Bonding, structure and properties of matter
Ionic bonding
ionic bonding occurs between a metal and a non-metal
metal atoms transfer their electrons to non-metal atoms, become ions.
electron transfer when these bonds form can be shown by a dot and cross diagram:
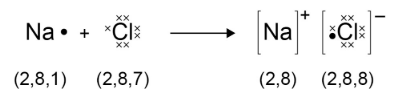 this diagram only shows electrons in the outer shell, but you get the picture.
this diagram only shows electrons in the outer shell, but you get the picture.
remember to draw brackets around the ions, and state their charge!!!
you need to have the same amount of atoms/ions on each side - you can’t just magically create an ion.
features of an ionic compounds:
strong electrostatic forces of attraction between oppositely charged ions, acting in all directions
takes a lot of energy to overcome these bonds, thus these compounds have high melting and boiling points
ionic lattice structure formed
compounds aren’t easy to separate
conduct electricity when molten or aqueous, because their ions can move freely and carry their charge in this state
solid ionic compounds cannot conduct electricity
ionic compounds can also be shown as 3D diagrams, like these, called ball and stick diagrams.
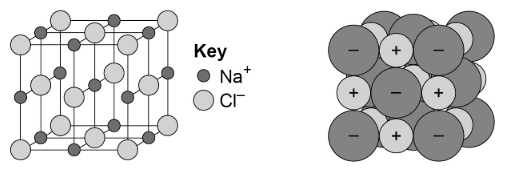
the dot and cross diagrams are not well suited for showing large numbers of bonds in a lattice, and don’t convey the idea of a lattice like the ball and stick diagrams. ball and stick diagrams, however, can become quite difficult to draw and interpret.
Covalent bonds
non-metals share one or more pairs of electrons to complete their outer shells and become stable
polymers are large, covalently bonded molecules.
atoms are linked with strong covalent bonds
intermolecular forces between molecules are strong, so substances are solid at room temperature
giant covalent structures double down on that, consisting of many atoms, covalently bonded, in a lattice structure. e.g diamond and graphite (which there’s more detail on later)
these substances have very high melting points, because all of their atoms are linked to other atoms by strong covalent bonds that have to be overcome to melt or boil. thus they’re solid at room temperature (and also far into high temperatures too)
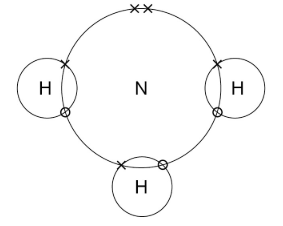
this is the dot and cross diagram for ammonia, NH3
nitrogen is in group 5, so has 5 electrons in its outer shell. it shares 3 of these with hydrogen atoms that only have one electron in their outer shells to form stable ammonia. this can also be represented by:
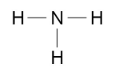
the number of lines between the atoms shows how many electrons they share.
Properties of small molecules
substances with small molecules are usually gases or liquids (at room temperature) with low melting and boiling points
there are weak intermolecular forces between molecules, overcome when the substance melts or boils, instead of the strong covalent bonds. intermolecular forces increase with size of the molecules, so larger molecules have higher melting and boiling points anyway.
don’t conduct electricity because molecules have no overall charge
Metallic bonding
bonds in a metal consist of positive ions and their delocalised electrons, arranged in a regular pattern.
delocalised electrons are free to move throughout the structure. this has a few affects:
bonds are strong because electrons shared throughout substance
can carry electrical charge and thermal energy through the material, making metals good conductors
in pure metals, atoms are arranged in layers, which allows metals to be bent and shaped (ductile)
giant structure of atoms with strong metallic bonding means most metals have high melting and boiling points.
alloys are made of 2 or more different types of metal. the different sizes of atoms distort the layers in the structure of the metal, making it hard for them to slide over each other, and thus making alloys harder than pure metals
States of matter
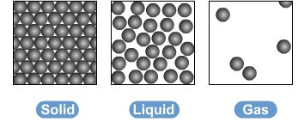
this model helps to explain how state changes occur - the amount of energy to change state depends on strength of the forces between the particles of a substance. stronger forces also means higher melting and boiling points
however this model has some limitations:
no forces shown
all particles are represented as solid, inelastic spheres
this is a problem because state changes involve changes in strength of intermolecular forces, which isn’t shown. also doesn’t show the increases in kinetic energy of particles between state changes
Structure and bonding of carbon (covalent bonds)
diamond
every carbon atom is joined to 4 other carbon atoms, the maximum possible (because carbon has 4 electrons in its outer shell) thus makes it hard
high melting point
does not conduct electricity because there’s no free electrons/ions to carry charge
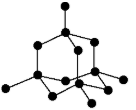
graphite
every carbon atom is joined to 3 other carbon atoms, forming layers of hexagonal rings. there aren’t covalent bonds between the layers. this means the layers can slide over each other, so graphite is soft and slippery
there’s one electron delocalised from each carbon atom, so it conducts electricity.
graphene
graphene is a single layer of graphite.
it’s very strong because the atoms in the layer are tightly bonded, and elastic because planes of atoms flex without breaking apart. it’s thin nature and ability to conduct electricity like graphite makes it good for use in electrical circuits and in composites, which are basically combinations of materials
fullerenes
fullerenes are molecules of carbon atoms that are hollow. they may be formed from the hexagonal rings of graphine (3 carbon bonds), or may contain rings with 5 or 7 carbon atoms
the first fullerene discovered was C60, which is spherical.
nanotubes are fullerenes made into a cylindrical shape, giving them a high length to diameter and thus surface area to volume ratio. this makes them good for use in things like:
drug delivery systems in the human body
catalysts
reinforcing materials
lubricants
Nanoparticles
nanoparticles are anywhere between 1 and 100 nanometres across, containing just a few hundred atoms. in the ‘really super small particle’ pecking order, they’re the smallest:
nanoparticles: 1 to 100 nm diameter
fine particles: 100 to 2500 nm
coarse particles (aka dust): 2500 to 10000nm
nanoparticles often have different characteristics to the same material in bulk because of their small size, which makes way for a very high surface area to volume ratio. this may mean smaller quantities are needed to be effective compared to a normal molecule.
nanoparticles have a few uses you need to know:
as catalysts, due to high SA:V ratio
could be used to make electronic sensors, and also to conduct electricity in small circuits
may be used to make building materials lighter yet stronger
in cosmetics like suncream - nanoparticles with UV protecting properties get absorbed into the skin
lubricants
they do, however, come with a few disadvantages:
may be absorbed into cells, then into the bloodstream, and then into crucial parts of the body like the brain where they may cause harm
we have no idea about their long term effects yet