Folliculogenesis
Learning Objectives
Understand the time span of follicle and oocyte development
Describe the structure of the follicle and the function of each cellular layer
Explain the selection of the dominant follicle that is destined to be ovulated and the mechanisms involved in chromosome number reduction
Discuss differing steroid production from both the granulosa and theca cells of the follicle
Time Span of Follicle and Oocyte Development
Requirements for Reproduction:
Differentiation into male or female
Sexual maturation
Production, storage and release of a sufficient supply of eggs and sperm and the correct number of chromosomes in eggs and sperm
Egg and sperm have to meet
Creation of a new individual with genes from both parents
To nurture the individual until capable of an “independent life”
Prenatal Development:
Primordial Germ Cells (PGCs) are germ cells entering the gonad and that will differentiate eggs or sperm
They increase in number during mitosis and are identifiable in the yolk sac of a developing fetus about 3 weeks after conception:
They migrate to the genital ridge and differentiate into oogonia, which multiply via mitosis during fetal life.
Once the PGCs enter the ovary and become oocytes. The germ cells become the oogonia when they are in the ovary and oogonia are egg precursors and multiply by mitosis
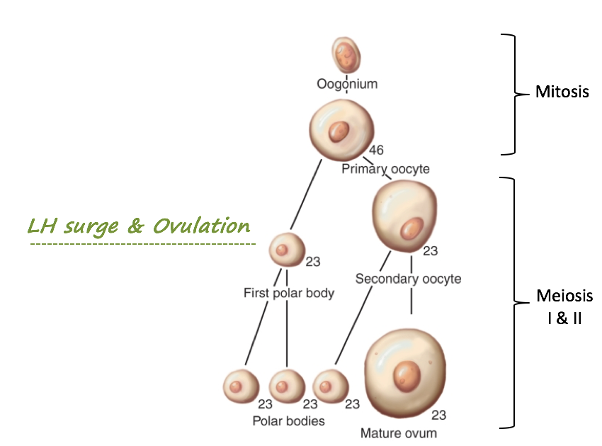
Once mitosis stops, they enter meiosis and are known as primary oocytes.
By 20 weeks of gestation, all oogonia enter meiosis I, becoming primary oocytes and arresting in prophase I.
These primary oocytes remain in meiotic arrest until ovulation, a process that could span decades.
All the eggs a woman will ever have are made at this stage so the mitotic divisions are critical and this determines the level of fertility
Massive loss of oocytes occurs before birth due to atresia, leaving about 1–2 million at birth.
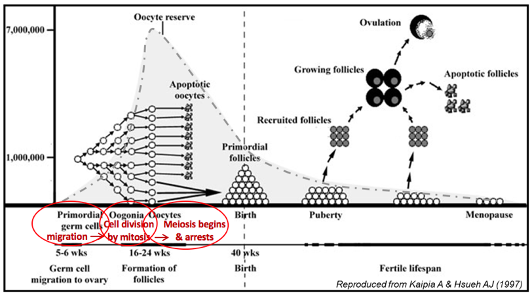
This graph shows that there is a large increase in egg number by mitosis and reached a peak in the womb
After birth, there is. massive loss in eggs and in menopause there is. alow egg number and can’t have children after this point - reproductive lifespan
Postnatal Development:
After puberty, a small cohort of follicles begins to grow each day from the pool of resting follicles in a process called folliculogenesis.
It takes ~3 months for a follicle to progress from a primordial follicle to a dominant follicle ready for ovulation.
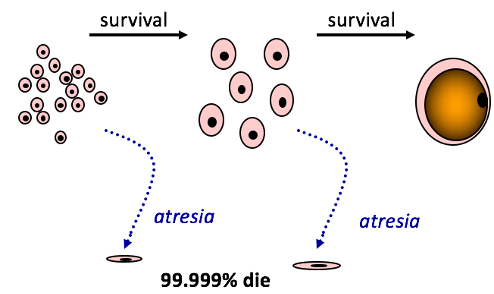
Approximately 400–600 oocytes are ovulated over a lifetime, with the remainder undergoing atresia.
Resting follicles that will go on to grow, are only a small cohort (of about 40 or 50) that initiate growth and you have initiation (A cohort of early follicle leave the resting pool and grow continuously) and recruitment (They will NOT continue to grow unless they reach the size at which they respond to changes in FSH that occur in the menstrual cycle) out of the resting pool.
They then reach a certain size and stage, needing FSH to keep the pool going and progress to keep growth occurring.
You need the FSH rise in the menstrual cycle to continue growth, and so therefore, follicles at the right stage are recruited into the menstrual cycle, which are a small number of of follicles.
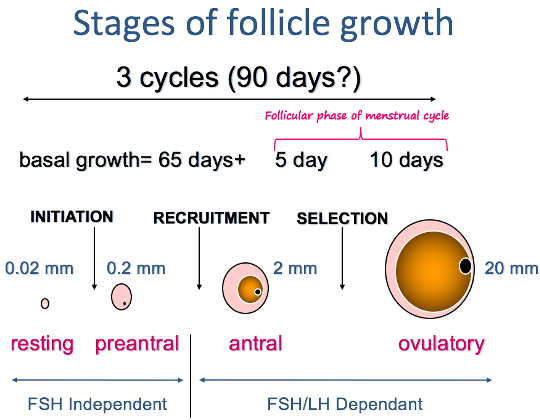
This process and the stages of follicular growth is very slow (3 months) and the growth is continuous.
You don’t need FSH for this early growth, but after the antral stage, you have FSH and LH dependent selection.
You need the FSH for the theca cells to keep growing and from this smaller cohort, about 5-10 are selected for ovulation and this is the follicular phase of the menstrual cycle.
This is dependant on the gonadotrophins FSH and LH.
Thousands of the primordial follicles are made but most die through atresia and only a few make it into the menstrual cycle.
There is stress released from what happens before, not just what happens now. Around 400-600 eggs are ovulated and the majority of the eggs die off.
This shows a HNE stain of the antral ovarian follicle. This is a standard concept used where you take the tissue and fix it, then dehydrate it through a series of alcohol washes to remove the fluid. Then you put wax on it to imbed it and stain it with various dyes. You can then see cellular structures of things that you couldn’t see under the microscope.
Structure of the Follicle and Function of Each Cellular Layer
Folliculogenesis: growth and development of follicles from the earliest “resting” stages as laid down in the foetus through to ovulation.
Most follicles in the ovary aren’t growing (after puberty - a few grow each day)
As follicles start to grow, the granulosa cell multiplies and the oocyte secretes another protective acellular layer (zona pellucida) which stays attached after ovulation
Once the growth of the follicles starts, a second layers of cells differentiate around the basal lamina known as the theca
As the oocyte gets bigger, they secrete the zona pellucida protein layer, which arranges to the cell. The multiplication of the granulosa cell (originally formed in the fetus) in puberty, you get another layer of cells called the theca cells that form around the granulosa cells and these sustain growth around the follicle. There are steroids produced from both the granulosa and theca cells.
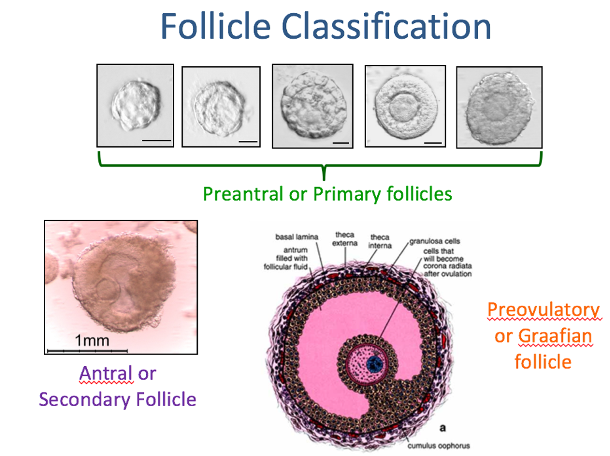
Stages of Follicular Development:
Primordial Follicle:
Structure: Primary oocyte surrounded by a single layer of flat granulosa cells and a basal lamina.
Function: Dormant stage, waiting for initiation of growth.
Primary Follicle:
Structure: Oocyte enlarges; granulosa cells become cuboidal and start to proliferate.
Function: Early development begins, independent of gonadotropins.
Secondary (Antral) Follicle:
Structure: Multiple layers of granulosa cells, theca cells form around the basal lamina, and fluid-filled spaces (antrum) appear.
Function: Theca cells provide androgens; granulosa cells convert androgens to estrogens, supporting follicular growth.
Graafian (Dominant) Follicle:
Structure: Enlarged follicle with a large antrum; oocyte surrounded by cumulus granulosa cells.
Function: Prepares for ovulation, driven by FSH and LH.
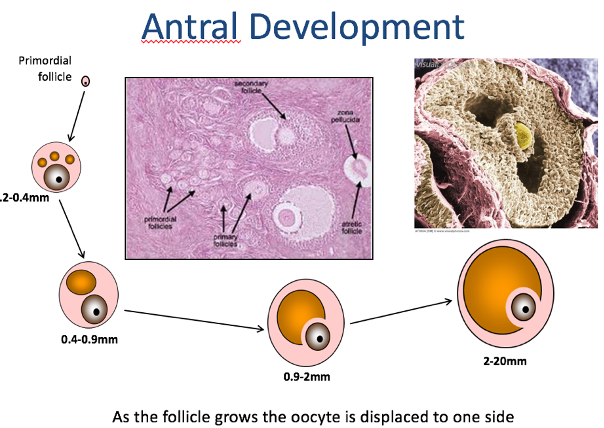
Follicular Growth:
Not hormone controlled; FSH drives most of folliculogenesis but early growth is independent of FSH and is driven by local factors
FSH-deficient patients or those with. amputation of FSHr can still undergo early growth of folliculogenesis
As the follicle starts to grow it increases rapidly in diameter and granulosa cells divisions increase. The granulosa cells are vascular and have no blood supply.
Gaps begin to form in the granulosa cell layers due to theca cell formation. These gaps consist of fluid-filled spaces which form an antrum & are filled with follicular fluid.
Once the theca cells forms, the vasculature does too with hormones that can influence it.
The fluid is then filtered out of the blood and fills the space in the cavity and form antrum follicles.
Cellular Layers of the Follicle:
Oocyte:
Function: Carries the genetic material for fertilization. Arrests in meiosis I until ovulation. Produces factors that regulate surrounding granulosa cells.
Granulosa Cells:
Function:
Early stages: Provide nutrients to the oocyte and secrete protective zona pellucida.
Later stages: Convert androgens (from theca cells) to estrogens using the enzyme aromatase.
Lacks blood supply; relies on diffusion of nutrients and hormones.
Theca Cells:
Layers:
Theca Interna: Produces androgens under LH stimulation.
Theca Externa: Provides structural support.
Function: Supplies granulosa cells with androgen precursors for estrogen production.
Antrum:
Function: Stores follicular fluid rich in growth factors, hormones, and nutrients, which regulate follicle maturation.
Selection of the Dominant Follicle and Chromosome Reduction Mechanism
Dominant Follicle Selection:
Mechanism:
A cohort of antral follicles begins to grow in response to FSH.
The dominant follicle emerges as the one most sensitive to FSH, requiring lower levels to sustain growth.
Produces high levels of estradiol, which exerts negative feedback on FSH, suppressing the growth of other follicles.
Outcome:
Dominant follicle develops LH receptors, preparing it for the LH surge that triggers ovulation.
The remaining follicles undergo atresia.
In order for the follicle to survive, the theca brings out the blood supply and it enables the follicles to keep growing. You have the corpus luteum is formed from the follicle that has released its egg and is about to collapse.
The vasculature of the theca is important to the follicle as it brings with it growth factors and hormones. It is needed in order for the follicle to grow.

Chromosome Reduction (Meiosis):
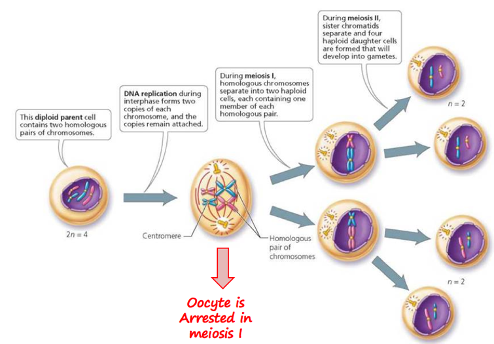
In the fetal ovary, the oocyte is arrested and the oogonia forms by mitosis and enters meiosis 1. They line up on the spindle and arrest and remain until the LH surge which isn’t until late in the menstrual cycle.
When the chromosomes are being replicated during the S phase, the chromosome copies remain attached.
The two copies, each an exact replicate of the original chromosome, stay attached at a region called the centromere.
As long as the replicate copies remain attached, each copy is called a chromatid.
The two attached chromatids are genetically identical and are called sister chromatids.
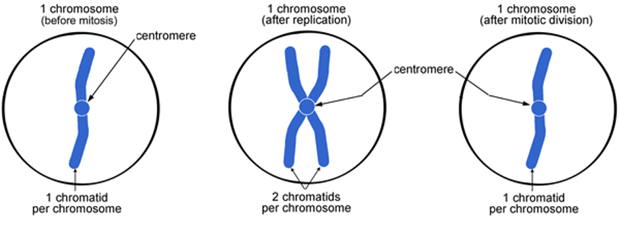
Chromosomes replicate during S-phase of cell cycle
Remain attached at the centromere
Each copy known as a chromatid → the 2 copies are identical to each other → “sister” chromatids
Exact copy of original chromosomes
You have 2 identical daughter cells as a result of this.
Mitosis has 4 stages, prophase, metaphase, anaphase and telophase.
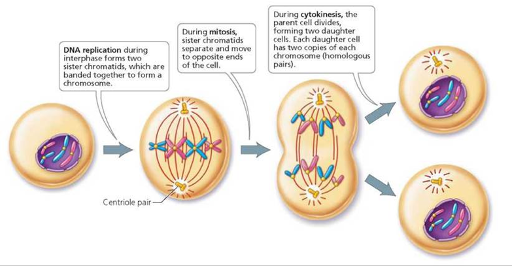
Meiosis:
Meiosis and mitosis begin the same way.
Both are preceded by the same event—the replication of chromosomes.
Unlike mitosis, however, meiosis involves two divisions.
In the first division, the chromosome number is reduced, because the two homologues of each pair of chromosomes (each replicated into two chromatids attached by a centromere) are separated into two cells so that each cell has one member of each homologous pair of chromosomes.
In the second division, the replicated chromatids of each chromosome are separated. We see, then, that meiosis begins with one diploid cell and, two divisions later, produces four haploid cells.
The orderly movements of chromosomes during meiosis ensure that each haploid gamete produced contains one member of each homologous pair of chromosomes.
The chromosomes all line up adjacent to each other and crossing over occurs here which leads to biological variation. Duplicated chromosomes then pull apart. The diploid has 1 and haploid, in separation has 2. You then produce 23 single chromosomes per cell.
Steroid Production by Granulosa and Theca Cells
Theca Cells:
Steroid Produced: Androgens (e.g., androstenedione and testosterone).
Mechanism:
Cholesterol is converted to androgens via the delta-4 pathway using enzymes such as CYP17.
Stimulated by LH.
Granulosa Cells:
Steroid Produced: Estrogens (e.g., estradiol).
Mechanism:
Aromatase converts theca-derived androgens to estrogens under FSH stimulation.
Granulosa cells lack CYP17, so they depend on theca cells for androgen precursors.
Two-Cell, Two-Gonadotropin Model:
Theca Cells: LH stimulates androgen production.
Granulosa Cells: FSH drives aromatization of androgens to estrogens.
According to the two-cell–two-gonadotropin theory, luteinizing hormone stimulates theca cells to produce androgens, and follicle-stimulating hormone stimulates granulosa cells to produce oestrogens from androgens. The ovarian follicle produces steroids.
The granulosa and theca cells produce different steroids as they are regulated differently and a different load of androgens are produced form the theca and these are produced from cholesterol. The theca cells form the granulosa cells and ten the oestrogen drives the granulosa cells and the goes into the circulation done by FSH binding to it’s receptor. Androgen formation is driven by LH and a few others things.
Aromatase is increased in production with oestrogen and it is only present in granulosa cells, none are in the theca. Therefore the androgens are only made in the theca and you need both the cells to be present to drive granulosa formation.
The starting point for steroid biosynthesis is the conversion of cholesterol in pregnenolone by P450scc. One route for pregnenolone metabolism is the delta-5 pathway (red arrows) through CYP17 (P450c17). Pregnenolone hydroxylation at the C17a position forms 17-hydroxypregnenolone, and subsequent removal of the acetyl group forms the androgen precursor dehydroepiandrosterone (DHEA).
An additional route for pregnenolone metabolism is the delta-4 pathway (purple arrows), in which pregnenolone is converted to progesterone by 3b-HSD (an irreversible conversion). Progesterone is then converted to 17-hydroxyprogesterone by CYP17. In humans, 17-hydroxyprogesterone cannot be further metabolized.
Importantly, CYP17 is exclusively located in thecal and interstitial cells in the ovary extrafollicular compartment, whereas CYP19 (aromatase), which converts androgens to estrogens, is expressed exclusively in GCs, which are in the intrafollicular compartment. Androgen aromatization to estrogens is a distinct activity that occurs in the granulosa layer, and it is induced by FSH via P450 aromatase (P450arom) gene activation.
There are various enzymes that convert the products into different things, so for example, you can go from cholesterol to progesterone. For all the antral follicles, the boxed section above only occurs in the dominant follicle after the LH receptors become present, as these are not a part of regular steroidogenesis.
There is steroid feedback to the hypothalamus and the follicle that is destined to become the final releasing follicle would have started its growth a few cycles before.