Energetics
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
What doe we call chemical reactions that release energy
Exothermic
What do we call chemical reactions that take in energy
Endothermic
Definition for enthalpy of formation
The enthalpy of formation of a compound is defined as the enthalpy change (heat energy) when one mole of the compound is formed from its elements. When all reactants and products are in their standard states under standard conditions
Example of enthalpy of formation
Basically solve the equation normally but then once its solved find out 1 mole of the product by diving everything by the coefficient of the product
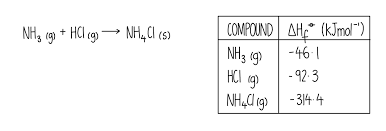
Definition of enthalpy of combustion
The enthalpy change when one mole of a substance undergoes complete combustion in sufficient oxygen. When all reactants and products are in their standard states under standar conditions
Example of enthalpy of combustion

What are the standard conditions
298K (25 degrees) and 100KPa
Definition for Enthalpy of reaction
The amount of heat taken in or evolved under standard conditions when the quantities shown by an equation react completely to form a product. When all reactants and products are in their standard states under standard conditions
What are standard states
The most stable version/allotrope of themselves in standard conditions.
Equation for heat energy
q=mc △T
Equation for enthalpy of combustion
△Hoc = q-1000/moles
Equation for ethalpy of reaction
△Hro = - q/1000/moles