Lecture 27: Reaction Rates
Reaction Rates: Chem 101
Quiz on Hypotonic Solutions
What happens to blood cells in a hypotonic solution?
They swell up and explode (hemolysis)
Demo: Fireworks on Ice
Initial Reaction:
Mix powders, nothing happens initially.
Adding water triggers a reaction.
Key Questions:
Why do reactions occur under some conditions, but not others?
the water dissolves some of the ions which allowed the ions to find other ions and react
the water was a catalyst
Why are some fast, while others are slow?
Why do some reactions go 100% while others have very low yields?
Can we influence the reaction rate or yield?
Collision Theory of Reactions
Kinetic theory of reactions:
molecules are constantly moving
molecules must collide to react
collisions must have sufficient energy
collision must have correct orientation

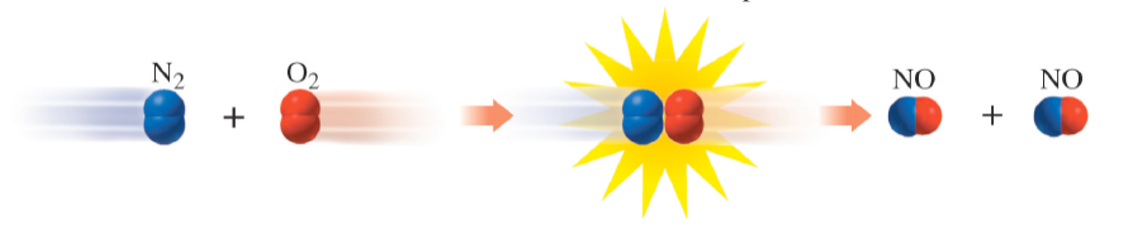
Activation Energy
Definition:
Minimum energy needed for a reaction to occur upon proper collision of reactants.
Reaction Rate
Definition:
the speed at which reactant is used up, or amount of reactants consumed per unit time
is the speed at which product forms or amount of products formed per unit time
Factors Affecting Reaction Rate
Concentration:
Increasing the concentration of reactants increases the number of collisions which increases the reaction rate
Temperature:
an increase in kinetic energy —> more collisions
an increase in ave. K. E. —> more molecules with enough energy to break bonds
therefore an increase in reaction rate
Catalyst:
lowers the energy of activation
often improves the orientation of the collision
speeds up the tare of a reaction
is not used up during the reaction
Summary: Increasing Reaction Rate
Requirements:
Higher concentration.
Higher temperature.
Use of a catalyst.
To slow down a reaction:
Lower concentration.
Lower temperature.
Remove the catalyst.
Quiz on Reaction Rate Over Time
What happens to the reaction rate over time?
a) Remains constant
b) Speeds up
c) Slows down
d) Not enough information
Answer: Using up reactant = decreasing reactant concentration = slower reaction rate.
Demo: Reversible Reactions
Many reactions are reversible.
Notation:
Forward reaction: 22()—> 24()
Reverse reaction: 24()→22()
Characteristics of Reversible Reactions
Proceed in either direction depending on conditions.
Represented by half arrows.
Reach equilibrium in a closed system at constant temperature
What happens at equilibrium?
Rate of forward reaction = rate of reverse reaction.
Concentrations of species remain constant.
Quiz on Reversible Reactions
Write the forward and reverse reactions for the following reversible reaction:
Forward: CH4(g) + 2H2S(g) → CS2(g) + 4H2(g)
Reverse: CS2(g) + 4H2(g) → CH4(g) + 2H2S(g)
Chemical Equilibrium
A reversible reaction is in equilibrium when:
Rate of forward reaction = Rate of reverse reaction
the concentrations of reactants and products stop changing
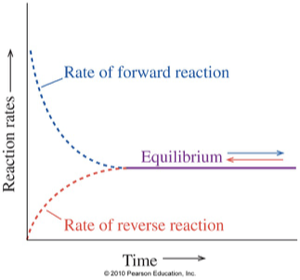
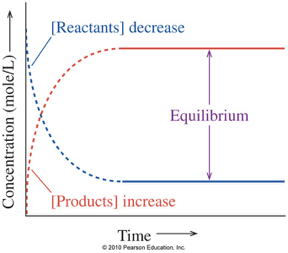
Quiz on Equilibrium
Why do reactant and product concentrations stop changing at equilibrium?
a) Forward and reverse reactions stop occurring
b) Molecules no longer have enough energy to overcome activation barrier
c) Rate at which reactants and products are formed is the same
d) Reactant and product concentrations are now the same
Equilibrium Examples
Examples of equilibrium:
Vapor pressure above a liquid.
Phase changes.
Osmosis.
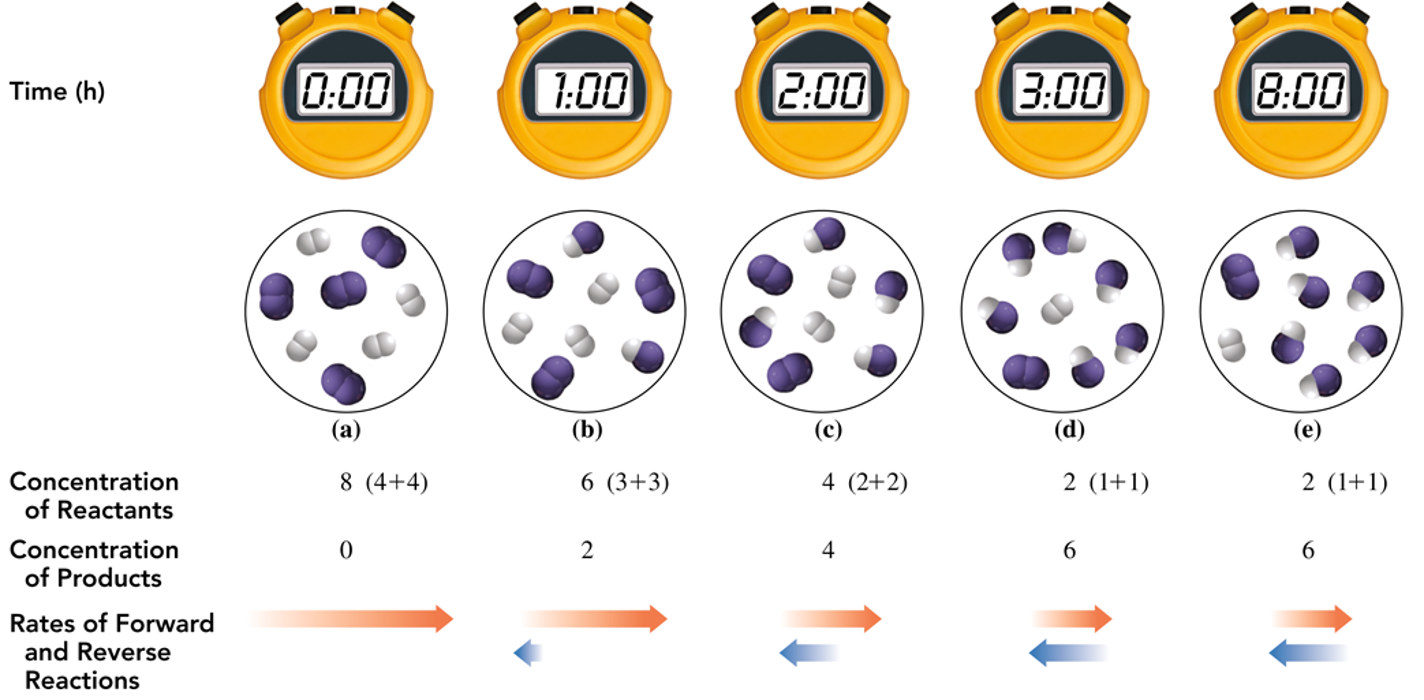
Rates of Forward and Reverse Reactions
Example Reaction:
H2(g) + I2(g) ⇌ 2HI(g)
2 SO2 (g) + O2(g) ⇌ 2SO3
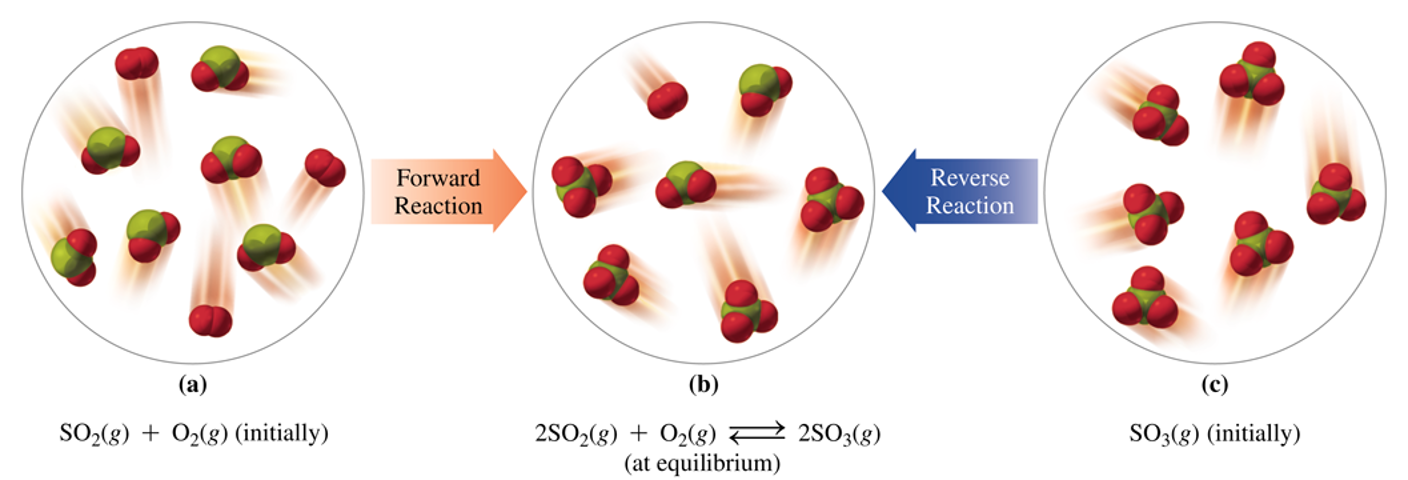
Reaction progression from initial state to equilibrium, with concentration changes over time.