Cycles of Matter
Cycles of Matter Overview
Water's Role in Biogeochemical Cycles
Water is a universal solvent due to its chemical and physical properties including hydrogen bonding.
Essential for transporting nutrients and participating in the cycling of matter in ecosystems.
Importance of Nutrients in Plants
All living organisms, including plants, require matter to build cellular structures and generate energy.
Key nutrients for plants include sulfur, nitrogen, and phosphorus, which are essential for growth and energy metabolism.
Elements Transfer in the Environment
Elements like Oxygen (O), Carbon (C), Nitrogen (N), and Sulfur (S) can be easily transferred globally.
Elements such as Iron or Phosphorus are found in soil but not typically in the atmosphere, affecting their availability.
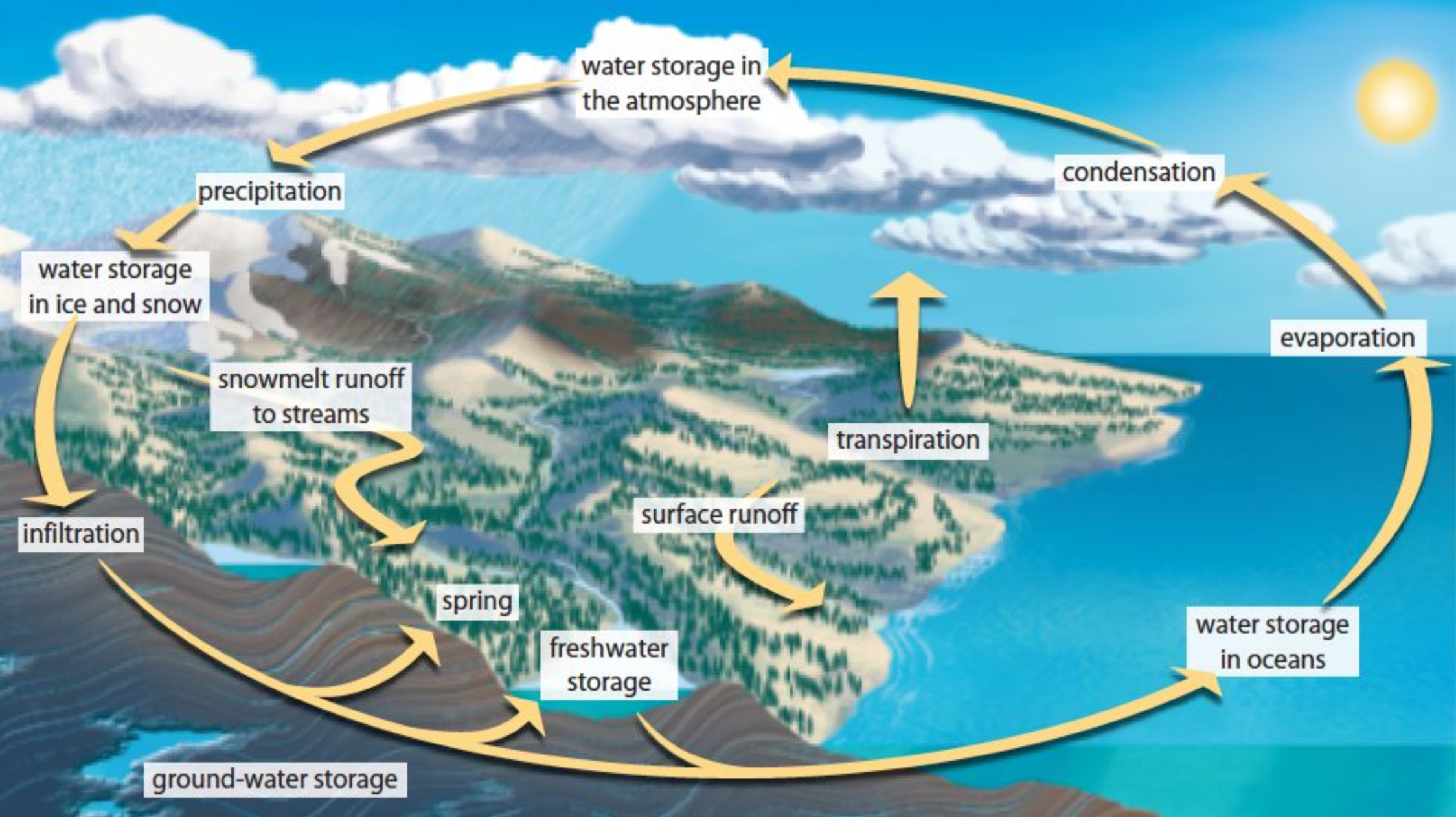
Hydrologic Cycle
Water in Different States:
About 97% of Earth's water is liquid; 2.1% is frozen as ice; and significant amounts vaporize from oceans (86%).
Water’s ability to exist in different states plays a crucial role in climate and weather patterns.
Properties of Water
Bonds in Water:
Water consists of a polar molecule formed by covalent bonds between hydrogen and oxygen, resulting in hydrogen bonding between water molecules.
Specific Heat Capacity:
Water requires a high amount of energy to change states (high specific heat), stabilizing temperatures in environments.
Density, Cohesion, and Adhesion
Density: Cold water is less dense due to loosened hydrogen bonds, allowing ice to float.
Cohesion and Adhesion: Water molecules attract each other (cohesion) and to other substances (adhesion), facilitating processes like transpiration in plants.
Water’s Function in Life
Clean freshwater is essential for all life, making up ~70% of the adult human body and ~95% of plant tissues.
Metabolism relies on water balance, including consumption, retention, and loss.
Human Impact on Cycles
Increased CO2 emissions from human activities lead to a buildup of greenhouse gases, impacting ecosystems.
This vicious cycle results in reduced transpiration by plants, consequently affecting water availability.
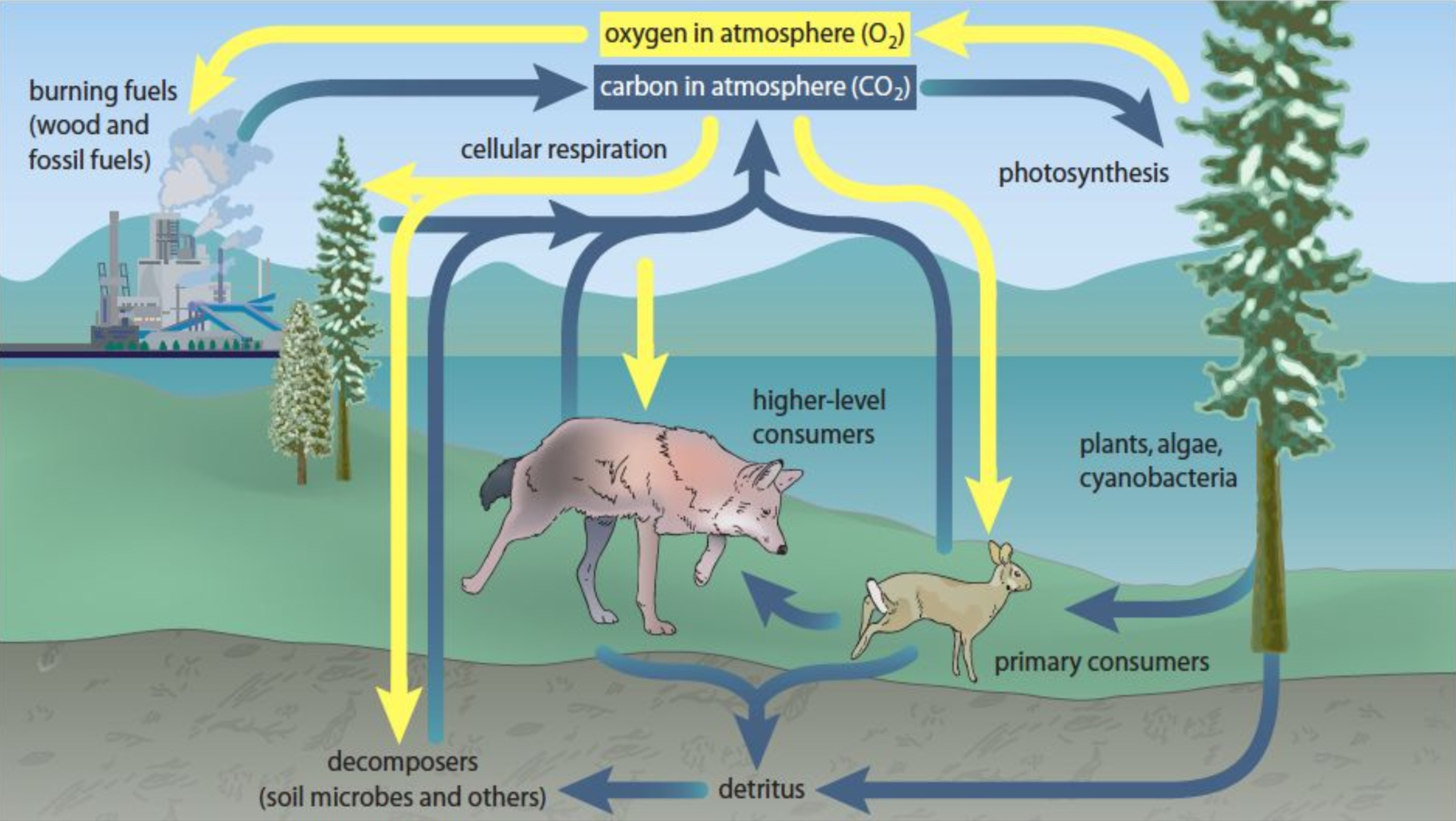
Carbon and Oxygen Cycles
Nutrients cycle through reservoirs (soil, air, water, organisms) and can be categorized into rapid (biological processes) and slow cycling (long-term storage like fossil fuels).
Rapid cycling involves processes like photosynthesis, respiration, and decomposition, while the slow carbon cycle incorporates long-term sequestration in geological formations.
Carbon Sinks vs. Sources
Carbon Sinks: Absorb more carbon than they emit (e.g., forests).
Carbon Sources: Emit more carbon than absorbed (e.g., fossil fuel combustion).
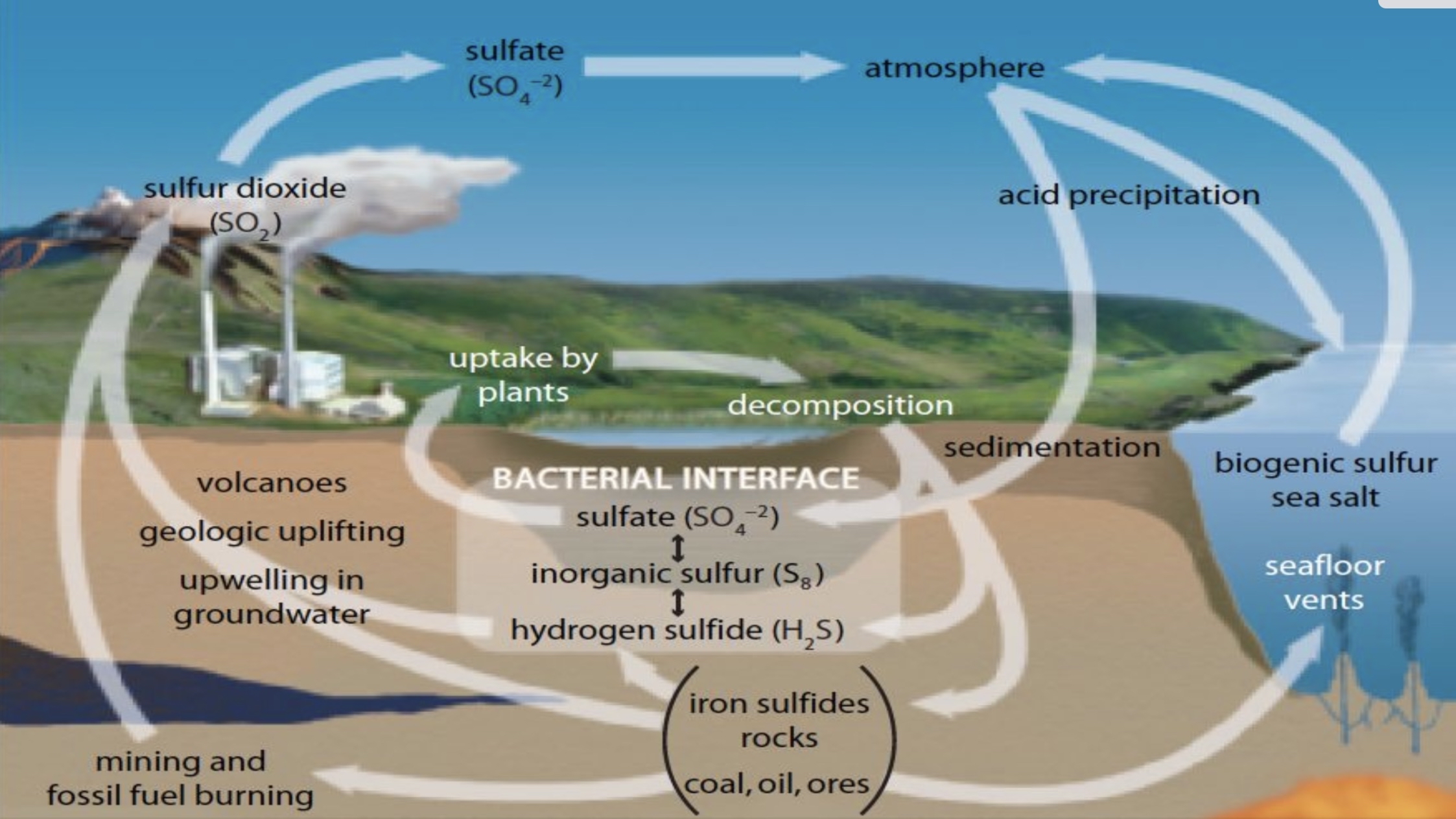
Sulfur Cycle
Sulfur from rocks and fossil fuels is released into the atmosphere through volcanic activity and human actions (e.g., burning fossil fuels).
Acid precipitation can harm ecosystems, demonstrating the impact of elevated sulfur levels from anthropogenic sources.
Acid precipitation (harms ecosystems)
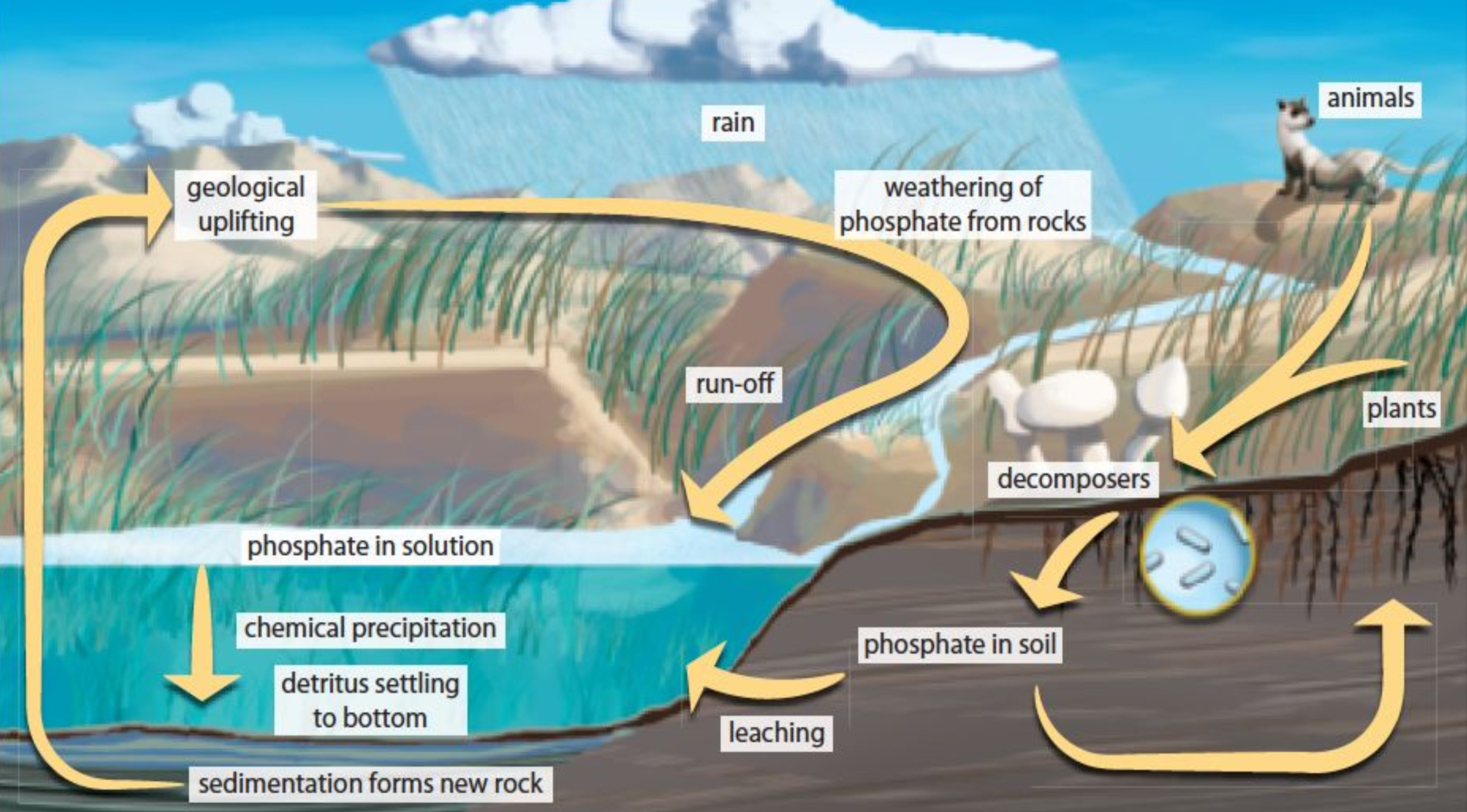
Phosphorus Cycle
Phosphorus, crucial for living organisms, mainly cycles through soil and water and is not found in the atmosphere.
Limited in supply, phosphorus must be transferred from rocks through weathering and often is applied as fertilizer in agriculture.
Not in the atmosphere (found in rocks in bodies of water)
Found in DNA and ATP (energy)
Algal Blooms
Excessive phosphorus can lead to algal blooms, which disrupt aquatic ecosystems by depleting oxygen and harming aquatic life. (Promote algae growth taking up oxygen)
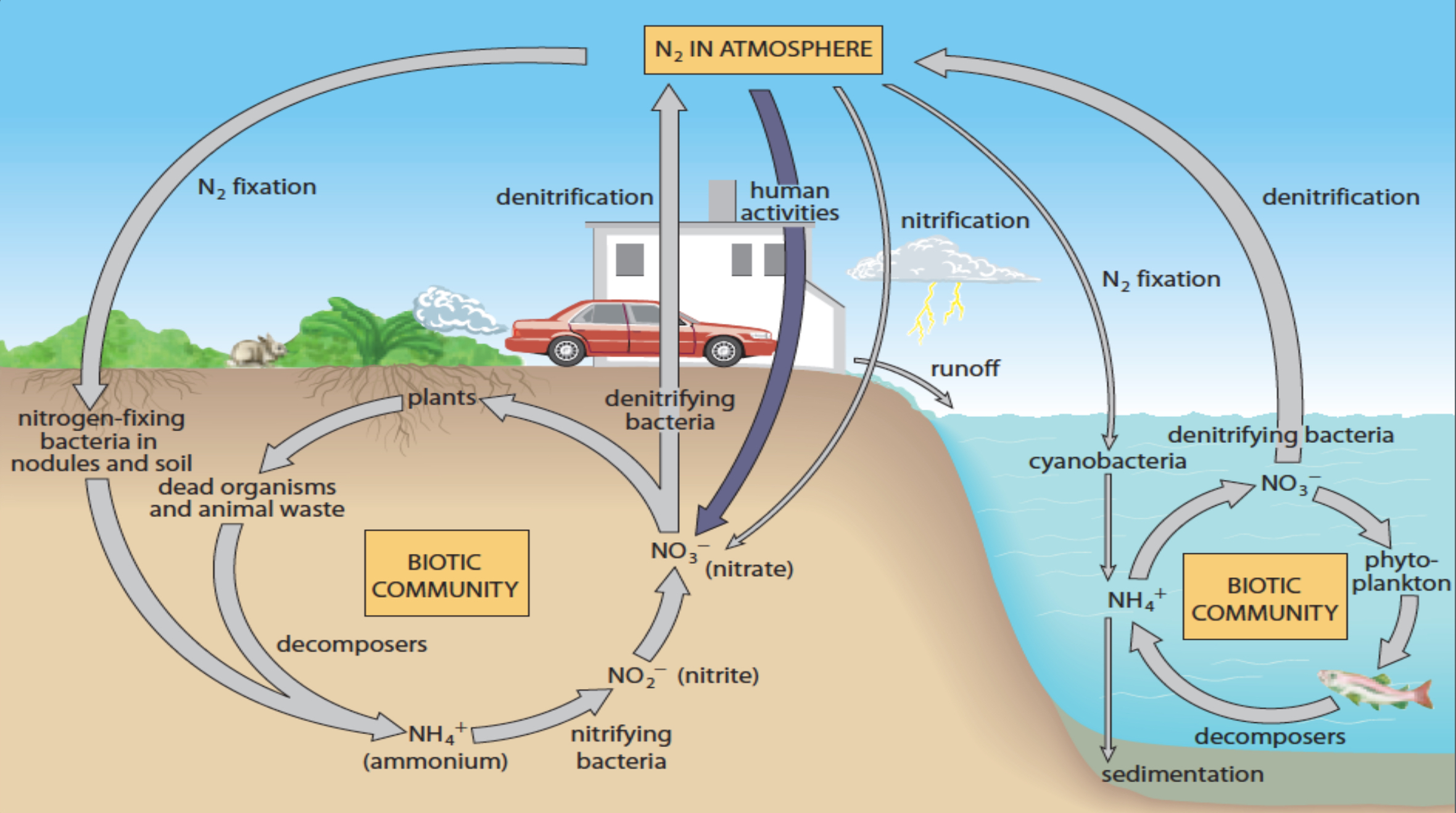
Nitrogen Cycle
Nitrogen fixation by bacteria converts atmospheric nitrogen into forms usable by plants (e.g., ammonium, nitrates), essential for proteins and nucleic acids.
Decomposition and human activities can affect nitrogen levels in soil, impacting plant growth significantly.
Interrelationship of Energy and Matter
Ecosystem productivity reflects the rate at which producers convert solar energy into organic compounds and can be influenced by factors like light availability and nutrient levels.
Sustainability of ecosystems depends on balanced energy and matter exchanges, which can be affected by human activities like pollution and land use changes.
Productivity
Productivity is amount of energy made by photosynthesis (changes between
Factors
-number of producers
-amount of light and heat
-amount of rain
-nutrients
Eutrophication
Dead zones: regions of lakes or oceans in which aquatic life has suffocated due to algal blooms
Nutrients washed into rivers by rain
Sewage washed into the body of water that has a lot of phosphate & nitrate
Surface runoff that bring manure from livestock
Runoff from fertilizer
Wetlands (bogs, marshes, swamps) act as a giant filters as they are saturated with water.
Conclusion
Understanding the interconnected cycles of matter and energy is critical for maintaining ecosystem health and supporting sustainability initiatives in response to environmental changes and human impacts.