Applications of Hess's Law
0.0(0)
0.0(0)
Card Sorting
1/5
There's no tags or description
Looks like no tags are added yet.
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
6 Terms
1
New cards
Hess’s Law
energy in a reaction system must be conserved, cannot be created or destroyed
-overall enthalpy change for a reaction is equal, regardless of route taken
2
New cards
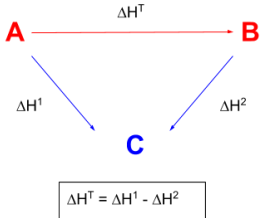
Hess’s Law triangular circle
3
New cards
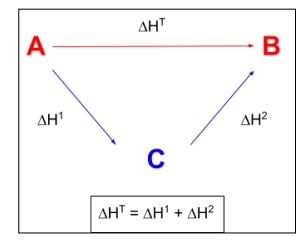
enthalpies of formation - arrows point from central product C

4
New cards
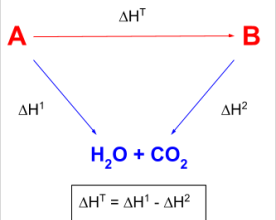
enthalpies of combustion - arrows point towards central product (always H2O + CO2)
5
New cards
bond enthalpy data is average data representing →
energy required to break one mole of the stated bond in a gaseous state, under standard conditions
6
New cards
overall energy change = arrows always point to central product