CH 13 Solutions and Colligative Properties: Chemistry Concepts and Calculations
1/68
Earn XP
Description and Tags
On CH 13- Solubility & Solutions
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
69 Terms
Solutions
Homogeneous mixtures where the solute and solvent are mixed uniformly.
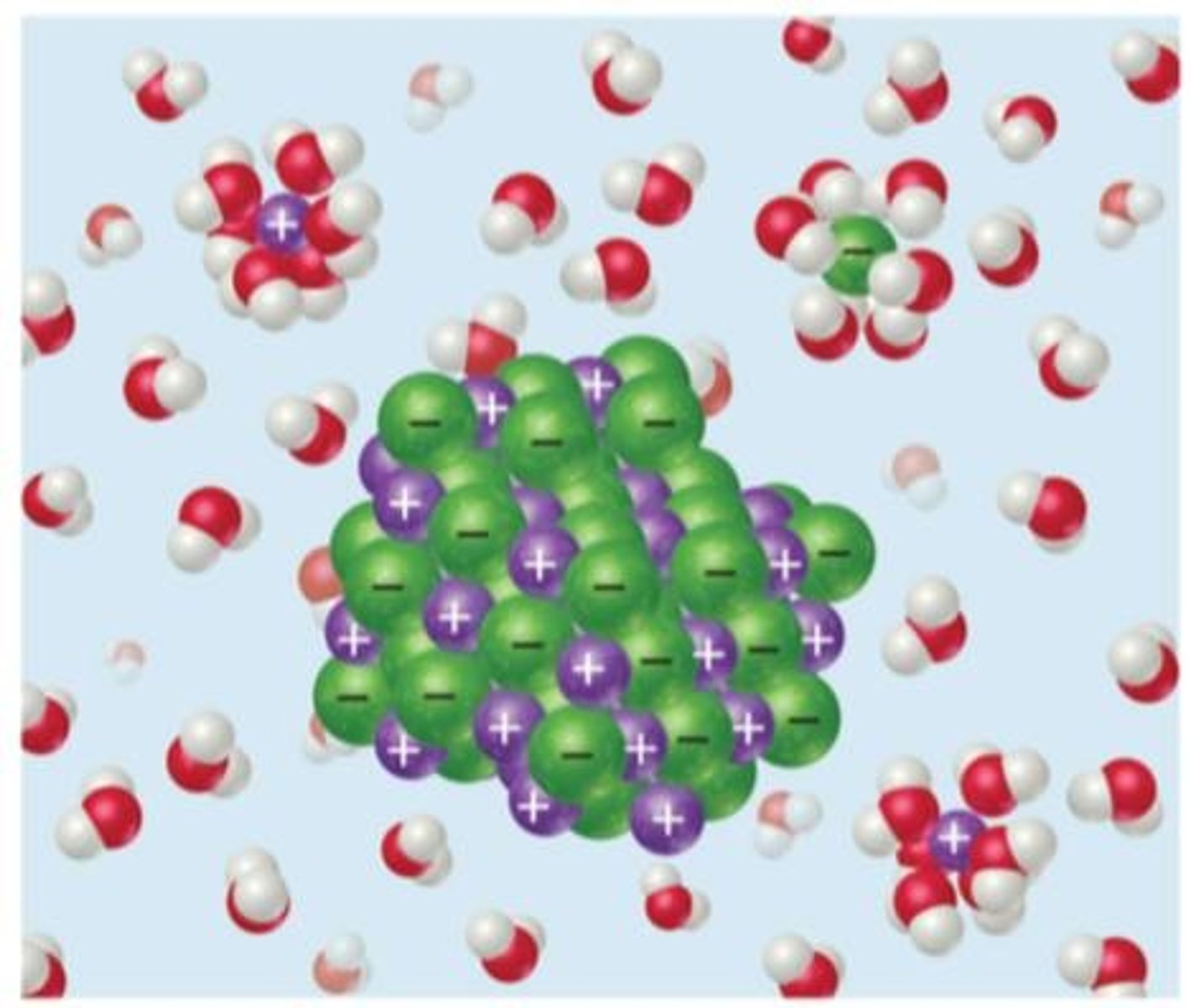
Solute
Substance there is less of in a solution.
Solvent
Substance there is more of in a solution.
Solution
Result of mixing solute and solvent.
Dissolution
A physical change where molecules remain intact when they dissolve.
Ionic compounds
Dissociate to form solvated ions when dissolved.
Covalent bonds
Do not break during dissolution; only intermolecular forces are affected.
Solubility
The ability of a substance to dissolve in another substance.
Miscibility
The ability of substances to mix in any proportions.
Polar molecules
Molecules that will dissolve in other polar molecules.
Nonpolar molecules
Molecules that will dissolve in other nonpolar molecules.
Energy of Mixing
The energy associated with the mixing of solute and solvent.
Entropy
The natural tendency towards mixing of substances.
Enthalpy
The energy of mixing that can affect solubility.
Dispersion forces
Weak forces always present in nonpolar substances.
Dipole-dipole forces
Forces present when both molecules are polar.
H-bonding
Attractive forces between H-bond donors and acceptors.
Ion-dipole forces
Forces between ionic solutes and polar solvents.
Colligative Properties
Changes in physical properties of solutions that depend on solute concentration.
Vapor pressure lowering
A colligative property where the vapor pressure decreases due to dissolved solute.
Boiling point elevation
A colligative property where the boiling point increases due to dissolved solute.
Freezing point depression
A colligative property where the freezing point decreases due to dissolved solute.
Colligative Property
A solution will have lower vapor pressure and increase the boiling point.
Raoult's Law
Pvap of the solution is given by Psolution = Xsolvent•P°solvent.
P°solvent
Vapor pressure of the pure solvent.
Psolution
Vapor pressure of the solution.
Xsolvent
Mole fraction of the solvent.
Decrease in Pvap
∆P = Psolution - P°solvent, proportional to the mole fraction of solute.
∆P
Change in vapor pressure, calculated as ∆P = Xsolute•P°solvent.
Partial Pressures
Partial pressures are proportional to the mole fractions of the species in solution and are additive.
PA
Partial pressure of A in the solution, calculated as PA = CA•P°A.
PB
Partial pressure of B in the solution, calculated as PB = CB•P°B.
Ptotal
Total vapor pressure of the solution, given by Ptotal = PA + PB.
Vapor Pressure of Pure Water
The vapor pressure of pure water at 20 °C is 0.0231 atm.
Mole Fraction of Water
For seawater with a mole fraction of water XH2O = 0.980 at 20 °C, calculate Pvap.
Vapor Pressure at 100 °C
Calculate the vapor pressure of a solution prepared by mixing 1.000 L of ethylene glycol with 1.000 L of water.
Vapor Pressure of Methanol
Methanol has a vapor pressure of 0.81 atm at a given temperature.
Vapor Pressure of Ethanol
Ethanol has a vapor pressure of 0.45 atm at the same temperature.
Partial Pressure of Methanol
Calculate the partial pressure of methanol in a solution prepared from 2 mol methanol and 1 mol ethanol.
Partial Pressure of Ethanol
Calculate the partial pressure of ethanol in a solution prepared from 2 mol methanol and 1 mol ethanol.
Total Vapor Pressure of Solution
Calculate the total vapor pressure of the solution prepared from 2 mol methanol and 1 mol ethanol.
Ideal Solutions
Ideal solutions obey Raoult's Law and hold when solute and solvent have similar-strength interactions.
Vapor Composition and Distillation
A component with a higher vapor pressure will make up a greater composition of the vapors than it does in the solution.
Fractional Distillation
Repeated distillations or evaporation/condensation cycles yield purified liquids.
Boiling-Point Elevation
∆Tb = i•Kb•m, where Tb is raised proportionally to the molality of the solute.
Van't Hoff Factor
i is the Van't Hoff factor, always 1 for molecular solutes.
Molal Concentration of Ethylene Glycol
What molal concentration of ethylene glycol would give an aqueous solution that boils at 105 ºC?
Freezing-Point Depression
∆Tf is lowered (∆Tf = Tf solution - Tf solvent) proportionally to the molality of the solution: ∆Tf = -i•Kf•m
Freezing Point Elevation
The boiling point is raised (∆Tb > 0) and the freezing point is lowered (∆Tf < 0)
Henry's Law
Solubility of gases is usually low, decreases with temperature, and increases with pressure (Sg = khPg)
Van't Hoff Factor, i
The number of dissolved particles per species; for molecular compounds, i = 1 and for electrolytes, i > 1
Osmosis
Across a semipermeable barrier, solvent will move from low to high concentration.
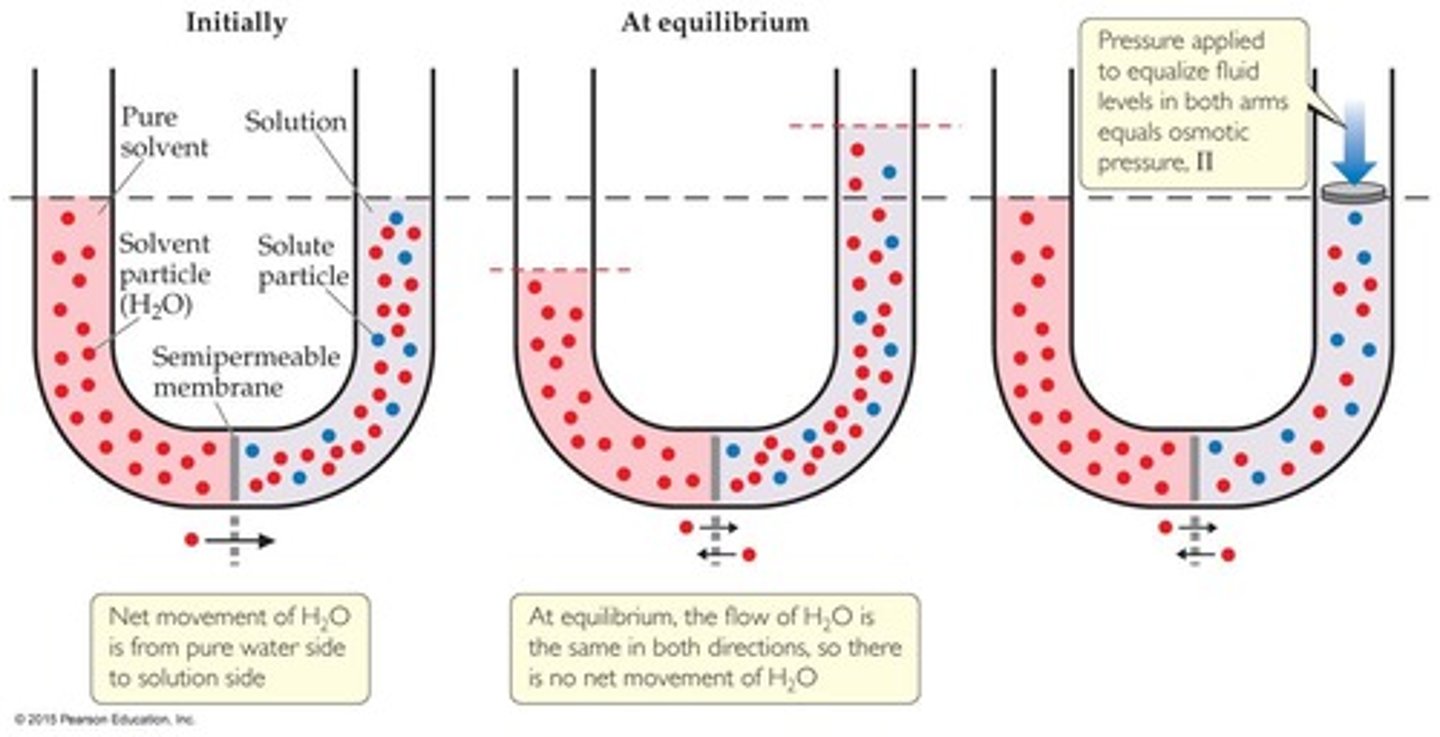
Osmotic Pressure
Movement towards equilibrium acts as a pressure.
Supersaturated Solutions
Solutions with more solute than 'allowed'; if [solute] > [solubility], the extra solute will be eliminated.
Solubility of Gases
Solubility of gases increases with pressure and decreases with temperature.
Molarity of Glycerol Solution
A solution of glycerol in water has a freezing point of -0.56 °C.
Freezing Point of Ethylene Glycol Solution
What is the freezing point of a 2.0 m solution of ethylene glycol in water?
Concentration of Lactic Acid
Two aqueous solutions of equal volume, one containing 20 g of lactic acid (C3H6O3) and one containing 10 g of glucose (C6H12O6) are separated by a semipermeable membrane.
Direction of Water Flow (KNO3 and Glucose)
If a 0.1 M solution of KNO3 and a 0.2 M solution of glucose (C6H12O6) are connected by a semipermeable membrane.
Osmotic Pressure Comparison
Which of the following aqueous solutions will have the highest osmotic pressure?
Types of Solutions
The solute concentration of seawater is approximately 1.15 M.
Isotonic Solutions
Isotonic solutions for red blood cells have the same osmotic pressure as 5.0% m/v glucose.
Hypertonic Solutions
In hypertonic solutions (higher [solute] than in the cell), cells shrink (crenation).
![<p>In hypertonic solutions (higher [solute] than in the cell), cells shrink (crenation).</p>](https://knowt-user-attachments.s3.amazonaws.com/909abc9d-4f67-4060-a085-dc1bee1a3ee5.jpg)
Hypotonic Solutions
In hypotonic solutions (lower [solute] than in cell), cells swell until they burst (hemolysis).
Solubility of Solids
Solubility usually increases at higher T.
Crystallization
You can form crystals by cooling a saturated solution.
Equilibrium in Freezing and Melting
At the freezing (melting) point, the rate of freezing equals the rate of melting.
Ionic Species Dissociation
In concentrated solutions, ionic species do not dissociate fully: some ions pair.
Measured vs Expected i
At higher concentrations, imeasured < iexpected.