RP2: Enthalpy Change
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
16 Terms
What is the main objective of this practical?
To measure the enthalpy change (H∆) for a chemical reaction (like neutralisation) or a physical process (like dissolving a salt) using simple calorimetry.
What is the key apparatus used as a calorimeter in this experiment, and why?
A polystyrene cup. It is a good insulator, which minimises heat loss to (or heat gain from) the surroundings, making the measured temperature change more accurate.
What is the purpose of placing a lid on the polystyrene cup?
To further reduce heat loss to the surroundings, specifically by evaporation and convection.
What is the equation used to calculate the heat energy (q) absorbed or released?
q = mc∆T
q = heat energy change (in Joules)
m = mass of the solution being heated (in grams)
c = specific heat capacity (in $J g-1 K-1)
∆T = the change in temperature (in K or °C)
What two key assumptions are made about the 'm' and 'c' values when using q = mc∆T
Mass (m): The density of the (aqueous) solution is assumed to be the same as water (1.0 g cm-3). Therefore, 50 cm3 of solution has a mass of 50 g.
Heat Capacity (c): The specific heat capacity of the solution is assumed to be the same as pure water (4.18 J g-1 K-1).
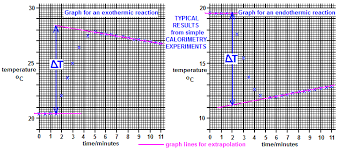
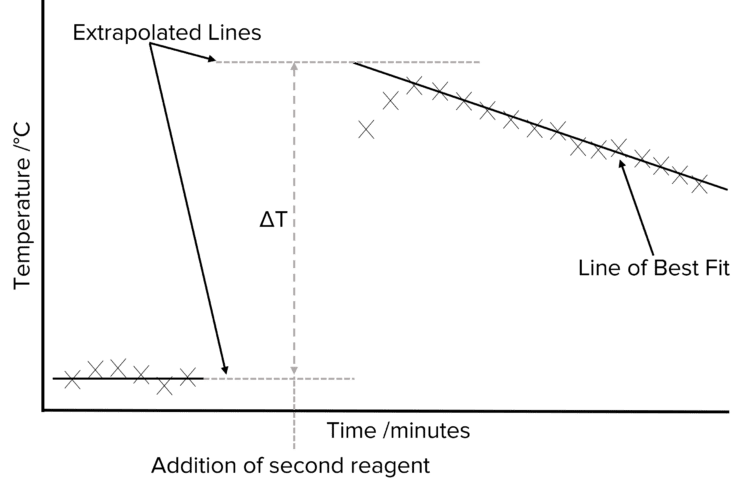
How is the maximum temperature change (∆T) found most accurately?
By plotting a graph of temperature vs. time.
Plot the cooling section of the graph.
Extrapolate the cooling curve (a line of best fit) back to the time of mixing (time = 0).
∆T is the difference between this extrapolated temperature and the initial temperature.
What is the equation used to convert the heat energy (q) into the molar enthalpy change (∆H)?
∆H = -q / moles
q must be in kJ (divide J by 1000).
moles is usually the moles of the limiting reactant, or moles of water formed in neutralisation.
The negative sign corrects the sign: if the reaction is exothermic, ∆H is positive, 'q' is positive, but ∆H must be negative.
What is the main source of inaccuracy in this experiment?
Heat loss to the surroundings (and to the apparatus, e.g., the thermometer).
This makes the measured ∆T smaller than the true value, making the calculated enthalpy change less exothermic (or less endothermic).
⚠ SAFETY What is the main safety hazard when using acids (like HCl) and alkalis (like NaOH)?
They are corrosive (or irritant at low concentrations). They can cause severe damage to skin and eyes.
⚠ SAFETY What is the main safety hazard associated with solid sodium hydroxide?
It is corrosive and can cause severe burns. It is also hygroscopic (absorbs moisture) and the dissolving process is highly exothermic.
⚠ SAFETY What are the two essential safety precautions that must be taken during this practical?
Wear eye protection (goggles) at all times to protect against splashes of corrosive chemicals.
Wear a lab coat to protect skin and clothing. (Also: clear up spills immediately, and if chemicals make contact with skin, wash with copious amounts of water).
Question 1 (1 mark) State the main reason why a polystyrene cup is a suitable container for this experiment.
Answer: It is a good insulator / it minimises heat loss. (1)
Question 2 (2 marks) In an experiment to measure the enthalpy of neutralisation, 25.0 cm³ of acid was added to 25.0 cm³ of alkali. When calculating the heat energy change (q), state the two assumptions made about the solution.
The density of the solution is the same as water (1.0 g cm-3). (1)
The specific heat capacity of the solution is the same as water (4.18 $J g-1 K-1). (1)




Question 5 (6 marks) Describe a method to find the enthalpy of neutralisation between 1.0 mol dm-3 ethanoic acid and 1.0 mol dm-3 sodium hydroxide, explaining how you would obtain an accurate value for the temperature change.
Rinse and fill a burette with 1.0 $mol dm-3 ethanoic acid. Rinse a pipette with 1.0 $mol dm-3 NaOH.
Use the pipette to add an accurate volume (e.g., 25.0 cm3) of the NaOH into a polystyrene cup.
Place the cup in a beaker (for stability) and place a lid on top. Use a thermometer (reading to ± 0.1 °C) inserted through a hole in the lid.
Measure the temperature of the NaOH solution every minute for 3 minutes to get a stable initial temperature.
At the 4th minute, add 25.0 cm³ of the ethanoic acid from the burette to the cup. Do not record the temperature at this time.
Continue recording the temperature of the mixture every minute from 5 minutes to 10 minutes, stirring gently.
To find ∆T: Plot a graph of temperature (y-axis) vs. time (x-axis). Extrapolate the cooling curve (the sloping line after the peak) back to 4 minutes. Read the temperature ($T_{max}$).
∆T = $Tmax - Tinitial.
Calculate q using q = mc∆T (where m = 50.0 g) and ∆H using ∆H = -q /moles (where moles = 0.025 mol).