Chem Quiz 4- Election and Molecular Geometrics
1/34
Earn XP
Description and Tags
The fucking bullshit we have to know or we fail chem and loose 2k
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
35 Terms
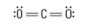
How many Electron groups?
2
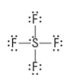
How many electron groups?
5
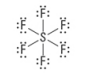
How many electron groups?
6
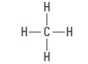
How many bonding groups?
4
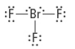
How many bonding groups?
3
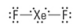
How many bonding groups?
2
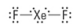
How many lone pairs?
3
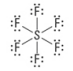
How many lone pairs?
0
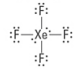
How many lone pairs?
2
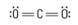
What are the Electron and Molecular Geometries?
Linear, Linear
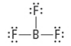
What are the Electron and Molecular Geometries?
Trigonal planar, trigonal planar
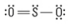
What are the Electron and Molecular Geometries?
Trigonal planar, bent
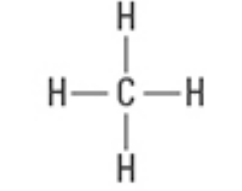
What are the Electron and Molecular Geometries?
Tetrahedral, Tetrahedral
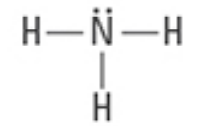
What are the Electron and Molecular Geometries?
Tetrahedral, Trigonal pyramidal
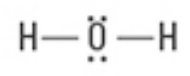
What are the Electron and Molecular Geometries?
Tetrahedral, Bent

What are the Electron and Molecular Geometries?
Trigonal bipyramidal, Trigonal bipyramidal
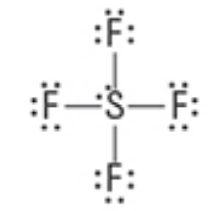
What are the Electron and Molecular Geometries?
Trigonal bipyramidal, Seesaw
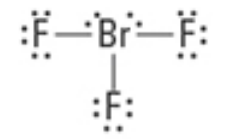
What are the Electron and Molecular Geometries?
Trigonal bipyramidal, T-Shaped
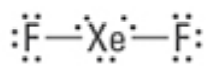
What are the Electron and Molecular Geometries?
Trigonal Bipyramidal, Linear
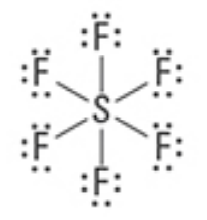
What are the Electron and Molecular Geometries?
Octahedral, Octahedral
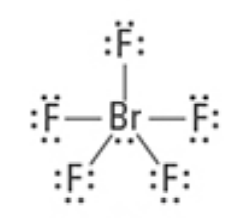
What are the Electron and Molecular Geometries?
Octahedral, Square Pyramidal
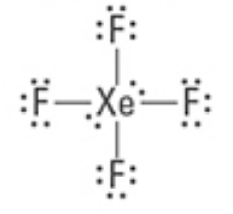
What are the Electron and Molecular Geometries?
Octahedral, Square Planar
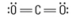
What are the approximate bond angles?
180

What are the approximate bond angles?
120
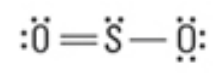
What are the approximate bond angles?
<120
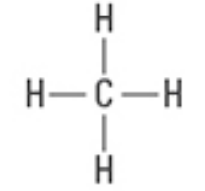
What are the approximate bond angles?
109.5

What are the approximate bond angles?
<109.5
What are the approximate bond angles?
<109.5
What are the approximate bond angles?
120 (equatorial), 90 (axial)
What are the approximate bond angles?
120 (equatorial), 90 (axial)
What are the approximate bond angles?
<90
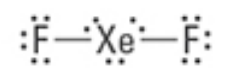
What are the approximate bond angles?
180

What are the approximate bond angles?
90
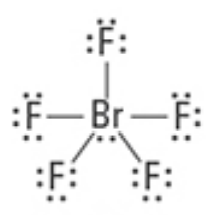
What are the approximate bond angles?
<90

What are the approximate bond angles?
90