1. Basic principle of pharmacology
Basic Principles of Pharmacology
Drug names
Drug Definition: substance put into body to change physiological function
Pharmacotherapeutics: use of drugs to treat,control, or prevent diseases
Pharmacokinetics: what body does to drug
HOWS DOES BODY METABOLIZE
Pharmacodynamics: what drug does to body
MECHANISM / FUNCTION
how to identify drug
chemical name
generic name
trade/brand name
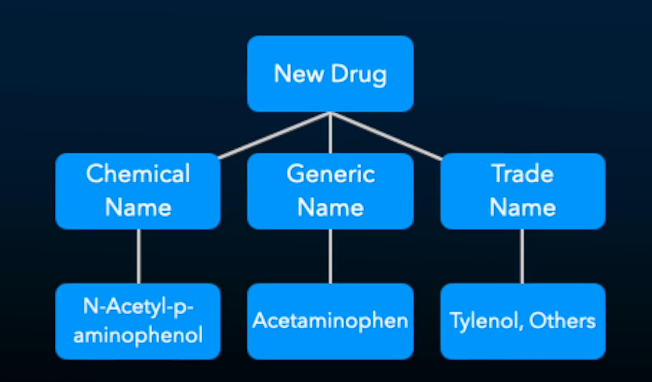
Most generic names are shortened versions of the chemical name
A drug’s ___ name is often shortened or condensed to form the drug’s ___ name. —> chemical, generic
Generic Equivalents
Drug is marketed by generic name after patent expiration
expiration allows companies to market under generic name
Drugs are marketed by ___ name after patent expiration? —> Generic
Generics are equivalent when
same type and amount of active ingredient
administered in same way
same pharmacokinetic profile (same absorption, metabolize, excretion)
same therapeutic effects
Distinguish between a drug’s chemical, generic and trade names
Chemical is chemical structure of drug, generic is shortened chem name, trade is brand name
1.2 Drug Approval and classification
FDA tests/approves drugs for use in patients and clients
decide if prescription vs. over-counter
Phases of drug testing
Preclinical (animal) trials ~1-2 years
Clinical trials
phase 1: healthy volunteers <1 year
phase 2: small patient sample 2 years
test on small group of people with condition for drug (200-300 people)
phase 3: larger sample 3 years
2,000 - 3,000 people
Phase 2 purpose: is it EFFECTIVE
Phase 3 purpose: is it SAFE
3. Approved for marketing
Phase 4: post-marketing surveillance
market studies/phone calls to see how/where drug is being used and if things appear that didn’t in phase 2/3
Every 5000 compounds tested for animals, around 5 make it to human trial, 1 approved
Expedited review when drug has exceptional need or beneficial effects
still tested after administered to gen. pop.
Orphan drugs
for <200,000 people in USA
not cost-effective for companies since not widely used
Implication of drug testing/approval
drug costs
developing company takes on 7-9 years of development
cost a lot for developing other drugs
availability in U.S vs other countries
FDA requires safety and effective
others only safety, leave to practitioner/patient to decide effective
Failure to identify side effects
if testing in 2-3K people, side effects could appear 1in 20,000
If there are serious side effects, BUT drug is effective, Black Box Warning highlights problems and who it SHOULD NOT be used in
Off-Label Prescribing
Prescribing drug for the unintended purpose
Legal bc FDA cannot tell physicians how to practice medicine
off-label is 40-60% of prescriptions
1.3 Basic Concepts in Drug Therapy and Safety
Discuss concepts such as dose-response curves, potency, and therapeutic index.
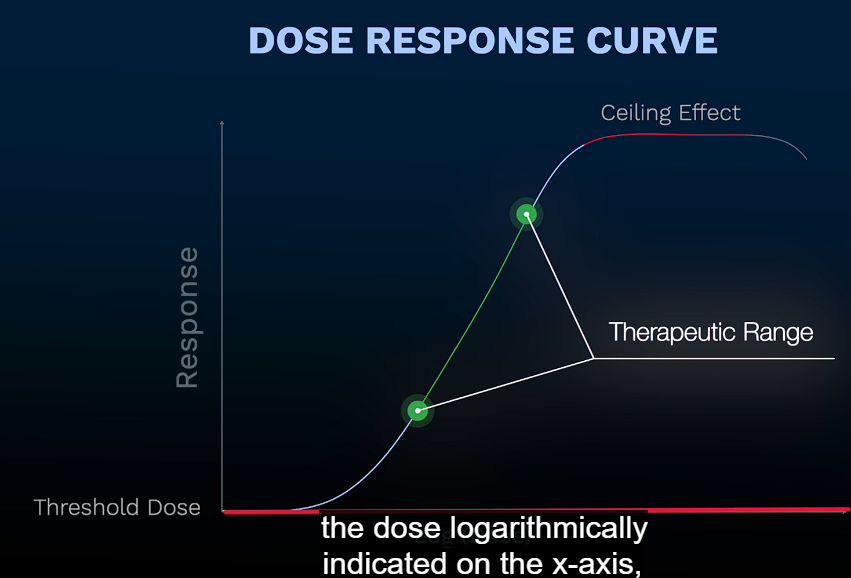
Discuss dose response curves—> refers to dosage of drugs and that effects beginning to appear at a certain level. The dosage and response can increase to a certain level until it plateaus, ceiling effect/maximal efficacy.
Potency —> refers to dosage of drug required to reach certain effect. If first drug achieves the same response as second drug but at a lower dose, first is more potent, not more effective.
Theraputic Index —> refers to ratio of the toxic dose to effective dose with a higher TI indicating safer drug. NO CUTOFF
2. Explain how these concepts can give an indication of a drug’s effects and safety.
Theraputic index refers to the ratio of a toxic dose to the effective dose. The higher the TI, the safer the drug. The dose response curve helps determine the ranges of doses of a certain drug for effects to appear until response plateaus. Potency helps determine between two drugs that may have same responses but one having a lower doseage to achieve the same effects as the other.
Dose Response Curves
no response if too low
response starts to appear at certain “dose/level
Response increase to certain point
response plateau AKA maximal efficacy/ ceiling effect
Drug Potency (used incorrectly)
Potency: how much of a drug is needed to get a certain effect
2 drugs: response at lower dose is more potent
DOESN’T MEAN “MORE EFFECTIVE”
just lower dose achieves SAME RESPONSE for second drug at higher dose
Quantal (cumulative) D-R Curve: Finds correct dosage for certain response in group
Determines drug safety
Median Effective Dose (ED50): dose for beneficial effect in half of sample
Median Toxic Dose (TD50): dose for toxic effect in half of sample
Theraputic Index (TI)
Ratio/number for drug safety dose
TI = TD50 / ED50
Higher TI = safer drug
NO CUTOFF FOR SAFE THERAPUTIC INDEX
1.4 Pharmacokinetics
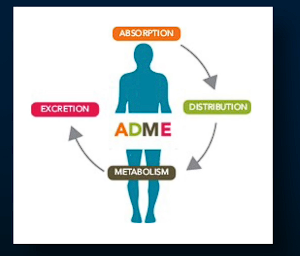
Pharmacokinetics: how body handles/deliver drug
ADME
absoroption, distribution, metabolism, excretion
Absorption (HOW was drug administred)
must move from site administered to another tissue or “central compartment” (bloodstream)
related to route of administration
Golden Rule of Pharmacology
Administer drug directly on tissue that needs it for better effect, less drugs, less side effect
what is the golden rule of pharmacology —> administer drug directly to tissue to better the effect, use less drug, less chance side effect
Routes of Administration
Enteral (alimentary canal)
oral
lingual/sublingual (under tongue)
rectal
buccal (between cheek and gum)
Easy to do
Less predictable absorption
Parental
injection
inhalation
topical (suface of skin issue)
transdermal (will move through skin to bloodstream)
eye drops/ear drops
Usually more difficult
more predictable
Absorption
Bioavailability: % of administered dose that appears in bloodstream
applies to oral medication
IV is 100% bioavailable
First pass effect
if orally, must pass through liver from GI and hepatic portal vein
some of drug destroyed
aware of what % of drug won’t survive “first pass”
Distribution
drug cross membranes and tissue as hurdles
factors
administration route
physiocochemical prop. of drug
good at passing membranes?
binding to plasma proteins
various barriers and carriers
blood brain barrier
Volume of distribution (Vd)
Vd = amt. drug adminis. / concentration in plasma
Vd compared to 42 (liters of water in body)
if close to 42, even thru body
<< 42, plasma retains
>> 42, tissue retains
Drug Storage/Accumulation
Primary sites
fat, muscle, bone, liver, kidneys, other tissue/organ
Primary problem
local tissue damage
bone storage can lead to fractures
Redistribution
stored in tissue then leaks later
anesthesia
Metabolism
active form of drug is changed chemically to an inactive or less active byproduct or “metabolite”
More polar, water soluble metabolite
excreted by kidneys
“biotransformation”
Liver is primary site for metabolism
kidney, lung, gi tract, skin
enzyme metabolize drug via:
phase 1: oxidation: add oxygen, remove hydrogen, reduction, hydrolysis (break apart drug)
phase 2: conjugation
2nd molecule to phase 1 byproduct
Excretion
Kidney (primary role)
also lungs, gi tract
Minor sites
sweat, saliva, breast milk
25-30% drugs excreted “intact”
urine pH affects excretion vs reabsorption
1.5 Pharmacokinetic Variables
Drug elimination
Clearance (CL)
rate of drug removal from body
measures one organ or sum of all organs involved
Depends on
how much blood is going to organ that does clearance
Extraction ratio
how good is organ taking out of bloodstream to metabolize
Extraction Ratio

Use for Clearance
drugs cleared well if organ has high blood flow (Q) and good extraction ratio
disease/illness affecting organ’s blood flow or extrac. ratio will impair ability to clear drugs
decrease in liver enzymes, reduction in kidney bloodflow
Lower CL prolong drug effects
Dosing Schedules
IV drip, Patches
continuous level of drug in bloodstream
Drugs given intermittently (pills) cause peaks and troughs in plasma levels
Correlate patients in dosing cycle (peaks/troughs) if odd things occur
Oral drugs peak 30-60 min after taking
Effects can be increased by heat exercise, massage
Factors affecting normal pharmacokinetics
disease
age
genetics
gender, body comp, diet, other chem (alc/nicotine), physical factors
