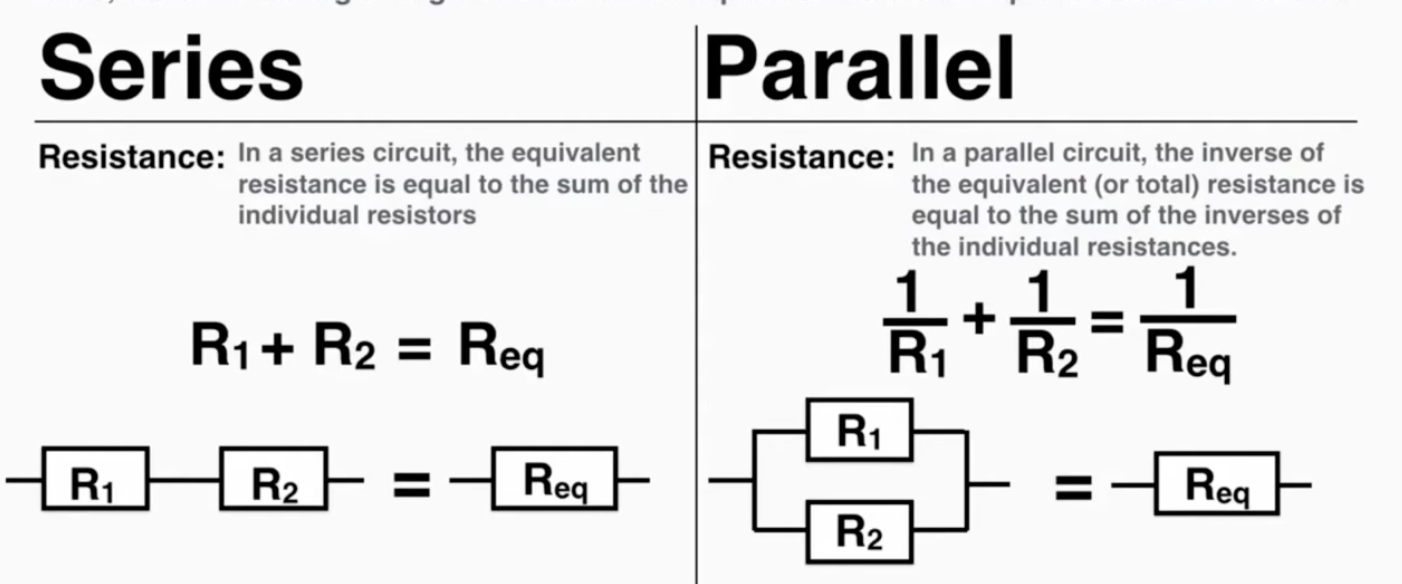
Complex Circuits: A complex circuit is a circuit with a combination of series and parallel arrangements. To simplify a complex circuit, find arrangements inside of it that are simple series or parallel circuits, simplify them to a single resistor, and repeat until the entire circuit is one resistor.
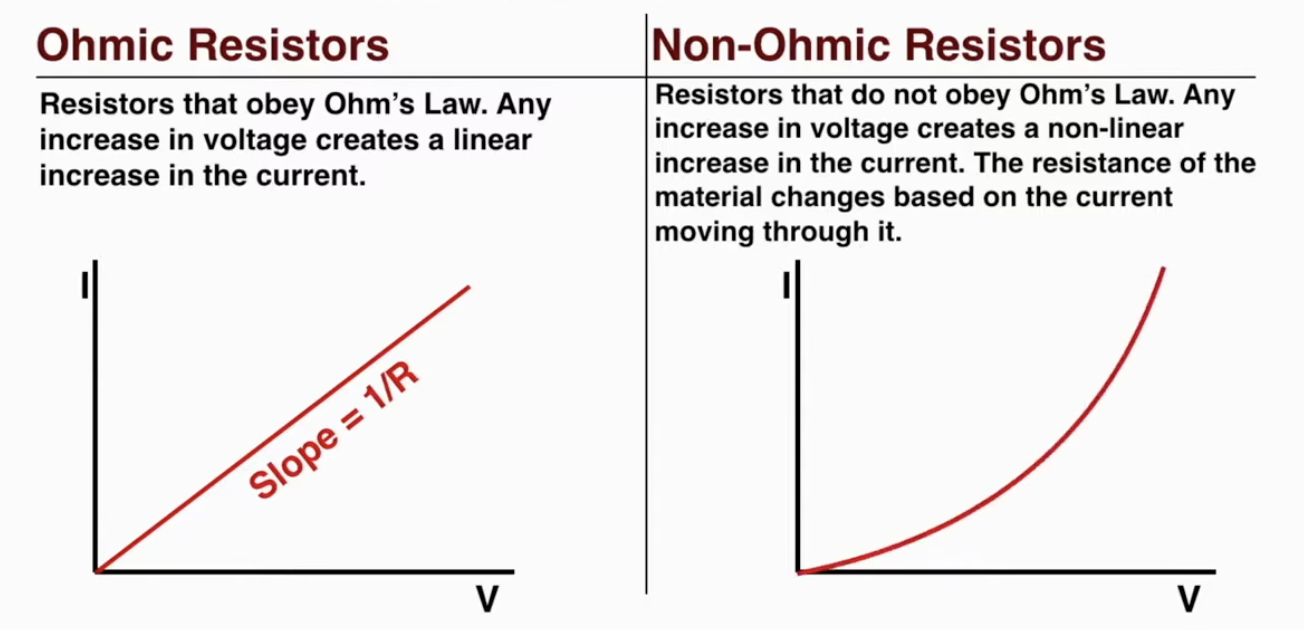
Ohm’s law : V = IR
Ohmic resistors: obeys ohm’s law
non-ohmic resistors: do not obey ohm’s law

Power in circuits:
P = IV = I²R = V²/R
Internal resistance (r) refers to the resistance inside of a battery. No battery is
ideal and all contain at least a little resistance. We call the voltage that the battery delivers to the rest of the circuit (excluding the voltage drop in the internal resistance) the terminal voltage.
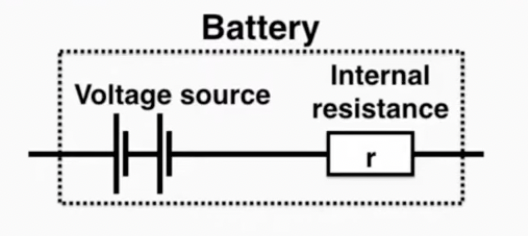
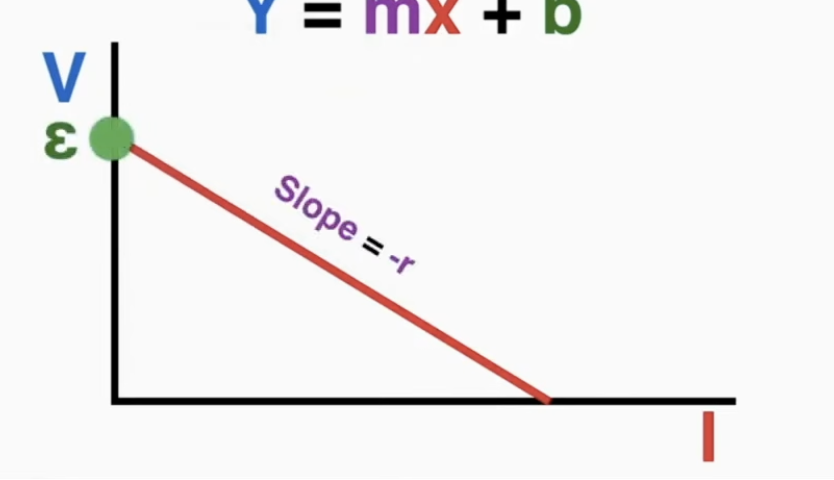
voltage source: electromotive force/EMF (ε) is the voltage causing elecyrons to move
ε = Ir + V = I(R + r)
the voltage of the battery usually refers to the terminal voltage (actual volateg coming out of the battery)
voltage current graph: