5.1-5.3 Redox reactions and Oxidation and reduction
- Oxidation is the loss of electrons, gain of oxygen
- Reduction is gain of electrons, loss of oxygen
- ^^u can remember this by OILRIG^^
- ^^Oxidation is Loss of electrons Reduction is Gain of electrons^^
- sometimes oxidation and reduction happen together this reaction or process is called Redox Reactions.
- Redox reactions can also be defined in terms of electron transfer
- Oxidation is a reaction in which an element, ion or compound loses electrons * The oxidation state of the element is increased * This can be shown in a half equation, e.g. when silver reacts with chlorine, silver is oxidised to silver ions: * Ag → Ag+ + e-
- Reduction is a reaction in which an element, ion or compound gains electrons * The oxidation state of the element is decreased * This can be shown in a half equation, e.g. when oxygen reacts with magnesium, oxygen is reduced to oxide ions:
O2 + 4e- → 2O2-
- Oxidation and reduction in terms of electron transfer can be remembered by the mnemonic 'OIL RIG': Oxidation Is Loss of electrons, Reduction Is Gain of electrons
\ Mnemonic to remember oxidation and reduction in terms of electron transfer
\n \n
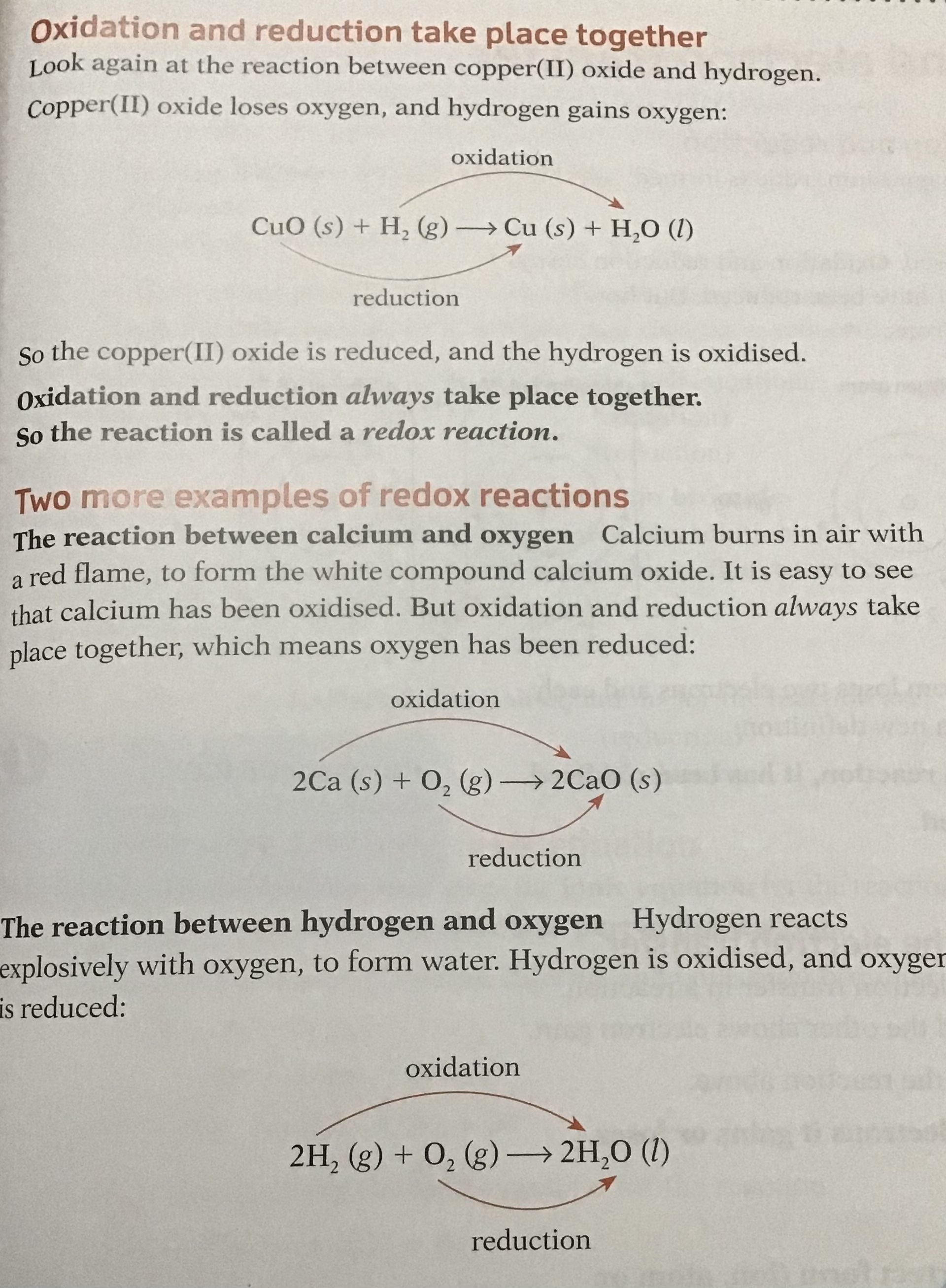
Names using oxidation states
- Transition elements can bond in different ways by forming ions with different charges
- When naming, the charge on the ion is shown by using a Roman numeral after the element's name * e.g. iron can form ions with a 2+ charge, called iron(II) ions or a 3+ charge, called iron(III) ions
- The Roman numeral is the oxidation state of the element
- When iron reacts with oxygen to form iron oxide, the formula depends on the oxidation state of the iron ions * The compound where iron has a 2+ charge has the formula FeO and is called iron(II) oxide * The compound where iron has a 3+ charge has the formula Fe2O3 and is called iron(III) oxide
Oxidation number
- The oxidation state (also called oxidation number) is a number assigned to an atom or ion in a compound which indicates the degree of oxidation (or reduction)
- The oxidation state helps you to keep track of the movement of electrons in a redox process
- It is written as a +/- sign followed by a number (not to be confused with charge which is written by a number followed by a +/- sign)
- E.g. aluminium in a compound usually has the oxidation state +3
Assigning the oxidation state
- Oxidation state refers to a single atom or ion only
- The oxidation number of a compound is 0 and of an element (for example Br in Br2) is also 0
- The oxidation number of oxygen in a compound is always -2 (except in peroxide R-O-O-R, where it is -1)
- For example in FeO, oxygen is -2 then Fe must have an oxidation number of +2 as the overall oxidation number for the compound must be 0 * Table to show some common oxidation states of elements within compounds * Example redox equation: electron loss/gain and oxidation state * zinc + copper sulphate → zinc sulphate + copper * Zn + CuSO4 → ZnSO4 + Cu
- Writing all of the ions present and including state symbols we get: * Zn(s) + Cu2+(aq) + SO42-(aq) →Zn2+(aq) + SO42-(aq) + Cu(s)
- The spectator ions (those that do not change) are SO42-(aq), removing these we can write the ionic equation as: * Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
- By analysing the ionic equation, we can split the reaction into two half equations by adding in the electrons to show how the changes in charge have occurred.
- It then becomes clear that zinc has been oxidised as its oxidation state has increased from 0 in Zn to +2 in Zn2+and it has lost electrons: * Zn(s) → Zn2+(aq) + 2e-
- Copper ions have been reduced as the oxidation state has decreased from +2 in Cu2+ to 0 in Cu and they have gained electrons: * Cu2+(aq) +2e- → Cu(s)
\
Oxidation and Reducing Agents
Oxidation Agent
- A substance that oxidises another substance, in so doing becoming itself reduced
- An oxidising agent gains electrons as another substance loses electrons
- Common examples include hydrogen peroxide, fluorine and chlorine
Reducing agent
- A substance that reduces another substance, in so doing becoming itself oxidised
- A reducing agent loses electrons as another substance gains electrons
- Common examples include carbon and hydrogen
- The process of reduction is very important in the chemical industry as a means of extracting metals from their ores
Example
- CuO + H2 → Cu + H2O
- In the above reaction, hydrogen is reducing the CuO and is itself oxidised as it has lost electrons, so the reducing agent is therefore hydrogen:
- H2 → 2H+ + 2e-
- The CuO is reduced to Cu by gaining electrons and has oxidised the hydrogen, so the oxidising agent is therefore copper oxide
- Cu2+ +2e- → Cu

\ \