CEM 141 MSU Exam 3
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
47 Terms
Ionization Energy
energy required to remove an electron from an atom in the gas phase
Left Side Electrons
have weaker attractions to remove electrons
easier to remove electrons from the left side of the table than the right side
Does it take MORE or LESS energy to remove an electron from Li than from Na?
MORE
Why? Because as we move down a group, the orbitals become larger and weaker. While going across the effective nuclear charge increases. (holds onto the electrons tighter)
Big Bang evidence
universe is expanding (red shift)
Where do atoms come from?
The Big Bang
What were elements formed by?
Nuclear reactions
Chemical reactions
involve rearrangements of valence electrons
the element undergoing a chemical reaction does not ever change
Ionization energies are affected by
-size of an atom
-size of positive charge
Periodic Trends
-small atoms have higher ionization energies
-they are inversely related
Atomic Radius Decreases ----> Ionization energy increases
when the radius decreases, it pulls the electrons closer. decreasing the size of an atom
Nuclear Reactions
Fusion and Fission
Fusion
two lighter nuclei combines to form a heavier nucleus (energy released)
Fission
a heavier nucleus is broken a part to form lighter, more stable nuclei energy
Atomic # =
# of protons
Atomic mass =
what's left over
Do isolated atoms or molecules have melting or boiling points?
No
Do isolated atoms or molecules exist in a state (solid, liquid, gas)?
No
Why do bonds form between atoms?
because valence electrons are attracted to the nuclei of other atoms
Covalent Bonds
two hydrogen atoms interact and form a covalent bond
What properties do metals have?
-hard
-conduct electricity
-shiny
-react w/water
-malleable
Bonding in Metals
atomic orbitals combine with each other to form molecular orbitals
Covalent bonds hold atoms together within..
molecules
Molecular interactions hold atoms together between...
molecules
T/F: Covalent bonds are stronger
True
Why are metals shiny?
photons of wavelengths are absorbed and then re-emitted
Changes of state (solid --> liquid ---> gas)
REQUIRES energy
Changes of state (gas --> liquid --> solid)
RELEASES energy
Where does the energy come from?
The surroundings
Where are the attractions coming from?
Between the molecules
Ionic Bonds -
are between a metal and non-metal
Bonding in Diamond
each carbon atom is bonded to make more carbon atoms
Valence Bond
atomic orbitals overlap to form a bond
Bonding in Graphite
one s and two p orbitals hybridize to give three Sp2 orbitals
Heterogeneous Compounds
bonding in carbon compound bonds to C,H,O,N,S,P
Lewis Structure
1. group number = how many valence electrons
2. for cations = (+) remove electrons
3. for anions = (-) add electrons
4. use 2 electrons for each atom
Formal Charge:
Valence electrons on free atom - #bonds to atoms - non bonded electrons = Formal charge
Example: OH-
6 from oxygen +1 from hydrogen +1because its minus 1 ion = 8 valence electrons
Electronegativity
ability of an element to attract electrons to another electron
Electronegativity ...
increases across the table
decreases down the table
Polar Bonds
when two atoms of different eleectronegativities bond, the electrons are not shared equally
- results in dipole
Which Bonds are Polar?
H-F
H-O
CH3F
H20
Which Bonds are Not Polar and have similar electrongeativity
H-C
C-C
CF4
CO2
SO3
Triple Bonds
C - sp hybridized
One sigma bond
Two pi bonds
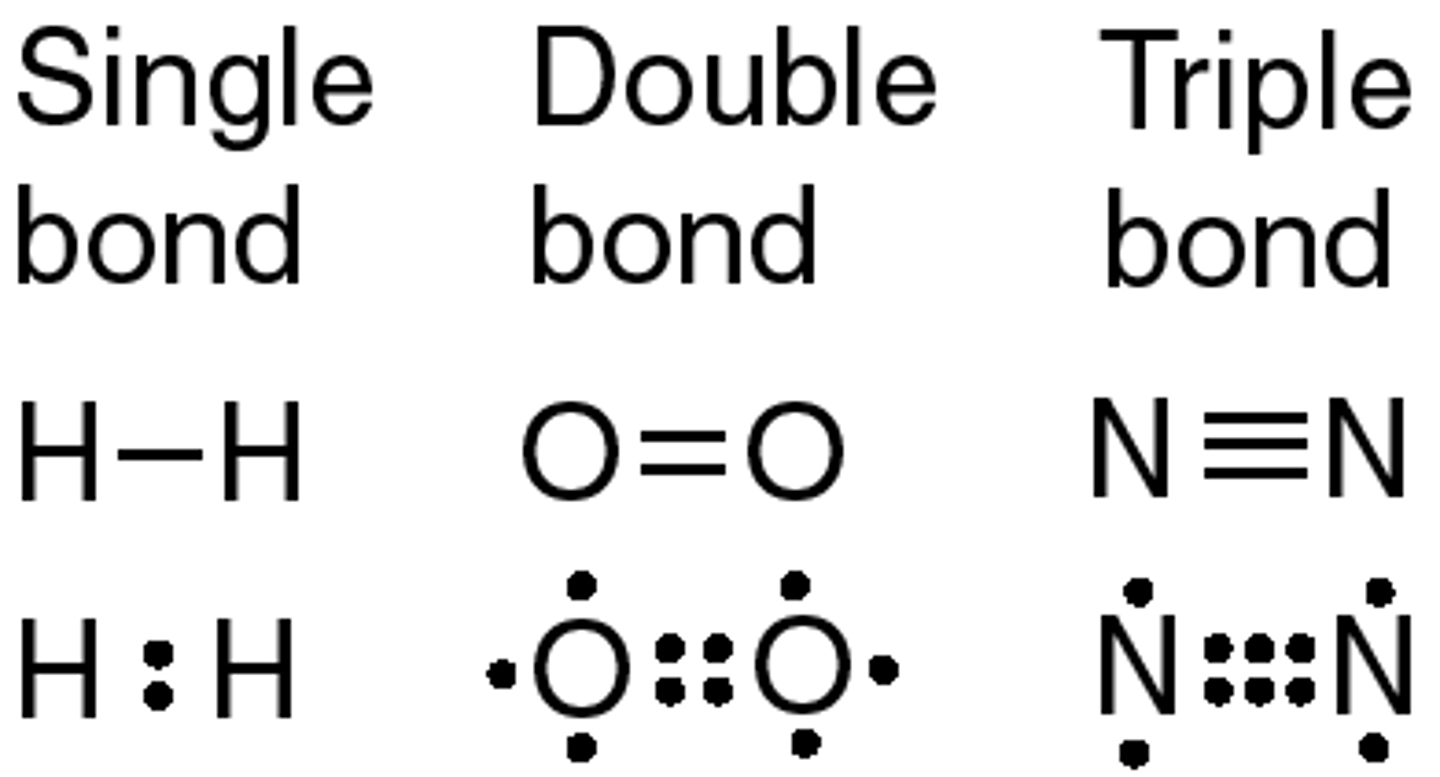
Types of Intermolecular Forces
LDFS
Dipole-dipole interactions
Hydrogen bonds
London Dispersion Forces
-Present in all substances
-Temporary fluctuating dipoles
-Depends on size, surface area, and shape of molecule
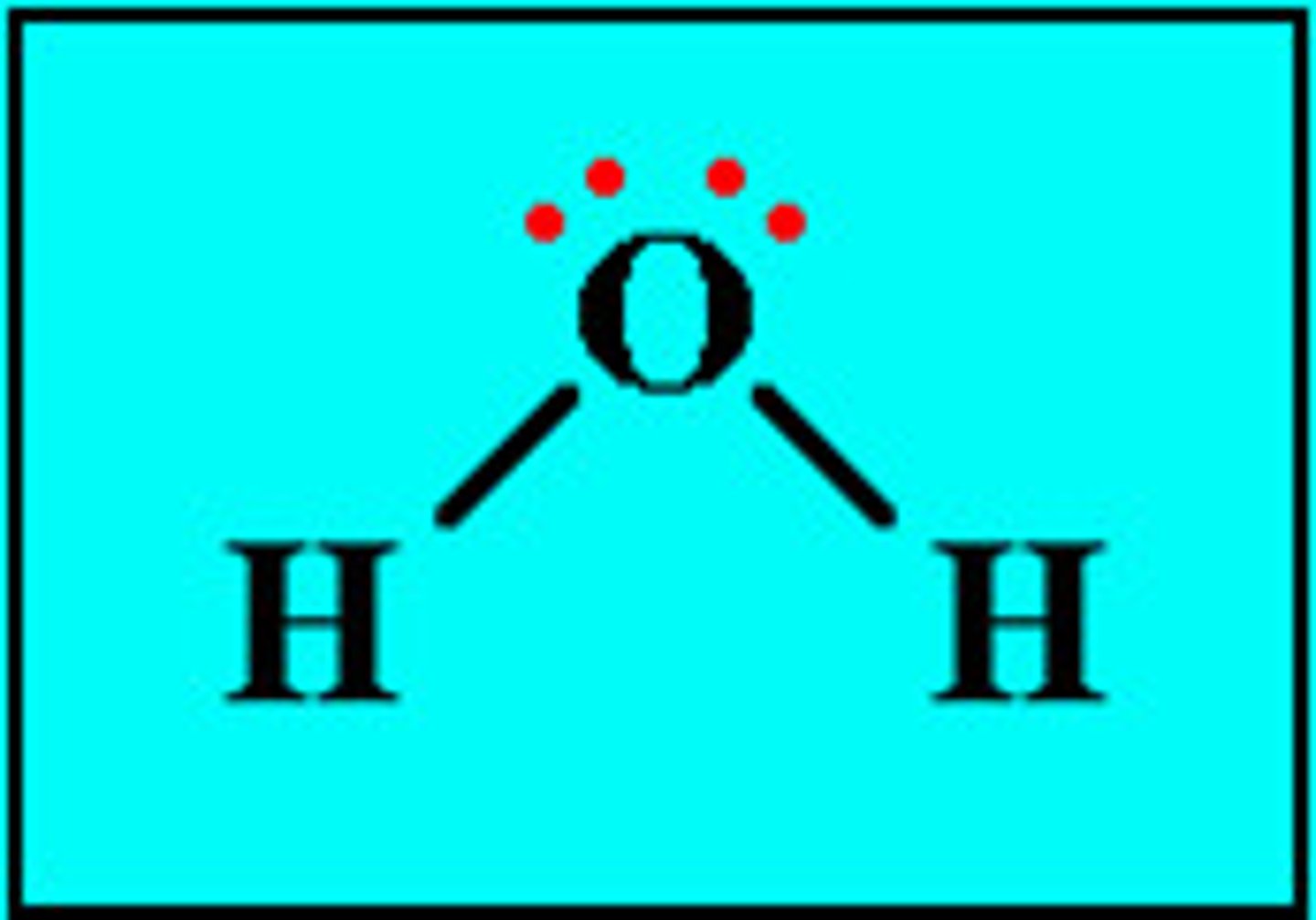
Lewis Dot Structure for H2O
Dipole-dipole Interactions
-Present in polar substances (along with LDF)
-Typically stronger than LDF
Examples: HCL, CH3, CH2O,Ch3OCH3
Hydrogen Bonding Interactions
-Present between two molecules
-H-bonds are extremely strong dipole interactions
Examples: H2O, CH3OH, CH3Ch2OH, HF, NH3