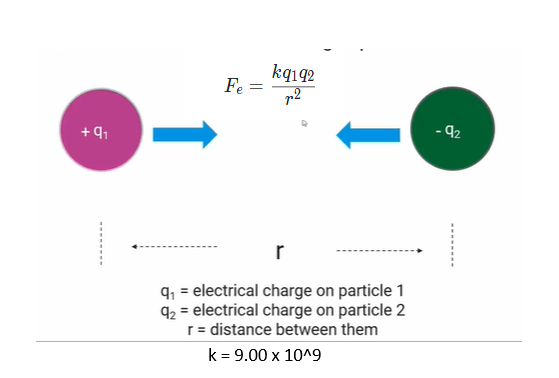
Coulomb's law
used to calculate the force between 2 charged particles,
Coulomb's law states that the magnitude of the electrostatic force is directly proportional to the product of the magnitudes of the charges and inversely proportional to the square of the distance between them.
F = force in terms of newtons
q1 and q2 are measured in coulombs.
the unit for radius is in meters
Coulomb’s law can be used to predict the amount of energy required to remove an electron from an atom.
This relationship is essential for understanding atomic interactions and the stability of matter. It does so by explaining why electrons and protons are held together, why further valence electrons are easier to remove, and why they require more energy to be added to another subshell.
Core electrons shield valence electrons from the pull of the nucleus (this is known as the shielding effect, resulting in a lower effective nuclear charge experienced by the valence electrons. This phenomenon helps to explain trends in ionization energy across the periodic table, as elements with more core electrons tend to have lower ionization energies. )
ionization energy is the energy required to remove an electron from an atom. The energy required to remove the first electron from an atom is the atom’sfirst ionization energy, while the energy required to remove subsequent electrons increases due to the greater positive charge experienced by the remaining electrons.
the Heisenberg uncertainty principle states, the exact position and velocity of a particle cannot be known at the same time. Therefore, we can only determine orbitals, which are found by calculating the Schrödinger equation.
The Schrödinger equation is the equation used to describe the wave function of a quantum system, allowing us to predict the probability of finding an electron in a particular orbital.
The principle quantum number (n) and angular momentum number ( I ) indicate the shell and subshell, respectively. The electron spin quantum number (m [subscript] s) describes the electron spin of all the electrons in that orbital.
All but the s orbital have degenerate orbitals, which are those that have the same energy, but different orientations in space

Aufbau principle: The Aufbau principle states that electrons occupy the lowest energy orbitals first before moving to higher energy levels, ensuring a stable electron configuration.
Hund’s rule: electrons will occupy an empty orbital before pairing up in an already occupied orbital, thereby maximizing the total spin and minimizing electron repulsion.
Pauli exclusion principle: no two electrons in an atom can have the same 4 quantum numbers.
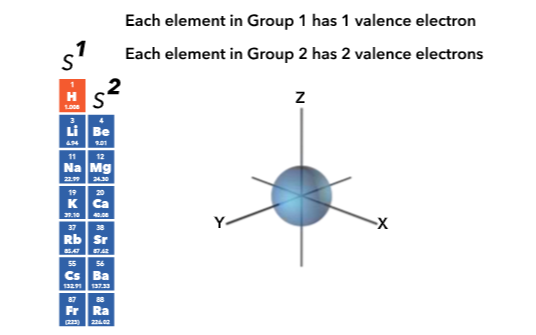
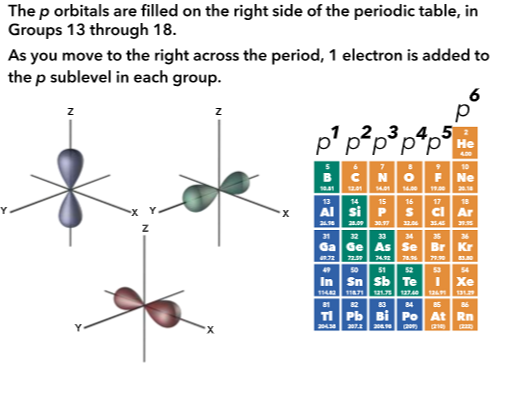
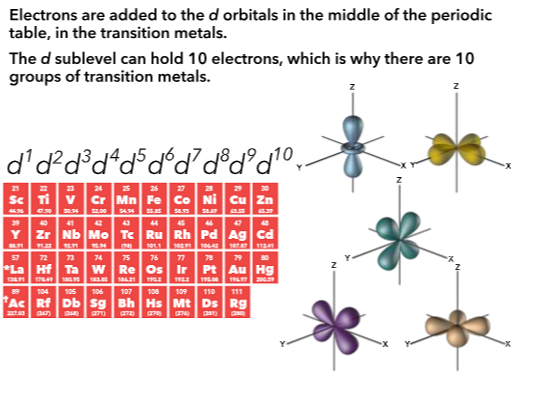
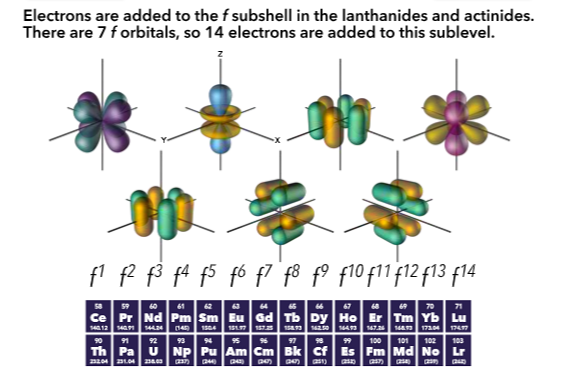
Relation to periodic table
The principle quantum number (n) is represented by the rows of the table
atomic radii (measured from nucleus to the outermost electron shell) tend to decrease across a period due to increased nuclear charge, which pulls electrons closer to the nucleus. )
electron affinity describes the energy released when an electron is added to an atom
electronegativity, electron affinity, and ionization energy increases across a period
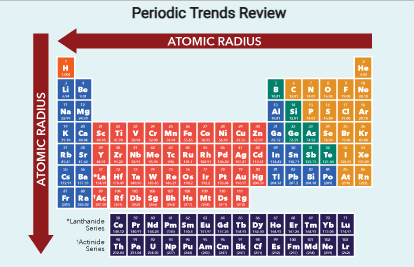
Ionization energy and atomic radius follow opposite trends.