Chapter 9
Lattice Energy: The energy released when oppositely charged ions come together OR the energy absorbed to break apart an ionic compound into its ions
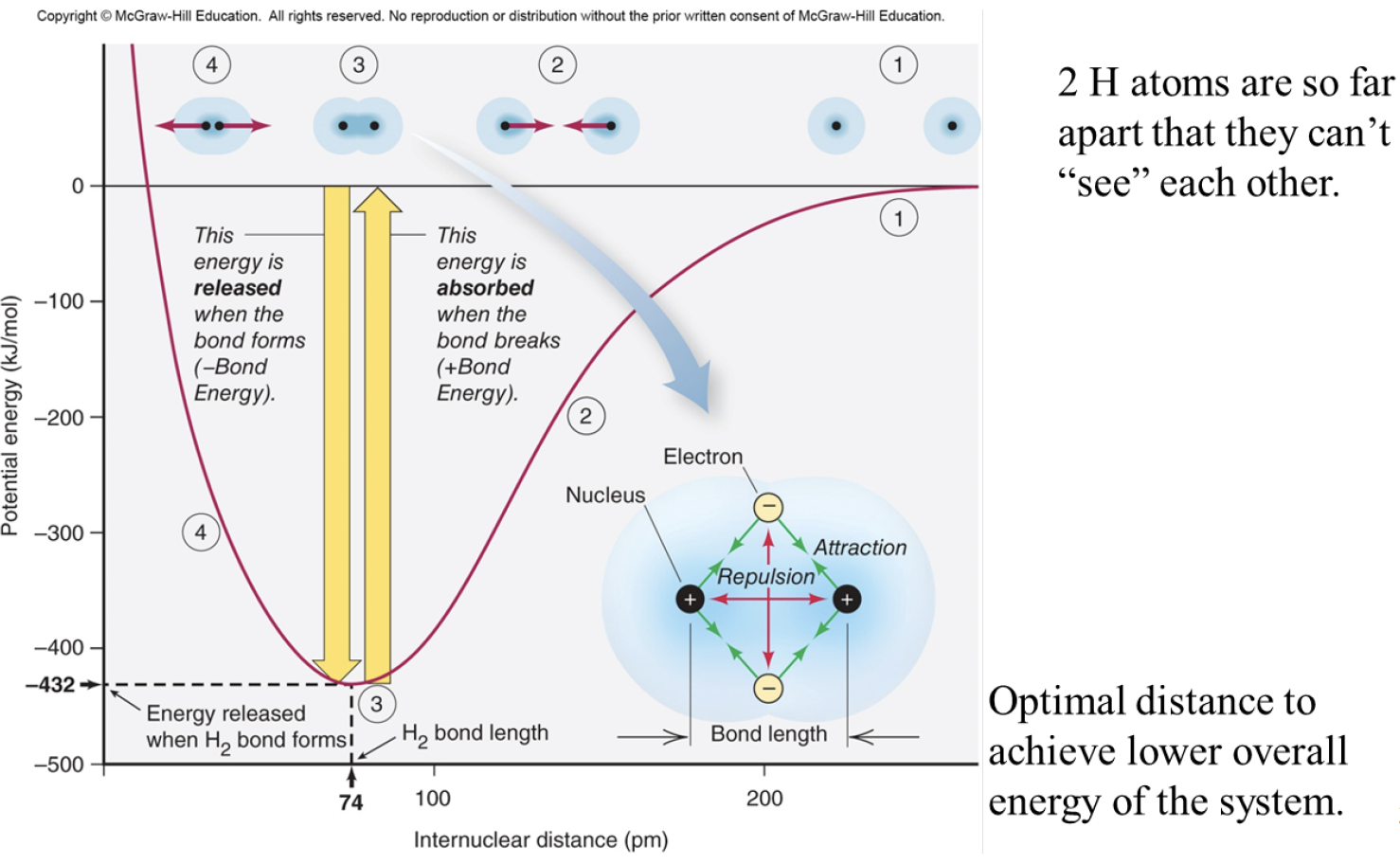
Bond length: the distance between the two
Bond Energy: energy needed to break a bond between atoms
Optimal distance to achieve lower overall energy of the system
Properties of Covalent Bonds
Bond length: distance btwn nuclei of the bonded atoms
Bond energy: energy needed to overcome the attraction between the nuclei and the shared electrons—energy required to break a bond. The stronger the bond, the higher the bond energy.
Bond order: the number of electron pairs being shared by a given pair of electrons