Types of Enthalpy Changes
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
What is the standard enthalpy change of atomisation?
The enthalpy change when one mole of gaseous atoms is formed from an element in its standard state.
*For solid elements, it is known as the enthalpy change of sublimation.
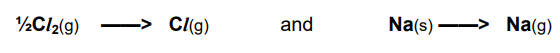
Why is the enthalpy change of atomisation endothermic?
Energy is required to break the bonds holding the atoms together.
What is the lattice formation enthalpy?
The enthalpy change when one mole of an ionic crystal lattice is formed from its gaseous ions under standard conditions.
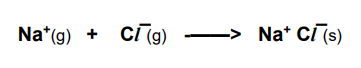
Why is the lattice formation enthalpy highly exothermic?
There are strong electrostatic attraction between ions of opposite charge; a large amount of energy is released when these bonds are formed.
What is the lattice dissociation enthalpy?
The enthalpy change when 1 mole of an ionic crystal lattice dissociates into its gaseous ions under standard conditions.

Why is the lattice dissociation enthalpy highly endothermic?
There are strong electrostatic attraction between ions of opposite charge; a large amount of energy is required to overcome this attraction.
What is the standard enthalpy change of formation?
The enthalpy change when 1 mole of a compound is formed in its standard state from its elements in their standard states.
It is usually exothermic.
Note: Elements in their standard states have an enthalpy change of formation of 0.
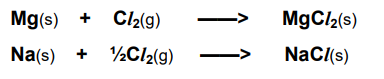
What is the first ionisation energy?
The energy required to remove 1 mole of electrons from 1 mole of gaseous atoms to form 1 mole of gaseous 1+ ions.

Why is the first ionisation energy always endothermic?
Energy is required to overcome the pull of the nucleus on the electron.
What is the first electron affinity?
The enthalpy change when 1 mole of gaseous atoms acquire 1 mole of electrons to form 1 mole of gaseous 1- ions.
Why is the first electron affinity always exothermic?
Energy is released due to the electron being strongly attracted to the nucleus.
What is the 2nd electron affinity?
The enthalpy change when 1 mole of gaseous 1- ions acquire 1 mole of electrons to form 1 mole of gaseous 2- ions.
Why is the second electron affinity endothermic?
Energy is required to overcome the electrostatic force of repulsion between the electron and the negatively charged ion.